Self-cleaning pH/thermo-responsive cotton fabric with smart-control and reusable functions for oil/water separation†
Abstract
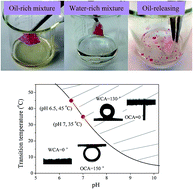
Nowadays, the environmental problem caused by oily wastewater discharge and crude oil leakage has attracted worldwide attention. Recently developed technologies for oil/water separation have been based mainly on materials with special wettability. In this work, a self-cleaning system with smart-control and reusable functions for oil/water separation has been built on cotton fabric via surface-initiated atom transfer radical polymerization (ATRP) of 2-dimethylaminoethyl methacrylate (DMAEMA). The fabric showed high pH- and thermo-responsibility, which was strongly dependent on the pDMAEMA grafting ratio. And by modulating pH or temperature, the fabric switched from superhydrophilic (WCA ∼ 0°) to highly hydrophobic (WCA ∼ 130°), and therefore, were suitable both for the separation of water-rich or oil-rich oil/water mixtures as a type of adsorbent material. The fabric could adsorb oil over 4 times its own weight, and reversibly release it in acidic water, making the fabric easily recyclable.

 Please wait while we load your content...
Please wait while we load your content...