One-pot synthesis of acyloxy carbonyl compounds from ketones using a Pybox–copper(ii) catalyst†
Abstract
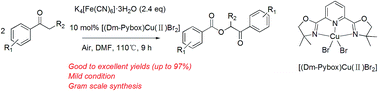
We have described the first efficient one-pot method for the synthesis of acyloxy carbonyl compounds from ketones catalyzed by a Pybox–Cu(II) complex under mild conditions. A series of α-acyloxy ketone products were obtained in good to excellent yields.

 Please wait while we load your content...
Please wait while we load your content...