Constructing nitrogen-doped nanoporous carbon/graphene networks as promising electrode materials for supercapacitive energy storage†
Abstract
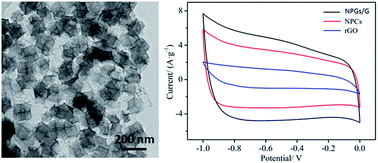
Nitrogen-doped nanoporous carbon/graphene networks (NPC/G) have been prepared by zeolitic imidazolate frameworks (ZIF-8) homogeneously encapsulated in graphene oxide (GO) networks, which were carbonized at high-temperature under a N2 atmosphere and washed to get rid of impurities. The obtained NPC/G composites are highly graphitizing and exhibit a high specific surface area up to 703 m2 g−1. By optimizing the annealing conditions, the nanoporous frameworks derived from ZIF-8 can be effectively maintained, which are beneficial for electrolyte ion adsorption and transportation. Furthermore, the NPC/G composites are used as electrode materials for supercapacitive energy storage and show a high specific capacitance of 235 F g−1 at a current density of 1 A g−1, which is larger than that of NPCs and rGO. This provides a new route for designing novel structures to improve electrochemical performance. More interestingly, these electrode materials also present excellent rate capability and 85% retention of its initial capacitance after 1000 cycles in 1 M KOH aqueous solution.

 Please wait while we load your content...
Please wait while we load your content...