Dealuminated BEA zeolite for selective synthesis of five-membered cyclic acetal from glycerol under ambient conditions
N. J. Venkatesha*,
Y. S. Bhat and
B. S. Jai Prakash
Chemistry Research Centre, Bangalore Institute of Technology, K. R. Road, Bangalore 560 004, India. E-mail: venkatesha.312@gmail.com; Tel: +91 9901003120
First published on 9th February 2016
Abstract
BEA zeolite was treated with phenoldisulfonic acid (PDSA) of different concentrations from 0.1 M to 1.0 M to obtain dealuminated zeolites. The treated BEA samples were characterized by XRD, 27Al-MAS NMR, SEM-EDX, TPD NH3, pyridine-FTIR and BET measurements. Modified zeolites showed an increase in surface area and pore volume and a decrease in acid amount with an increase in PDSA concentration. An untreated BEA sample catalyzed the reaction between glycerol and acetone to form glycerol acteals to yield two products, five-membered dioxalane and six-membered dioxane. Under optimized reaction conditions, the untreated BEA sample showed 83% dioxalane and 17% dioxane while the modified samples showed an increase in dioxalane selectivity with an increase in the acid concentration used for the treatment. The 1 M PDSA treated sample exhibited 100% selectivity for dioxalane. This is attributed to an increase in pore volume and decrease in acid amount of the modified BEA samples with an increase in PDSA concentration. The combined effect of pore volume and acidity in determining dioxalane selectivity is discussed.
1. Introduction
The condensation of glycerol with aldehydes or ketones gives a mixture of cyclic acetal products, 1,3 dioxalane, a 5-membered solketal and 1,3 dioxane, a 6-membered solketal with an excess of one product or approximately in equimolar amounts.1,2 This phenomenon seems to be related to the nearly identical reactivity of the primary and secondary hydroxyl group in glycerol and to similar tendencies for the formation of the 5- and 6-membered ketals. Only by changing their physical properties by converting them into their derivatives, is isolation of one or both of the acetals on a large scale feasible.3 However, this procedure is disadvantageous because of expensive purification steps which result in considerable loss of the target product.3–5Selective protection of carbonyl groups by glycerol during the manipulation of multifunctional organic molecules6 is employed in production of fragrances, food and beverage additives, pharmaceuticals, detergents, lacquers and also as fuel additives by blending with petrol to reduce the cost and enhance efficiency by controlling the knocking.6–10
The general method for acetal synthesis involves reaction of carbonyl compounds with an alcohol or an orthoester in the presence of protic or Lewis acid catalysts. Protic acids such as pTSA, HCl, H3PO4, H2SO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 and Lewis acids such as AlCl3, FeCl3, ZnCl2, SnCl2
11 and Lewis acids such as AlCl3, FeCl3, ZnCl2, SnCl2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12,13 are largely used in the homogeneous medium for acetal synthesis. These acids, however, have limitations which include tedious work-up procedure and production of undesired wastes in addition to their costs. Heterogeneous acid catalysts have obvious advantages over homogeneous acids such as their easy separation, product selectivity, solventless conditions and reusability.13,14 There are several reports available on the synthesis of glycerol acetals by different carbonyl compounds like formaldehyde, cinnamaldehyde, benzaldehyde, furfural, acetone, benzophenone, cycloheaxanone1,7,9,14–16 using various catalysts such as, Amberlyst, MCM, Nafion-H1, alumina,17 montmorillonite,18,19 zeolites,9,20–22 mesoporous aluminosilicates,23,24 ion exchange resins,12,25 [Cp*IrCl2]2, Lu(OTf)3
12,13 are largely used in the homogeneous medium for acetal synthesis. These acids, however, have limitations which include tedious work-up procedure and production of undesired wastes in addition to their costs. Heterogeneous acid catalysts have obvious advantages over homogeneous acids such as their easy separation, product selectivity, solventless conditions and reusability.13,14 There are several reports available on the synthesis of glycerol acetals by different carbonyl compounds like formaldehyde, cinnamaldehyde, benzaldehyde, furfural, acetone, benzophenone, cycloheaxanone1,7,9,14–16 using various catalysts such as, Amberlyst, MCM, Nafion-H1, alumina,17 montmorillonite,18,19 zeolites,9,20–22 mesoporous aluminosilicates,23,24 ion exchange resins,12,25 [Cp*IrCl2]2, Lu(OTf)3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 26 and supported silica.27–29
26 and supported silica.27–29
Modified zeolites have been used as heterogeneous acid catalysts for organic synthesis. Post synthesis modification of zeolites is commonly practiced to vary their catalytic activity. Dealumination of zeolites is an effective post synthesis method of varying the Si to Al ratio (SAR) and acidity. Mineral acids such as HCl, HNO3, H2SO4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30,31 and organic acids like pTSA,32 citric acid, oxalic acid, acetic acid and dicarboxylic acid33–36 have been used as dealuminating agents for zeolite samples. When treated with mineral acids, dealumination is caused by the acid hydrolysis of Al–O–Si bonds whereas organic acids essentially remove Al by complexation.32–36 In both cases, post modification results in a zeolite having steady catalytic performance for a long period in acid catalyzed reactions such as alkylation, acylation, esterification and hexane cracking reactions.32–36
30,31 and organic acids like pTSA,32 citric acid, oxalic acid, acetic acid and dicarboxylic acid33–36 have been used as dealuminating agents for zeolite samples. When treated with mineral acids, dealumination is caused by the acid hydrolysis of Al–O–Si bonds whereas organic acids essentially remove Al by complexation.32–36 In both cases, post modification results in a zeolite having steady catalytic performance for a long period in acid catalyzed reactions such as alkylation, acylation, esterification and hexane cracking reactions.32–36
In the recent years, microwave heating is used in the post synthesis modification like dealumination of zeolite. Microwave heating brings about the dealumination considerably faster. Apart from saving energy, it also results in more pronounced effect on the surface and acidic properties compared to conventional heating. Thus, use of microwave in the zeolite modification is a valuable green chemistry process.32,37,38
In the present study, dealumination of BEA zeolite sample was brought about by treatment with organic acids such as phenoldisulfonic acid (PDSA), methanesulfonic acid (MSA) and para-toluenesulfonic acid (pTSA). The objective was to investigate the catalytic activity of the dealuminated samples for the reaction between glycerol and acetone to form glycerol acetals. Selectivity aspect of five and six membered cyclic acetals formed as a result of dealumination is also examined.
2. Experimental
2.1 Materials and methods
The BEA zeolite sample having Si to Al ratio (SAR) 25 was obtained from Süd chemie, Vadodara, Gujarat, India. Commercial zeolite obtained is in Na+ form, which was further converted into H+ form by ion exchanging with 1 M NH4NO3 solution followed by calcination at 540 °C. MSA and pTSA were obtained from SD fine chemicals, PDSA was procured from Loba chemicals, glycerol was supplied by Merck chemicals and acetone was received from CDH chemicals. Glycerol formal [mixture of dioxalane (64%) and dioxane (36%)] and dioxalane were procured from Sigma Aldrich.2.2 Catalyst preparation
The modification of zeolite samples by organic acid treatment was performed under microwave irradiation. Milestone, “START-S” microwave lab station for synthesis, Italy has been used. This has a facility to control reaction temperature with the aid of infrared sensor by regulating microwave power output has been used. Variable power up to 1200 W was applied by microprocessor-controlled single-magnetron system.Acid treatment of zeolites was performed by following the procedure reported elsewhere.32 PDSA solution of different concentrations (0.1 M, 0.25 M, 0.5 M and 1.0 M) were prepared using deionized water in 100 cm3 volumetric flasks. BEA zeolite samples (10 g each, H-form) was separately dispersed with continuous stirring in PDSA solutions of different concentrations. The suspensions were subjected to refluxing under microwave irradiation for 30 min initially by applying 800 W of energy to reach 110 °C. Then the same temperature was maintained for 29 min by applying the energy in the range of 200–450 W. After that the mixture was cooled to ambient temperature, centrifuged and repeatedly washed to remove the excess of acid from the sample. The centrifuged solid sample was dried at 120 °C, finely ground and subjected to calcination at 540 °C for 6 hours. Similarly, BEA was treated with 1 M each of pTSA and MSA. The samples were designated as 0.1PDSABEA, 0.25PDSABEA, 0.5PDSABEA, 1.0PDSABEA, 1.0pTSABEA and 1.0 M SABEA.
2.3 Characterization
Different acid treated zeolite samples were characterized using various techniques such as pyridine adsorbed by FTIR, NH3-TPD, 27Al MAS NMR, powder XRD, SEM-EDX and BET analyses.Acidity of samples was recorded before and after organic acid treatment of the zeolite. All the samples were activated by degassing at 150 °C for 2 hours, then cooled under vacuum and saturated with liquid pyridine for 1 hour. Samples were again heated to 110 °C to remove physisorbed pyridine. Spectra of the samples were recorded by KBr pellet technique using Shimadzu, IRAffinity-1 instrument with 4 cm−1 resolution of 40 scans in the wavenumber range 1700–1400 cm−1.
Surface area and pore characteristics of the parent and modified zeolite samples were characterized under liquid nitrogen temperature using Quanta Chrome Nova-1000 surface analyzer instrument. BET and de Boer t-plot methods were used to determine surface area and pore volume.
Structural integrity of the catalyst samples was checked by powder XRD. The data were recorded by step scanning at 2θ = 0.020° per second from 3° to 80° on PANalytical X'Pert PRO MPD X-ray diffractometer with graphite monochromatized Cu Kα radiation (λ = 0.15406 nm).
27Al MAS NMR spectra of untreated and dealuminated BEA zeolites were recorded using Joel-ECX instrument having 8 mm rotor of speed 10k.
Morphology and Si/Al ratio of BEA and acid treated zeolite samples were recorded using ZEISS ULTRA-55 Field Emission Scanning Electron Microscope (FESEM) having Energy Dispersive X-ray Analysis (EDXA) under high vacuum mode.
Strength of the zeolite acid sites was evaluated by NH3-TPD method. Typically it involved around 0.1 g of sample, which was placed in a U-shaped quartz sample tube. Prior to the TPD measurement, the catalyst was pretreated with He at 120 °C for 2 h. A mixture of NH3 in He (10%) was passed at the rate of 30 cm3 min−1 at 150 °C for 30 min. The sample was then flushed with He (30 cm3 min−1) at 150 °C for 1 h. The TPD measurements were carried out in the range of 100–600 °C at a heating rate of 10 °C min−1. Ammonia concentration in the effluent was measured with a gold-plated filament thermal conductivity detector.
Molecular dimensions of the reactant and product molecules were calculated using Chem Bio 3D (version 7) and Gaussian software theoretically.
2.4 Catalytic activity tests
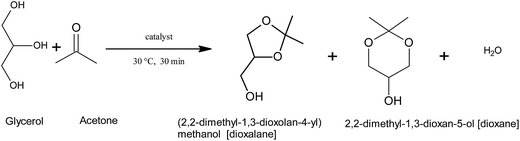
Ten mmol each of glycerol and acetone were mixed with 0.5 g catalyst sample in 50 ml reaction vessel and stirred on a magnetic stirrer for 30 min. Reaction temperature was maintained using a thermostat. After the reaction, the mixture was extracted by stirring with 4 ml of n-pentanol for 5 minutes and filtered to remove the catalyst. The reaction mixture was analyzed using Chemito GC-1000 gas chromatograph with TR-Wax capillary column and flame ionization detector. The cyclic acetal products [5-membered cyclic ketal (2,2-dimethyl-1,3-dioxolan-4-yl) methanol simply called dioxalane and 6-membered cyclic ketal (2,2-dimethyl-1,3-dioxan-5-ol)] simply called dioxane were analyzed using n-heptane as internal standard. The products were also confirmed by GC-MS analysis. The conversion and selectivity of products were quantified with respect to standard samples of dioxalane, glycerol formal and glycerol. Reaction occurring is shown in Scheme 1.3. Results and discussion
Table 1 gives the surface characteristics and acidity of zeolite samples before and after modification with organic sulfonic acids. BEA zeolite having SAR 25 was used for the experiments. Before modification, it possessed a measured surface area of 538 m2 g−1. Surface area and pore volume of BEA zeolite, before and after modification, are also presented in Table 1. Three different organic sulfonic acids such as pTSA, MSA and PDSA were used for modification. All the three organic acids caused removal of structural Al accompanied by increase in surface area and pore volume; the changes observed was in the sequence PDSA > MSA > pTSA.| Catalyst samples | Si/Al ratio | Surface area, SBET (m2 g−1) | Pore volume, V (cm3 g−1) | Average pore diameter (Å) | Pyridine FT-IR acidity mmol g−1 | |
|---|---|---|---|---|---|---|
| B | L | |||||
| a Standard deviation values: Si/Al-±1%, SBET-±2.1%, V-±0.35%, average pore diameter-±1.2%, pyridine FT-IR acidity-±0.42%, Si/Al ratio was calculated using SEM-EDX analysis. | ||||||
| BEA | 25 | 538 | 0.307 | 31.04 | 0.644 | 0.394 |
| 1.0pTSABEA | 48 | 579 | 0.364 | 30.9 | 0.626 | 0.372 |
| 1.0 M SABEA | 69 | 628 | 0.388 | 30.01 | 0.602 | 0.366 |
| 1.0PDSABEA | 96 | 674 | 0.412 | 29.24 | 0.531 | 0.347 |
3.1 Acidity
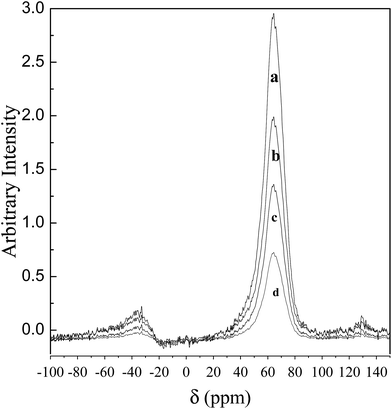
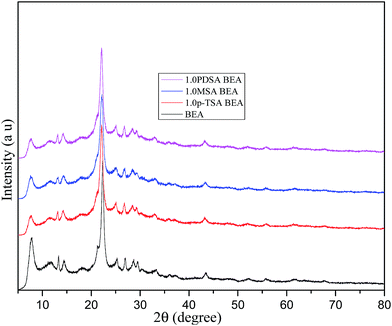
Acidity in zeolite is mainly due to isomorphous substitution of aluminum, generating a negative charge on the framework. This charge is compensated by H+ ions in H-form of BEA and is responsible for Bronsted acidity. Aluminum present on the framework edges is the Lewis acid site. Organic sulfonic acid treatment of the zeolite removes aluminum from the framework and also from edges of the framework. This causes decrease in both Bronsted and Lewis acidity. The changes in the Bronsted (B) and Lewis (L) acidities of different organic sulfonic acid treated BEA samples were measured by the pyridine adsorption using FTIR method reported elsewhere.39,40 The results are given in Table 1 and the pyridine-FTIR patterns are shown in Fig. 1.Both the methods of acidity measurement showed a decrease in the acid amounts of BEA on treatment with organic acids. The decrease in the Bronsted and Lewis acid amounts due to dealumination was found to be in the order PDSA > MSA > pTSA. Different organic acids have the ability to remove structural (tetrahedral) Al to different extents thereby altering the Si/Al ratio of the treated BEA zeolite. PDSA is able to remove more Al than the other two acids. This is confirmed by decrease in the intensity of the band at 60 ppm in the 27Al MAS NMR spectra corresponding to the tetrahedral aluminum present in the zeolite framework (Fig. 2). From the 27Al MAS NMR spectra it can be seen that the decrease in the Al content of the organic acid treated zeolite follows the order PDSA > MSA > pTSA. Consequently, among the organic acid treated zeolites, PDSA treated zeolite shows the lowest acid amounts and pTSA treated zeolite shows the highest acid amount. Al removal from the zeolite structure is expected to cause enhancement in surface area and pore volume. This is supported by the fact that the increase in surface area, pore volume and Si/Al ratio for the treated zeolites is in the order pTSA < MSA < PDSA (Table 1). Also there is no change in structural integrity of the acid treated BEA samples, as evident from the XRD patterns (Fig. 3). Morphology of zeolite samples, as shown by FESEM images, does not show any major change upon organic acid treatment (Fig. 4).
3.2 Reaction parameter studies with untreated BEA
The effect of different reaction parameters such as temperature, catalyst amount, mole ratio and reaction time on conversion and selectivity was carried out in the presence of untreated BEA catalyst. Each experiment was performed three times and the mean of the obtained values was considered for the calculation of standard deviation. Internal and external diffusion effects were calculated under similar experimental conditions using Mear criteria (3.54 × 10−7) for external diffusion and Weisz–Prater (2.984 × 10−7) and Koros–Nowak criteria for internal diffusion. Both the diffusion effects are found to be negligible. The results of variation of reaction parameters on catalytic activity are discussed in the ensuing sections.3.3 Comparison with dealuminated BEA samples
Under similar reaction conditions catalytic reactions using organic sulfonic acid treated BEA samples were carried out to study their behavior in catalytic activity and selectivity in acetalization of glycerol with acetone. Untreated BEA sample showed 76% conversion with selectivity 83% for dioxalane and 17% for dioxane. Conversion and selectivity of products were found to be higher in modified BEAs (Table 2). 1.0 M SABEA and 1.0pTSABEA showed almost the same conversion and selectivity. 1.0PDSABEA however showed 100% selectivity for the dioxalane with no formation of dioxane. This is perhaps due to the rearrangement of dioxane to dioxalane over 1.0PDSABEA.| Catalyst samples | % conversion of glycerol | % selectivity of dioxalane | % selectivity of dioxane | Amount of NH3-desorbed (mmol) | Total acidity (mmol) | |
|---|---|---|---|---|---|---|
| T1 | T2 | |||||
a Reaction conditions: time; 30 min, temperature; 30 °C, catalyst amount; 0.5 g, mole ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 of glycerol and acetone. T1 = temperature range 100–300 °C, T2 = temperature range 325–550 °C. Standard deviation values: glycerol conversion-±1.6%, dioxalane selectivity-±2.1%, dioxane-±1.35%, amount of NH3 desorbed-±0.52%. 1 of glycerol and acetone. T1 = temperature range 100–300 °C, T2 = temperature range 325–550 °C. Standard deviation values: glycerol conversion-±1.6%, dioxalane selectivity-±2.1%, dioxane-±1.35%, amount of NH3 desorbed-±0.52%. |
||||||
| BEA | 76 | 83 | 17 | 0.64 | 0.47 | 1.11 |
| 1.0pTSABEA | 82 | 90 | 10 | 0.45 | 0.51 | 0.96 |
| 1.0 M SABEA | 84 | 93 | 07 | 0.29 | 0.58 | 0.87 |
| 1.0PDSABEA | 80 | 100 | 00 | 0.19 | 0.49 | 0.68 |
Catalytic acetalisation reaction mainly occurs on the internal surface (pores) of the zeolite. Molecular dimensions of the dioxane and dioxalane computed theoretically are given below (Fig. 8). It can be seen that dimensions of both reactant and product molecules are smaller than internal pore dimensions. Further, most of the active sites are located within the pores.
Generally glycerol acetalization reaction is a Bronsted acid catalyzed reaction but also can take place to a lesser extent in the presence of a Lewis acid. The rearrangement of dioxane to dioxalane however, can take place only in the presence of Bronsted acid sites. Accordingly, selectivity is only dependent on the Bronsted acid sites.
Strength of the acid sites was studied by using NH3-TPD method. Desorption of ammonia at different temperatures could be seen in the TPD profile (Fig. 9). It indicates desorption of ammonia at lower temperature range (T1 = 100–300 °C) corresponding to weak acid sites and desorption at higher temperature (T2 = 325–550 °C) corresponding to the stronger acid sites. The amounts of NH3 desorbed are given in Table 2.
On treatment with organic acids, BEA zeolite showed changes in strength of the acid sites – a decrease in weaker acid sites and a moderate increase in stronger acid sites (Fig. 9). Increase in strength of the acid sites enhanced the selectivity of dioxalane from dioxane with insignificant change in conversion of glycerol in the acetalization reaction.
3.4 Selective dioxalane formation
Catalytic activity with 1.0PDSABEA was carried out by varying reaction conditions such as temperature, catalyst amount and reaction time. The results obtained are shown in Tables 3–5. Though the glycerol conversion varied under different conditions, 1.0PDSABEA sample gave dioxalane as the single product. This shows that only catalyst characteristics are responsible for the selectivity and not the reaction conditions. Further studies were made with the catalyst samples dealuminated with 0.1 M to 0.5 M PDSA. This was to investigate the changes in acid amounts and pore volumes that arise upon acid treatment and their relation with dioxalane selectivity.| Catalyst amount (g) | % conversion of glycerol | Selectivity of dioxalane | Selectivity of dioxane |
|---|---|---|---|
a Reaction conditions: time; 30 min, temperature; 30 °C, mole ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 glycerol: acetone. Standard deviation values: glycerol conversion-±1.8%, dioxalane selectivity-±0.15%, dioxane-±0.2%. 1 glycerol: acetone. Standard deviation values: glycerol conversion-±1.8%, dioxalane selectivity-±0.15%, dioxane-±0.2%. |
|||
| 0.1 | 35 | 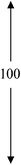 |
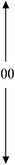 |
| 0.2 | 48 | ||
| 0.3 | 62 | ||
| 0.4 | 76 | ||
| 0.5 | 81 | ||
| 0.6 | 82 | ||
| 0.7 | 84 | ||
| 0.8 | 85 | ||
| 0.9 | 85 | ||
| 1.0 | 86 | ||
| Temperature (°C) | % conversion of glycerol | Selectivity of dioxalane | Selectivity of dioxane |
|---|---|---|---|
a Reaction conditions: time; 30 min, catalyst; 0.5 g, mole ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 glycerol: acetone. Standard deviation values: glycerol conversion-±1.48%, dioxalane selectivity-±0.09%, dioxane-±0.17%. 1 glycerol: acetone. Standard deviation values: glycerol conversion-±1.48%, dioxalane selectivity-±0.09%, dioxane-±0.17%. |
|||
| 30 | 56 | 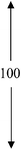 |
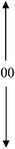 |
| 35 | 66 | ||
| 40 | 71 | ||
| 45 | 76 | ||
| 50 | 80 | ||
| 55 | 82 | ||
| 60 | 84 | ||
| 70 | 85 | ||
| 80 | 85 | ||
| Reaction time (min) | % conversion of glycerol | Selectivity of dioxalane | Selectivity of dioxane |
|---|---|---|---|
a Reaction conditions: catalyst; 0.5 g, temperature; 30 °C, mole ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 glycerol: acetone. Standard deviation values: glycerol conversion-±1.84%, dioxalane selectivity-±0.16%, dioxane-±0.20%. 1 glycerol: acetone. Standard deviation values: glycerol conversion-±1.84%, dioxalane selectivity-±0.16%, dioxane-±0.20%. |
|||
| 5 | 39 |  |
 |
| 10 | 48 | ||
| 20 | 62 | ||
| 30 | 76 | ||
| 40 | 80 | ||
| 50 | 78 | ||
| 60 | 80 | ||
| 90 | 82 | ||
| 120 | 84 | ||
3.5 Reusability of the samples
The reusability of the catalyst samples was checked for four cycles. The results (Table 6) are similar to fresh catalyst samples indicating negligible changes in catalyst characteristics after use in the reaction. Further catalyst samples were recovered after the reaction and subjected to pyridine FT-IR, XRD and BET analysis. These results do not show any significant change in catalyst characteristics before and after use in the reaction indicating there is no deactivation of zeolite sample.| Cycles | % conversion of glycerol | % selectivity of dioxalane | % selectivity of dioxane | Surface area (m2 g−1) | Pore volume (cm3 g−1) | Pyridine FT-IR acidity (mmol g−1) |
|---|---|---|---|---|---|---|
a Reaction conditions: time; 30 min, temperature; 30 °C, catalyst amount; 0.5 g, mole ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 of glycerol and acetone. Standard deviation values: glycerol conversion-±2%, dioxalane selectivity-±0.2%, dioxane-±0.25%, SBET-±2.4%, V-±0.28%, pyridine FT-IR acidity-±0.38%. 1 of glycerol and acetone. Standard deviation values: glycerol conversion-±2%, dioxalane selectivity-±0.2%, dioxane-±0.25%, SBET-±2.4%, V-±0.28%, pyridine FT-IR acidity-±0.38%. |
||||||
| 1st | 80 | 100 | 00 | 673 | 0.410 | 0.875 |
| 2nd | 78 | 100 | 00 | 671 | 0.409 | 0.869 |
| 3rd | 79 | 100 | 00 | 672 | 0.411 | 0.872 |
| 4th | 77 | 100 | 00 | 670 | 0.410 | 0.870 |
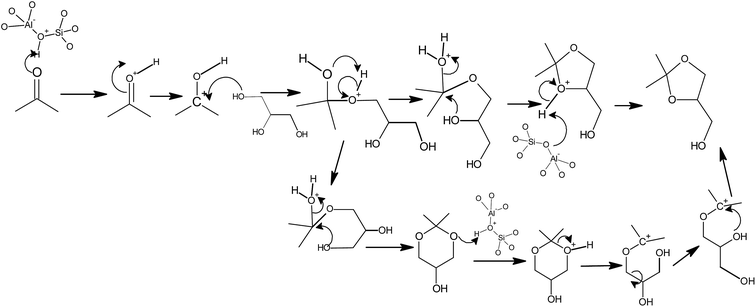
The possible reaction mechanism for dioxalane formation is shown in Scheme 2. The mechanism of formation of glycerol acetals involves protonation by Bronsted acid site or Lewis acid coordination to carbonyl oxygen of acetone generating positive charge on the carbonyl carbon (Scheme 2). The carbocation thus formed gets attacked readily by nucleophilic hydroxyl group of glycerol giving hemi acetal. This is further attacked by another hydroxyl group resulting in cyclic acetal with the elimination of a molecule of water. When 1,2 hydroxy groups are involved dioxalane [5-membered] is formed and dioxane [6-membered] is formed when 1,3 hydroxy groups are involved. Further, in the presence of Bronsted or Lewis acid sites, dioxane gets rearranged to dioxalane.
3.6 Role of pore volume and acidity in dioxalane selectivity
The selectivity for dioxalane and dioxane in presence of untreated BEA and selectivity for those treated with different concentrations of PDSA are shown in Table 7. It reveals that the selectivity for the dioxalane increased with the increase in PDSA concentration from 0.1 M to 1.0 M. The selectivity for dioxalane with 0.1PDSABEA was the same as that of untreated BEA catalyst (83%) but the selectivity increased to 100% with 1.0PDSABEA. Since this increase of 17% has significance with respect to mechanism, its relation with changes in various parameters observed in the catalyst samples upon acid treatment was studied.| Catalyst samples | Conversion of glycerol (%) | % selectivity of dioxalane | % selectivity of dioxane | Si/Al ratio | Acidity(Pyr-FTIR) (A) mmol g−1 | Pore volume (V) cm3 g−1 | Surface area (SBET) m2 g−1 |
|---|---|---|---|---|---|---|---|
a Reaction conditions: time, 30 min; temperature, 30 °C; catalyst amount, 0.5 g; glycerol and acetone mole ratio, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Standard deviation values: glycerol conversion-±1.6%, dioxalane selectivity-±2.1%, dioxane-±1.35%, Si/Al-±1%. 1. Standard deviation values: glycerol conversion-±1.6%, dioxalane selectivity-±2.1%, dioxane-±1.35%, Si/Al-±1%. |
|||||||
| 0.1PDSABEA | 72 | 83 | 17 | 48 | 0.992 | 0.345 | 556 |
| 0.25PDSABEA | 75 | 86 | 14 | 63 | 0.953 | 0.364 | 589 |
| 0.5PDSABEA | 76 | 95 | 05 | 84 | 0.928 | 0.392 | 635 |
| 1.0PDSA-BEA | 80 | 100 | 00 | 96 | 0.878 | 0.412 | 674 |
| R | — | — | — | — | −0.963 | 0.994 | 0.993 |
It could be seen from Table 7 that the catalyst properties that change on increase in the acid concentration used for treatment are surface area, pore volume and acidity. Increase in pore volume is evidently due to the removal of structural Al causing decrease in acid amount (Table 7). Acidity (A) was calculated by summing up the Bronsted and Lewis acidities by the pyridine-FTIR method. In order to know whether the change in pore volume and acidity are responsible for the increase in dioxalane selectivity, these characteristics were correlated with the increase in selectivity. The correlation coefficients are given in Table 7. The correlation coefficient (r) values show good correlation for the dioxalane selectivity with all the parameters. Surface area and pore volume showed very nearly the same r value. This is expected because these two parameters are caused due to dealumination and therefore there exists a good proportionality between surface area and pore volume. The acidity showed a decreasing trend with a simultaneous increase in pore volume. Since both pore volume and acidity appear to be responsible, some combination of both have to be considered in explaining the rearrangement of dioxane leading to 100% dioxalane selectivity.
3.7 Volume space acidity (VSA)
Increase in pore volume (by about 25%) on organic acid treatment is caused by the removal of framework Al thereby diminishing the acid amount (by about 14%) in the enhanced pore volume. A combination of both these factors, that is, acidity and pore volume, apparently determines the rearrangement of dioxane. Extent of rearrangement is dependent on the progressive removal of Al which causes decrease in acid amounts and increase in pore volume. The combination of both these factors is expressed as volume space acidity factor (VSA). VSA, described as the product of pore volume (V) and inverse acidity (A−1), provides a measure of volume space for each acid site available for the dioxane molecule to orient and undergo rearrangement.In the present work, the rearrangement of the dioxane to dioxalane is facilitated by the pore volume to approach the acid sites in such a way that the molecule readily orients to rearrange itself. With the increase in acid concentration used for treatment, the pore volume increases with proportional decrease in the acid amounts (or increase in VSA) till it reaches an optimum value to achieve 100% dioxalane selectivity with 1.0PDSABEA catalyst (Fig. 10). Correlation coefficient (r) between VSA and the dioxalane selectivity for catalysts treated with different acid concentrations also showed a high correlation (r = 0.989). Thus, combination of increase in pore volume and decrease in acid amounts favoring the rearrangement of the dioxane to dioxalane is probable.
 | ||
Fig. 10 Representation of relation between VSA and selectivity of dioxalane. Reaction conditions: time, 30 min; temperature, 30 °C; catalyst amount, 0.5 g; glycerol and acetone mole ratio, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. 1. | ||
3.8 Acidity, hydrophobicity and dioxalane selectivity
Increase in acid amounts is known to cause the hydrolysis of dioxane/dioxalane favoring the reverse reaction giving back the reactant molecules.20,26,29,41–45 On the contrary, decrease in framework Al brings down the acidity and consequently hydrolysis of the dioxane/dioxalane is reduced. Furthermore, Al removal causes increased hydrophobicity in the zeolite channels. Increased hydrophobicity prevents the dioxane molecules to hydrolyze back into the reactants thereby favoring the rearrangement of the dioxane molecules to dioxalane. This results in 100% selectivity for dioxalane in 1.0PDSABEA. High correlation factor observed between VSA and dioxalane selectivity is not surprising. On the other hand VSA correlation of acid amount of higher strength (as measured by the ammonia desorbed at higher temperature T2) with dioxalane selectivity showed a poor correlation (r = 0.846). It appears that variation of acid strength as measured by TPD-NH3 has no influence on the dioxalane selectivity. And also VSA of average pore diameter when correlated with the selectivity of dioxalane showed r value 0.953.4. Conclusion
Removal of Al from the framework on treatment with PDSA modifies the characteristics of BEA zeolite. Increase in PDSA concentration for treatment results in dealuminated zeolite with gradual increase in surface area and pore volume but decrease in acid amounts. Untreated BEA zeolite catalyzes the reaction between glycerol and acetone to form six-membered dioxane and five membered dioxalane, the maximum selectivity of the latter being 83%. BEA sample treated with 1 M PDSA, however, is found to exhibit complete rearrangement of six-membered dioxane into the five-membered dioxalane. Augmented selectivity for the five membered species is attributed to the increase in pore volume and decreased acid amount. A factor called volume space acidity (VSA) described as the product of pore volume (V) and inverse acidity (A−1), provides a measure of volume space available for molecular rearrangement on each acid site. VSA correlates well with the dioxalane selectivity exhibited by the modified BEA zeolite.Acknowledgements
The authors wish to thank the Rajya Vokkaligara sangha and Principal Bangalore Institute of Technology for the facilities provided. The authors also extend their thanks to Dr Arun Basrur, General Manager (R&D) Sud Chemie, India, for providing zeolite samples,L. V. Narendra NMRC IISc, Bengaluru for MAS NMR, Mr Shanmugam IIT madras for helping in molecular dimension calculation and PPISR for XRD and Mr Hareesh N. JU for NH3-TPD measurements.References
- J. Deutsch, A. Martin and H. Lieske, J. Catal., 2007, 245, 428–435 CrossRef CAS.
- A. J. Showler and P. A. Darley, Chem. Rev., 1967, 67, 427–440 CrossRef CAS PubMed.
- G. Aksnes, P. Albriktsen and P. Juvvik, Acta Chem. Scand., 1965, 19, 920–930 CrossRef CAS.
- P. H. J. Carlsen, K. Soerbye, T. Ulven and K. Aasboe, Acta Chem. Scand., 1996, 50, 185–187 CrossRef CAS.
- S. Bartoli, G. De Nicola and S. Roelens, J. Org. Chem., 2003, 68, 8149–8156 CrossRef CAS PubMed.
- T. W. Green and P. G. M. Wuts, Protective Groups in Organic Synthesis, Wiley, New York, 2nd edn, 1991, vol. 4, p. 212 Search PubMed.
- M. Pagliaro, R. Ciriminna, H. Kimura, M. Rossi and C. Della Pina, Angew. Chem., Int. Ed., 2007, 46, 4434–4439 CrossRef CAS PubMed.
- D. B. Tkatchenko, S. Chambrey, R. Keiski, R. Ligabue, L. Plasseraud, P. Richard and H. Turunen, Catal. Today, 2006, 115, 80–87 CrossRef.
- M. J. Climent, A. Corma and A. Velty, Appl. Catal., A, 2004, 263, 155–161 CrossRef CAS.
- K. Woelfel and T. G. Hartman, ACS Symp. Ser., 1998, 705, 193–194 CrossRef CAS.
- G. Cherkaev, S. A. Timonin, G. F. Yakovleva, L. Shutikova, A. S. Mikhailova and L. D. Shapiro, Chemical Abstract Service, 1987, 1, 337–342 Search PubMed.
- M. J. Ashton, C. Lawrence, J. A. Karlsson, K. A. J. Stuttle, C. G. Newton, B. Y. J. Vacher, S. Webber and M. J. Withnall, J. Med. Chem., 1996, 39, 4888–4896 CrossRef CAS PubMed.
- F. D. L. Menezes, M. D. O. Guimaraes and M. J. da Silva, Ind. Eng. Chem. Res., 2013, 52, 16709–16713 CrossRef CAS.
- H. R. Prakruthi, B. M. Chandrashekara, B. S. JaiPrakash and Y. S. Bhat, Catal. Sci. Technol., 2015, 5, 3667–3674 CAS.
- N. Narkhede and A. Patel, RSC Adv., 2014, 4, 19294–19301 RSC.
- B. L. Wegenhart, S. Liu, M. Thom, D. Stanley and M. M. Abu-Omar, ACS Catal., 2012, 2, 2524–2530 CrossRef CAS.
- T. Vu Moc and P. Maitte, Bull. Soc. Chim. Fr., 1975, 9, 2558–2560 Search PubMed.
- L. T. Shuang, L. S. Hui, L. J. Tai and L. H. Zhang, J. Chem. Res., Synop., 1997, 1, 286–287 Search PubMed.
- M. Csiba, J. Cleophax, A. Loupy, J. Malthete and S. D. Gero, Tetrahedron Lett., 1993, 34, 1787–1790 CrossRef CAS.
- C. X. A. da Silva, V. L. C. Goncalves and C. J. A. Mota, Green Chem., 2009, 11, 38–41 RSC.
- R. Ballini, G. Bosica, B. Frullanti, R. Maggi, G. Sartori and F. Schroer, Tetrahedron Lett., 1998, 39, 1615–1618 CrossRef CAS.
- I. Rodriguez, M. J. Climent, S. Iborra, V. Fornes and A. Corma, J. Catal., 2000, 192, 441–447 CrossRef CAS.
- C. A. Gonzalez, S. De and R. Luque, Catal. Sci. Technol., 2014, 4, 4242–4249 Search PubMed.
- Y. Tanaka, N. Sawamura and M. Iwamoto, Tetrahedron Lett., 1998, 39, 9457–9460 CrossRef CAS.
- A. Corma, L. T. Nemeth, M. Renz and S. Valencia, Nature, 2001, 412, 423–425 CrossRef CAS PubMed.
- A. W. Pierpont, E. R. Batista, R. L. Martin, W. Chen, J. K. Kim, C. B. Hoyt, J. C. Gordon, R. Michalczyk, L. A. Pete and R. Wu, ACS Catal., 2015, 5, 1013–1019 CrossRef CAS.
- P. Ferreira, I. M. Fonseca, A. M. Ramos, J. Vital and J. E. Castanheiro, Appl. Catal., B, 2010, 98, 94–99 CrossRef CAS.
- D. Nandan, P. Sreenivasulu, L. N. S. Konathala, M. Kumar and N. Viswanadham, Microporous Mesoporous Mater., 2013, 179, 182–190 CrossRef CAS.
- G. Vicente, J. A. Melero, G. Morales, M. Paniagua and E. Martın, Green Chem., 2010, 12, 899–907 RSC.
- Z. Chen, Y. Feng, T. Tong and A. Zeng, Appl. Catal., A, 2014, 482, 92–98 CrossRef CAS.
- D. M. Roberge, H. Hausmann and W. F. Holderich, Phys. Chem. Chem. Phys., 2002, 4, 3128–3135 RSC.
- B. M. Chandra Shekara, B. S. Jai Prakash and Y. S. Bhat, ACS Catal., 2011, 1, 193–199 CrossRef CAS.
- D. Zhao, J. Wang and J. Zhang, Catal. Lett., 2008, 126, 188–192 CrossRef CAS.
- R. Srivastava, N. Iwasa, S. I. Fujita and M. Arai, Catal. Lett., 2009, 130, 655–663 CrossRef CAS.
- M. R. Apelian, A. S. Fung, G. J. Kennedy and T. F. Degnan, J. Phys. Chem., 1996, 100, 16577–16583 CrossRef CAS.
- X. Zaiku, C. Qingling, Z. Chengfang, B. Jiaqing and C. Yuhua, J. Phys. Chem. B, 2000, 104, 2853–2859 CrossRef.
- M. D. Gonzalez, Y. Cesteros, P. Salagre, F. Medina and J. E. Sueiras, Microporous Mesoporous Mater., 2009, 118, 341–347 CrossRef CAS.
- M. D. González, Y. Cesteros and P. Salagre, Microporous Mesoporous Mater., 2011, 144, 162–170 CrossRef.
- F. Benaliouche, Y. Boucheffa, P. Ayrault, S. Mignard and P. Magnoux, Microporous Mesoporous Mater., 2008, 111, 80–88 CrossRef CAS.
- J. A. Lercher and A. Jentys, Infrared and Raman spectroscopy for characterizing zeolites, Introduction to Zeolite Science and Practice, Elsevier B. V., 3rd edn, 2007, pp. 436–476 Search PubMed.
- B. U. Shubhangi, V. K. Trupti, V. B. Ankush, P. Renu, C. Jyoti, K. D. Mohan, M. A. Sophie, L. Christine and P. J. Edmond, J. Mol. Catal. A: Chem., 2009, 310, 150–158 CrossRef.
- K. N. Tayade, M. Mishra, K. Munusamy and R. S. Somani, Catal. Sci. Technol., 2015, 5, 2427–2440 CAS.
- J. S. Clarkson, A. J. Walker and M. A. Wood, Org. Process Res. Dev., 2001, 5, 630–635 CrossRef CAS.
- L. Roldan, R. Mallada, J. M. Fraile, J. A. Mayoral and M. Menendez, Asia-Pac. J. Chem. Eng., 2009, 4, 279–284 CrossRef CAS.
- L. Li, T. I. Koranyi, B. F. Sels and P. P. Pescarmona, Green Chem., 2012, 14, 1611–1619 RSC.
| This journal is © The Royal Society of Chemistry 2016 |