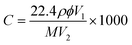Highly sensitive mixed-potential type ethanol sensors based on stabilized zirconia and ZnNb2O6 sensing electrode
Fangmeng Liu,
Xue Yang,
Zhangduo Yu,
Bin Wang,
Yehui Guan,
Xishuang Liang*,
Peng Sun,
Fengmin Liu,
Yuan Gao and
Geyu Lu*
State Key Laboratory on Integrated Optoelectronics, College of Electronic Science and Engineering, Jilin University, 2699 Qianjin Street, Changchun 130012, China. E-mail: liangxs@jlu.edu.cn; lugy@jlu.edu.cn; Fax: +86-431-85167808; Tel: +86-431-85167808
First published on 4th March 2016
Abstract
A mixed-potential type stabilized zirconia (YSZ)-based gas sensor using columbite type composite oxide sensing electrode was developed and fabricated, aiming at sensitive detection of ethanol. Among the different oxide sensing electrodes (SEs) developed, the sensor attached with ZnNb2O6-SE was found to achieve the largest sensitivity to ethanol at 625 °C. Furthermore, the result of the effect of sintering temperature on sensing characteristic showed that the sensor utilizing ZnNb2O6-SE sintered at 1000 °C displayed the highest response of −175 mV to 100 ppm ethanol and a low detection limit of 0.5 ppm at 625 °C. ΔV of the present sensor exhibited a segmentally linear relationship to the logarithm of ethanol concentration in the ranges of 0.5–5 ppm and 5–200 ppm, for which the sensitivities were −29 and −112 mV decade−1, respectively. Moreover, the fabricated device also displayed fast response and recovery times, good repeatability, small fluctuation during 30 days continuous high temperature of 625 °C measured periods, and acceptable selectivity to some other interfering gases. Additionally, the sensing mechanism involving mixed potential was further demonstrated by polarization curves.
1. Introduction
Ethanol processes flammable, volatile characteristics and much lower toxicity and harm to human health relative to methanol. However, ethanol detection is strongly required in many fields, such as drink driving, as a breath marker for specific diseases, and the control of fermentation processes.1,2 Under laboratory conditions, ethanol can conventionally be detected using techniques such as IR, mass spectrometry, or gas chromatography.3,4 Although these methods exhibit advantages in terms of accuracy, selectivity and detection of very low concentration, their application is limited by cost, instrumentation complexity and vast size of instrumentation. Thus, for low-cost, compact and portable application, gas sensors have attracted considerable attention for real-time detection of ethanol.Among various gas sensors, mixed-potential type solid-state electrochemical gas sensors utilizing yttria-stabilized zirconia (YSZ) solid electrolyte and metal oxide sensing electrode (SE) has been developed extensively over the past two decades, and are widely applied for high performance gas sensing detection, such as for NOx, H2, CO, NH3 and VOCs.5–19 The working principle of such sensors is related to a mixed-potential mechanism and has been described in detail by several research groups.10–22 For mixed-potential type gas sensors, the sensing signal (V) is generated at the sensing electrode due to the simultaneous occurrence of electrochemical reactions involving oxygen and the target gas. Thus, the selection of a suitable SE material is one of the most important points to achieve high sensing performance.
In this work, various columbite type composite oxide materials (ZnNb2O6, NiNb2O6 and CoNb2O6) were developed via a facile sol–gel method. For the first time, three kinds of oxide-SEs were used to fabricate planar mixed-potential type YSZ-based gas sensors for sensitive detection of ethanol and the corresponding sensing performances were examined. Among oxide-SEs developed, the sensor attached with ZnNb2O6-SE gave the highest sensitivity to ethanol at 625 °C. The effect of sintering temperature of ZnNb2O6-SE on sensing property was studied. Additionally, the gas sensing characteristics of the present devices to ethanol were systematically investigated and the sensing mechanism was also discussed.
2. Experimental
2.1 Preparation and characterization of columbite type sensing electrode material
ZnNb2O6 was synthesized via a sol–gel method from zinc nitrate hexahydrate (Zn(NO3)2·6H2O), niobium oxide (Nb2O5), HF acid (40%), aqueous ammonia (NH3·H2O), citric acid (CA), and ammonium nitrate (NH4NO3). All of reagents were of analytical grade which were used without further purification and provided by Sinopharm Corp., China. In a typical synthesis process, 3 mmol Nb2O5 was dissolved in a certain amount of HF acid after heating in a water-bath at 80 °C. Then, aqueous ammonia solution was dropwise added to the above solution until the pH value reached up to 9 to obtain Nb2O5·nH2O precipitate under continuous stirring. The precipitate was filtered off, washed, and dissolved in citric acid aqueous solution and stirred at 80 °C for 2 h. Then, stoichiometric Zn(NO3)2·6H2O and NH4NO3 (the molar ratio of Zn/Nb/NH4NO3 = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12) were added into this solution and then stirred at 80 °C until a gel was obtained. The resultant gel was maintained at 80 °C for 24 h in a vacuum drying oven. The MNb2O6 (M = Co and Ni) materials were prepared by the same method according to the procedure described above. The precursor gel was then introduced into a muffle furnace and sintered at 1000 °C for 2 h to obtain the target products, respectively. Additionally, in order to investigate the effect of sintering temperature on sensing performance, ZnNb2O6 was sintered at 800, 1000 and 1200 °C for 2 h, respectively.
12) were added into this solution and then stirred at 80 °C until a gel was obtained. The resultant gel was maintained at 80 °C for 24 h in a vacuum drying oven. The MNb2O6 (M = Co and Ni) materials were prepared by the same method according to the procedure described above. The precursor gel was then introduced into a muffle furnace and sintered at 1000 °C for 2 h to obtain the target products, respectively. Additionally, in order to investigate the effect of sintering temperature on sensing performance, ZnNb2O6 was sintered at 800, 1000 and 1200 °C for 2 h, respectively.
The structural properties of the products were characterized with a Rigaku wide-angle X-ray diffractometer (D/max rA, using Cu-Kα radiation of wavelength 0.1541 nm) in the angular range of 20–80°. Field-emission scanning electron microscopy (FESEM) measurements of the surface morphology of the ZnNb2O6-SE materials were performed using a JEOL JSM-7500F microscope with an accelerating voltage of 15 kV.
2.2 Fabrication and measurement of gas sensor
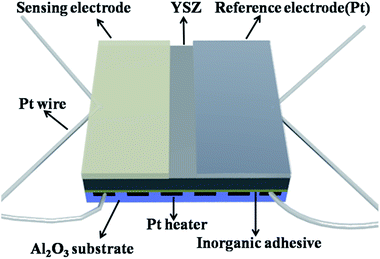
The sensor was fabricated using a YSZ plate (8 mol% Y2O3-doped, 2 mm × 2 mm square, 0.3 mm thickness, provided by Anpeisheng Corp., China). A point-shaped and a narrow stripe-shaped Pt electrode (reference electrode, RE) were formed on the two ends of the YSZ plate using a commercial Pt paste (Sino-platinum Metals Co., Ltd.), and sintered at 1000 °C. The various sensing electrode materials were mixed with a minimum quantity of deionized water to obtain required pastes, respectively. Then, the resultant paste was applied on the point-shaped Pt to form stripe-shaped sensing electrode (SE), and then the device was sintered at 800 °C for 2 h to achieve good contact between the sensing electrode and electrolyte. The Pt heater printed on the Al2O3 substrate was then fixed to the YSZ plate by the inorganic adhesive, which provided the required heating temperature for the sensor. A schematic diagram of the fabricated sensor is shown in Fig. 1.The gas sensing characteristics of the fabricated sensors were measured by a conventional static method.23,24 The electric potential difference (V) between the SE and the RE was measured with a digital electrometer (Rigol Technologies, Inc., DM3054, China) when the sensor was exposed to air or sample gas. The results obtained were recorded with a computer connected to the electrometer. The desired concentration of sample gas was obtained by the static liquid gas distribution method, which was calculated by the following formula:25,26
3. Results and discussion
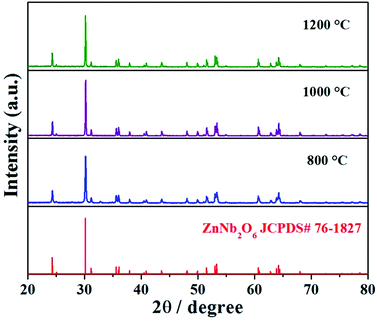
The typical XRD patterns of ZnNb2O6 composite oxide materials annealed at different temperatures are presented in Fig. 2. The sharp diffraction features suggest the good crystalline nature of the prepared ZnNb2O6 composite oxide sensing electrode materials. The diffraction peaks in the pattern of ZnNb2O6 sintered at 800, 1000 and 1200 °C can be readily indexed to columbite type structure of ZnNb2O6 (JCPDS#76-1827).27–29 No characteristic peaks from impurities in ZnNb2O6 samples are detected. The morphologies of ZnNb2O6-SE sintered at different temperatures (800, 1000 and 1200 °C) were studied by FESEM and results are shown in Fig. 3. Obviously, as shown in Fig. 3(a)–(c), porous structures were observed and such structure contributed to diffusion of gas molecules within the material. Additionally, the size of particles increased gradually with the increasing of sintering temperature. Fig. 3(d) displayed EDS mapping images of the surface of ZnNb2O6-SE sintered at 1000 °C. For the single columbite phase ZnNb2O6, the elemental mapping measurement further confirms the coexistence and homogeneous dispersion of Zn, Nb and O elements. | ||
| Fig. 3 SEM images of ZnNb2O6-SEs sintered at different temperatures: (a) 800 °C; (b) 1000 °C; (c) 1200 °C; (d) EDS mapping images for Zn, Nb and O of ZnNb2O6 sintered at 1000 °C. | ||
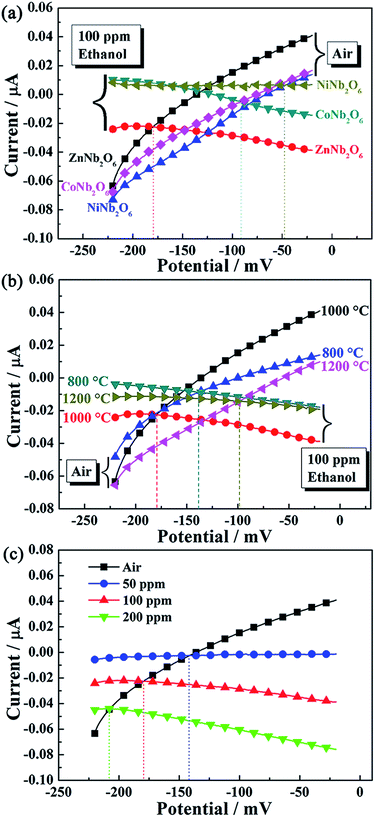
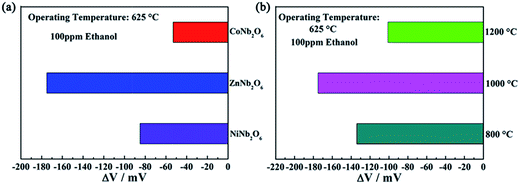
In order to find out the most suitable sensing electrode material, three kinds of columbite type oxides (ZnNb2O6, NiNb2O6 and CoNb2O6) were used to form the sensing electrode layer of YSZ-based sensors, and responses of the fabricated devices to 100 ppm ethanol were measured at 625 °C. It is noteworthy that, as shown in Fig. 4(a), the sensor utilizing ZnNb2O6-SE was found to exhibit the highest response value to ethanol, compared with devices attached with other oxide materials. Thus, the sensing properties of the sensor attached with ZnNb2O6-SE were investigated at 625 °C in more detail. For this kind of gas sensor, the sensing characteristics are affected by the sintering temperature of the sensing electrode material.30–32 Sensors using ZnNb2O6-SE calcined at 800, 1000 and 1200 °C were fabricated and the responses to 100 ppm ethanol for the fabricated devices are displayed in Fig. 4(b). It is apparent that the sensor utilizing ZnNb2O6-SE annealed at 1000 °C exhibited the largest response values to ethanol at the examined concentration when comparing with the devices attached with ZnNb2O6-SE sintered at other temperatures. The sensing characteristics of the present devices related to the mixed potential mechanism, and the sensor can be represented by the following electrochemical cells:
In air: O2, ZnNb2O6/YSZ/Pt, O2
In sample gas: C2H5OH + O2, ZnNb2O6/YSZ/Pt, C2H5OH + O2
 | ||
| Fig. 4 (a) Response to 100 ppm ethanol for sensors using different sensing electrodes; (b) response to 100 ppm of ethanol for sensors attached with ZnNb2O6-SE calcined at different temperatures. | ||
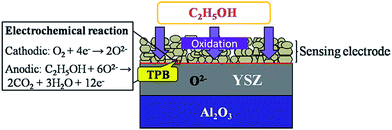
When the ethanol gas arrived at the TPB (triple phase boundary, the interface of ZnNb2O6-SE, ethanol and YSZ), the cathodic reaction of O2 (eqn (1)) and anodic reaction of ethanol (eqn (2)) take place simultaneously at the TPB and form a local cell, as shown in Fig. 5. The rates of two electrochemical reactions are equal to each other, dynamic equilibrium is reached, and the electrode potential is regarded as the mixed potential. The potential difference of the sensing electrode and reference electrode is expressed as the sensing signal.
| Cathodic reaction: O2 + 4e− → 2O2− | (1) |
| Anodic reaction: C2H5OH + 6O2− → 2CO2 + 3H2O + 12e− | (2) |
For the present device, the sensing signal is strongly dependent on the ethanol concentration at the TPB and degree of electrochemical reactions at TPB. Taking these factors into consideration, the porous structure of the SE layer and higher electrochemical reaction to ethanol will achieve higher sensing performance. The porous channels decreased the consumption of ethanol in the process of diffusion in ZnNb2O6 electrode layer and facilitate more ethanol gas to reach the TPB, which participates directly in electrochemical reactions. However, the microstructure of ZnNb2O6 particles should be taken into account, with a further increase of annealing temperature beyond 1000 °C, the particles became too large, so that while the interfacial area of the enlarged particles and YSZ increases, the TPB area is reduced and the number of electrochemically reactive active sites are decreased. In this case, the sensitivity of the sensor to ethanol will be decreased. As stated above, the crystallization of ZnNb2O6 increased with increasing sintering temperature, which affected the electrochemical activity. Therefore, both the degree of crystallization and microstructure of SE affect the response variation of the sensor to ethanol. The best balance of the electrochemical activity and TPB length was obtained when the annealing temperature is 1000 °C and the highest response to ethanol was achieved. Thus, the sensor using ZnNb2O6 sintered at 1000 °C was further investigated in the following sections.
For further clarification of the reasons for the highest response of the sensor utilizing ZnNb2O6-SE annealed at 1000 °C and validating the proposed mixed-potential mechanism, polarization curves of the sensor attached with MNb2O6 (M = Zn, Co, Ni)-SE and ZnNb2O6 sintered at different temperatures in air and 100 ppm ethanol and polarization curves of the sensor utilizing ZnNb2O6-SE sintered at 1000 °C in different concentrations of ethanol at 625 °C were measured and are shown in Fig. 6.33 The cathodic polarization curve was obtained in air, and the anodic polarization curve was obtained by subtracting values in air from values in sample gas (different concentrations of ethanol + air studied). From Fig. 6(a), it can be clearly seen that the polarization curve for the anodic reaction of ethanol for the device using ZnNb2O6-SE shifts downward to higher current values, compared with that of other SEs. This shift of polarization curve is considered as due to the electrochemical reaction of ethanol. It was assumed that the shift of current at an electrode potential was ascribed to the current due to anodic reaction (2) of ethanol. This indicates that sensor attached with ZnNb2O6-SE exhibits the highest electrochemical catalytic activity to anodic reaction (2) of ethanol. In this case, the sensor attached with ZnNb2O6-SE displayed the highest sensitivity to ethanol at 625 °C. Similarly, as shown in Fig. 6(b), the sensor attached with ZnNb2O6-SE sintered at 1000 °C also displays the highest electrochemical catalytic activity to anodic reaction of ethanol, by comparing the anodic polarization curves of the sensors using ZnNb2O6-SE sintered at different temperatures. Additionally, the mixed potential can be estimated from the intersection of the cathodic and anodic polarization curves.34,35 Based on the comparison of the mixed potential estimated values and the potential difference values experimentally observed for the three fabricated sensors and the device utilizing ZnNb2O6-SE annealed at 1000 °C to different concentrations of ethanol at 625 °C, in Table 1, the estimated values are in close proximity to those observed values. These results supported the sensing mechanism involving mixed potential.36–39
| Sensors | Ethanol conc./ppm | Mixed potential (estimated)/mV | Observed potential difference/mV |
|---|---|---|---|
| ZnNb2O6 (800 °C)-SE | 100 | −138.5 | −134 |
| ZnNb2O6 (1200 °C)-SE | 100 | −98 | −101 |
| ZnNb2O6 (1000 °C)-SE | 100 | −179 | −175 |
| ZnNb2O6 (1000 °C)-SE | 50 | −142 | −141 |
| ZnNb2O6 (1000 °C)-SE | 200 | −208 | −205 |
It is well known that the response of the sensor is strongly influenced by the operating temperature. In order to determine the optimal operating temperature to ethanol of the sensor attached with ZnNb2O6-SE annealed at 1000 °C, the responses of device fabricated to 50 ppm ethanol at different operating temperatures were tested and the results are presented in Fig. 7. It can be observed that the response to 50 ppm ethanol for the fabricated sensor tended to increase and reached the highest response value at an operating temperature of 625 °C, and then decreased with a further rise of operating temperature. The occurrence of the electrochemical reaction for the present device at TPB required a definite activation energy. The electrochemical reaction did not gain enough activation energy below 625 °C, thus, the sensitivity of the sensor to ethanol increased with increasing of temperature. However, the desorption process of ethanol became dominant above 625 °C, and the amount of ethanol reaching the TPB became less and less with further increase of operating temperature, and thus the response of the sensor to ethanol was reduced. Consequently, the optimal operating temperature for the present sensor was considered to be 625 °C.
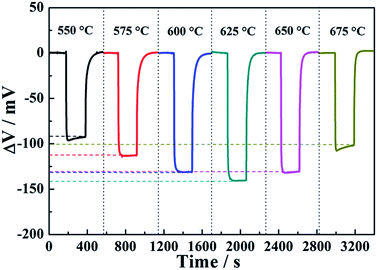 | ||
| Fig. 7 Response and recovery transients for the sensor using ZnNb2O6-SE sintered at 1000 °C to 50 ppm ethanol at different operating temperatures. | ||
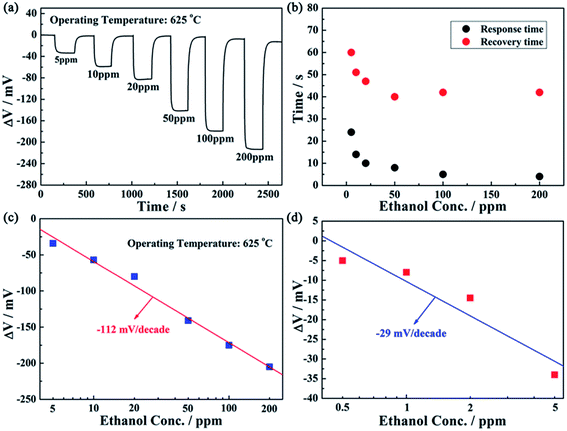
The response transients of the fabricated sensor attached with ZnNb2O6-SE annealed at 1000 °C toward different concentrations of ethanol in the range of 5–200 ppm was examined at 625 °C and the results obtained are shown in Fig. 8(a). It is obvious that the sensor quickly reached a steady state value when exposed to ethanol atmosphere, and the response increased with the increase of ethanol concentration. The response for the sensor attached with ZnNb2O6-SE annealed at 1000 °C to 5 and 200 ppm ethanol at 625 °C was −34 and −205 mV, respectively. Besides, the response and recovery times of the present sensor were also further evaluated according to Fig. 8(a). As shown in Fig. 8(b), the response and recovery process become shortened with increasing gas concentration, which may be attributed to a lower gas concentration requiring more time to reach equilibrium. The typical 90% response and recovery times of the present device toward 50 ppm of ethanol are 8 and 40 s, which shows fast response and recovery rates. Furthermore, the dependence of ΔV for the sensor attached with ZnNb2O6-SE sintered at 1000 °C on the ethanol concentration in the examined range at 625 °C is shown in Fig. 8(c). In this case, almost linear relationships between the ΔV and the logarithm of ethanol concentration in the range of 5–200 ppm at 625 °C were observed, which conforms to the mixed potential type model. The sensitivity (slope) of the present sensor is −112 mV decade−1. Moreover, the present sensor can detect 500 ppb ethanol, for which the response value is −5 mV. The sensitivity to ethanol in the range of 0.5–5 ppm for the fabricated sensor is reduced to −29 mV decade−1 (Fig. 8(d)). The occurrence of such a linear dependence of ethanol sensitivity can be rationalized. As is well known, the electrochemical reaction for the present mixed potential type ethanol sensor occurred at the TPB and the sensing signal of the sensor depended on the concentration of ethanol at the TPB of ZnNb2O6-SE and the amount of active sites of TPB. For the low concentration range of ethanol (0.5–5 ppm), the amount of TPB active sites for the present sensor are enough to provide full electrochemical reaction, therefore, the sensitivity of the sensor is mainly related to the amount of ethanol reaching the TPB of ZnNb2O6-SE. Owing to porous channels in the sensing electrode layer, a certain amount of gas is consumed in the process of diffusion, so that when the concentration of ethanol is at a lower level, such ethanol consumption is relatively larger than that at higher ethanol concentration (5–500 ppm). Therefore, a relatively low sensitivity to ethanol concentration in the range of 0.5–5 ppm was observed.
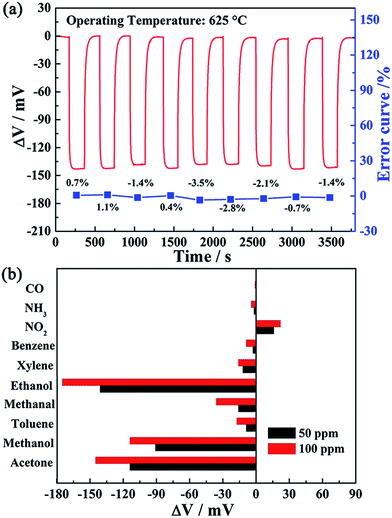
Moreover, the continuous response–recovery and selectivity for a gas sensor are important sensing performance parameters. The continuous response and recovery transients of the present sensor to 50 ppm ethanol at 625 °C is illustrated in Fig. 9(a). It is clearly seen that the responses of the present device to 50 ppm ethanol showed little fluctuation and the highest change error was −3.5% in the examined nine cycles, which indicated that the sensor displayed good repeatability. Fig. 9(b) shows the cross-sensitivities for the sensor attached with ZnNb2O6-SE annealed at 1000 °C to various gases at 625 °C, such as toluene, benzene, acetone, methanol, NO2 and NH3, etc. It is obvious that the sensor exhibited relatively high responses to 50 and 100 ppm of ethanol compared with those of other interfering gases. However, the effect of the acetone and methanol as interfering gases on the ethanol response for the fabricated sensor was substantial. As has been reported previously,40–42 these species are difficult to be differentiated using fabricated sensors. The reasons may be as follows: the sensing electrode material shows similar reactivity to similar kinds of chemical species (ethanol, acetone and methanol), i.e. the ZnNb2O6 sensing electrode material displayed similar electrochemical catalytic reaction activity towards ethanol, acetone and methanol, so leading to little difference in sensitivity. Thus, the present device is insufficient in selectivity of certain gases. Development of new sensing electrode materials with high electrochemical catalytic activity to certain signal gases, fabrication of an array of sensors or change of device structure may be a good future strategy to overcome this problem. The improvement of selectivity of ethanol relative to acetone and methanol remains to be further investigated in future work. On the whole, however, the present sensor still displayed acceptable selectivity to most of various interfering gases.
For a point of view of actual application of the sensor, the sensitivity to a target gas should not be affected under long-term working conditions. The stability of the present sensor was investigated by continuous working at high temperature of 625 °C during 30 days. The responses of the sensor to 50 ppm ethanol were measured every other day and the obtained results are depicted in Fig. 10. It can be seen that the change amplitude of ΔV for the sensor attached with ZnNb2O6-SE sintered at 1000 °C varied slightly towards 50 ppm ethanol during the 30 day measurement period. In order to further illustrate the change amplitude of ΔV with time, the change of ΔV (ΔVs) for the sensor is given by ΔVs = [(ΔVn − ΔV0)/ΔV0 × 100%], where ΔVn and ΔV0 denote ΔV of the sensor on the n and initial day, respectively. The quantitative result showed that ΔVs for the sensor to 50 ppm ethanol on the 30th day was less than 4%, which shows that the sensor has good stability. Based on the above results, the comparison of the ethanol sensing property for the fabricated sensor and those reported previously in the literature is presented in Table 2. Obviously, the present device exhibited better sensing performance in terms of response, sensitivity and low detection limit to ethanol than previously reported devices.
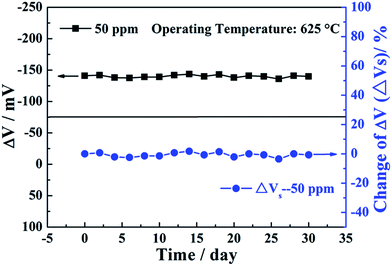 | ||
| Fig. 10 Long-term stability to 50 ppm ethanol at 625 °C for the sensor attached with ZnNb2O6-SE sintered at 1000 °C. | ||
| Material | Ethanol conc./ppm | Response/mV | Sensitivity/mV decade−1 | Low detection limit/ppm | Ref. |
|---|---|---|---|---|---|
| ZnNb2O6 | 50 | −141 | −112 | 0.5 (5 mV) | Present work |
| Pt/MoO3/SnO2 | 100 | 13 | 41 | — | 43 |
| Pt/CeO2/SnO2 | 100 | 28 | 49 | — | 43 |
| In2O3/SnO2 | 100 | 53.2 (R = Ra/Rg) | — | — | 2 |
| CdO/ZnO | 100 | 76 (R = Ra/Rg) | — | 0.5 (1.3) | 44 |
| Cr2O3/ZnO | 100 | 25 (R = Ra/Rg) | — | 1 (3.6) | 45 |
| In2−xNixO3 | 100 | 80 (R = Ra/Rg) | — | 1 (5) | 46 |
| SnO2/α-Fe2O3 | 100 | 16 (R = Ra/Rg) | — | 10 (3) | 47 |
4. Conclusions
In this paper, a compact solid-state electrochemical ethanol sensor based on YSZ and columbite type complex oxide sensing electrode synthesized via a facile sol–gel method was developed. Among the oxide SEs examined, ZnNb2O6 was found to be best suited as a sensing electrode of a YSZ-based ethanol device. The sensor attached with ZnNb2O6-SE sintered at 1000 °C displayed the highest response to ethanol at 625 °C. The present sensor showed a low detection limit of 500 ppb of ethanol, and ΔV of the device exhibited a segmentally linear relationship to the logarithm of ethanol concentration in the ranges of 0.5–5 ppm and 5–200 ppm, with slopes of −29 and −112 mV decade−1, respectively. Furthermore, the present device also exhibited good repeatability, acceptable selectivity and excellent stability during a measurement period of 30 days at high-temperature-aging of 625 °C. Given the above-mentioned sensing performance of the fabricated mixed potential type sensor, the present sensor can be considered as a potential candidate in detecting ethanol.Acknowledgements
This work is supported by the National Nature Science Foundation of China (No. 61134010, 61327804, 61374218, 61473132, 61533021, and 61520106003), Program for Chang Jiang Scholars and Innovative Research Team in University (No. IRT13018) and National High-Tech Research and Development Program of China (863 Program No. 2014AA06A505), Application and Basic Research of Jilin Province (2013010 2010JC).Notes and references
- A. Yazıcı, N. Dalbul, A. Altındal, B. Salih and Ö. Bekaroğlu, Sens. Actuators, B, 2014, 202, 14–22 CrossRef.
- Y. Liu, S. Yao, Q. Yang, P. Sun, Y. Gao, X. Liang, F. Liu and G. Lu, RSC Adv., 2015, 5, 52252–52258 RSC.
- M. Shnayderman, B. Mansfield, P. Yip, H. Clark, M. Krebs, S. Cohen, J. Zeskind, E. Ryan, H. Dorkin, M. Callahan, T. Stair, J. Gelfand, C. Gill, B. Hitt and C. Davis, Anal. Chem., 2005, 77, 5930–5937 CrossRef CAS PubMed.
- M. Franke, T. Koplin and U. Simon, Small, 2006, 2, 36–50 CrossRef CAS PubMed.
- N. Miura, H. Kurosawa, M. Hasei, G. Lu and N. Yamazoe, Solid State Ionics, 1996, 86–88, 1069–1073 CrossRef CAS.
- G. Lu, N. Miura and N. Yamazoe, J. Appl. Electrochem., 1998, 28, 1009–1011 CrossRef CAS.
- G. Lu, N. Miura and N. Yamazoe, Sens. Actuators, B, 2000, 65, 125–127 CrossRef CAS.
- Q. Diao, C. Yin, Y. Liu, J. Li, X. Gong, X. Liang, S. Yang, H. Chen and G. Lu, Sens. Actuators, B, 2013, 180, 90–95 CrossRef CAS.
- F. Liu, Y. Guan, M. Dai, H. Zhang, Y. Guan, R. Sun, X. Liang, P. Sun, F. Liu and G. Lu, Sens. Actuators, B, 2015, 216, 121–127 CrossRef CAS.
- S. Anggraini, M. Breedon and N. Miura, Sens. Actuators, B, 2013, 187, 58–64 CrossRef CAS.
- Y. Li, X. Li, Z. Tang, Z. Tang, J. Yu and J. Wang, Sens. Actuators, B, 2015, 206, 176–180 CrossRef CAS.
- T. Hübert, L. Boon-Brett, G. Black and U. Banach, Sens. Actuators, B, 2011, 157, 329–352 CrossRef.
- N. Miura, T. Raisen, G. Lu and N. Yamazoe, Sens. Actuators, B, 1998, 47, 84–91 CrossRef CAS.
- Y. Fujio, V. Plashnitsa, M. Breedon and N. Miura, Langmuir, 2012, 28, 1638–1645 CrossRef CAS PubMed.
- F. Liu, R. Sun, Y. Guan, X. Cheng, H. Zhang, Y. Guan, X. Liang, P. Sun and G. Lu, Sens. Actuators, B, 2015, 210, 795–802 CrossRef CAS.
- Y. Fujio, T. Sato and N. Miura, Solid State Ionics, 2014, 262, 266–269 CrossRef CAS.
- Y. Suetsugu, T. Sato, M. Breedon and N. Miura, Electrochim. Acta, 2012, 73, 118–122 CrossRef CAS.
- T. Sato, V. Plashnitsa, M. Utiyama and N. Miura, Electrochem. Commun., 2010, 12, 524–526 CrossRef CAS.
- F. Liu, Y. Guan, R. Sun, X. Liang, P. Sun, F. Liu and G. Lu, Sens. Actuators, B, 2015, 221, 673–680 CrossRef CAS.
- N. Miura, T. Sato, S. Anggraini, H. Ikeda and S. Zhuiykov, Ionics, 2014, 20, 901–925 CrossRef CAS.
- G. Lu, Q. Diao, C. Yin, S. Yang, Y. Guan, X. Cheng and X. Liang, Solid State Ionics, 2014, 262, 292–297 CrossRef CAS.
- R. Moos, K. Sahner, M. Fleischer, U. Guth, N. Barsan and U. Weimar, Sensors, 2009, 9, 4323–4365 CrossRef CAS PubMed.
- Q. Diao, C. Yin, Y. Guan, X. Liang, S. Wang, Y. Liu, Y. Hu, H. Chen and G. Lu, Sens. Actuators, B, 2013, 177, 397–403 CrossRef CAS.
- C. Wang, X. Cheng, X. Zhou, P. Sun, X. Hu, K. Shimanoe, G. Lu and N. Yamazoe, ACS Appl. Mater. Interfaces, 2014, 6, 12031–12037 CAS.
- H. Fan, Y. Zeng, H. Yang, X. Zheng, L. Liu and T. Zhang, Acta Phys.–Chim. Sin., 2008, 24, 1292–1296 CAS.
- P. Sun, X. Zhou, C. Wang, B. Wang, X. Xu and G. Lu, Sens. Actuators, B, 2014, 190, 32–39 CrossRef CAS.
- X. Huang, Y. Jing, J. Yang, J. Ju, R. Cong, W. Gao and T. Yang, Mater. Res. Bull., 2014, 51, 271–276 CrossRef CAS.
- H. Bafrooei, E. Nassaj, T. Ebadzadeh and C. Hu, Ceram. Int., 2014, 40, 14463–14470 CrossRef CAS.
- D. Xu, H. Yang, L. Li, Q. Zhou, H. Yuan and T. Cui, Cryst. Res. Technol., 2014, 49, 502–506 CrossRef CAS.
- H. Zhang, T. Zhong, R. Sun, X. Liang and G. Lu, RSC Adv., 2014, 4, 55334–55340 RSC.
- S. Anggraini, M. Breedon and N. Miura, J. Electrochem. Soc., 2013, 160, B164–B169 CrossRef CAS.
- L. Zhou, Q. Yuan, X. Li, J. Xu, F. Xia and J. Xiao, Sens. Actuators, B, 2015, 206, 311–318 CrossRef CAS.
- G. Lu, N. Miura and N. Yamazoe, Sens. Actuators, B, 1996, 35, 130–135 CrossRef CAS.
- N. Miura, H. Kurosawa, M. Hasei, G. Lu and N. Yamazoea, Solid State Ionics, 1996, 86–88, 1069–1073 CrossRef CAS.
- N. Miura, G. Lu and N. Yamazoe, Solid State Ionics, 2000, 136–137, 533–542 CrossRef CAS.
- J. Wang, P. Elumalai, D. Terada, M. Hasei and N. Miura, Solid State Ionics, 2006, 177, 2305–2311 CrossRef CAS.
- P. Elumalai, J. Wang, S. Zhuiykov, D. Terada, M. Hasei and N. Miura, J. Electrochem. Soc., 2005, 152, H95–H101 CrossRef CAS.
- G. Lu, N. Miura and N. Yamazoe, J. Mater. Chem., 1997, 7, 1445–1449 RSC.
- X. Liang, S. Yang, J. Li, H. Zhang, Q. Diao, W. Zhao and G. Lu, Sens. Actuators, B, 2011, 158, 1–8 CrossRef CAS.
- P. Sun, X. Zhou, C. Wang, K. Shimanoe, G. Lu and N. Yamazoe, J. Mater. Chem. A, 2014, 2, 1302–1308 CAS.
- F. Liu, Y. Guan, R. Sun, X. Liang, P. Sun, F. Liu and G. Lu, Sens. Actuators, B, 2015, 221, 673–680 CrossRef CAS.
- F. Qu, J. Liu, Y. Wang, S. Wen, Y. Chen, X. Li and S. Ruan, Sens. Actuators, B, 2014, 199, 346–353 CrossRef CAS.
- M. Kasalizadeh, A. Khodadadi and Y. Mortazavi, J. Electrochem. Soc., 2013, 160, B218–B224 CrossRef CAS.
- L. Zhou, C. Li, X. Zou, J. Zhao, P. Jin, L. Feng, M. Fan and G. Li, Sens. Actuators, B, 2014, 197, 370–375 CrossRef CAS.
- W. Wang, Z. Li, W. Zheng, H. Huang, C. Wang and J. Sun, Sens. Actuators, B, 2010, 143, 754–758 CrossRef CAS.
- C. Feng, W. Li, C. Li, L. Zhu, H. Zhang, Y. Zhang, S. Ruan, W. Chen and L. Yu, Sens. Actuators, B, 2012, 166–167, 83–88 CrossRef CAS.
- P. Sun, X. Zhou, C. Wang, K. Shimanoe, G. Lu and N. Yamazoe, J. Mater. Chem. A, 2014, 2, 1302–1308 CAS.
| This journal is © The Royal Society of Chemistry 2016 |