Polymer-templated mesoporous hybrid oxides of Al and Cu: highly porous sorbents for ammonia†
Hairong Tang‡
ab,
Chamila Gunathilake‡b,
Xuezhi Zhoua and
Mietek Jaroniec‡*b
aThe Institute of Chemical Defense, P.O. Box 1048, Beijing 102205, China
bDepartment of Chemistry and Biochemistry, Kent State University, Kent, Ohio 44242, USA. E-mail: jaroniec@kent.edu; Fax: +1-330-672-3816; Tel: +1-330-672-3790
First published on 13th April 2016
Abstract
Hybrid mesostructures consisting of copper and aluminum oxides were synthesized via co-condensation of suitable precursors in the presence of a triblock copolymer, Pluronic F127. These mesostructures were prepared using aluminum isopropoxide and copper nitrate as precursors. The aforementioned polymer-templated synthesis afforded copper oxide–alumina hybrid materials with well-developed mesoporosity and a high specific surface area (130–460 m2 g−1). Controlled heating of these materials in nitrogen resulted in template free copper oxide–alumina mesostructures showing high affinity toward NH3 and moderate affinity toward CO2 at elevated temperatures (∼50 °C). The temperature programmed desorption studies revealed a superior NH3 sorption capacity of the samples with a high Cu content at 50 °C (over 16 mmol g−1). The high affinity of mesoporous copper oxide–alumina materials toward NH3, their good thermal stability together with their high surface area make these materials attractive sorbents for various applications, where large and accessible pores with high loading of catalytically active metal oxides are needed.
Introduction
In recent years, mesoporous alumina (MA) or ordered mesoporous alumina (OMA) have been extensively studied because of their wide range of applications such as optical,1 electronic,2 catalytic,3–5 and biomedical fields.6,7 MA and OMA possess a large surface area, tunable porosity, high thermal stability, and high amount of surface Lewis acidic sites.8 On the other hand, cuprous oxide (Cu2O) and cupric oxide (CuO) are of great interest among transition metal oxides because of their catalytic and magnetic properties.9 Both CuO and Cu2O are extensively used in various areas such as removal of inorganic pollutants,10,11 nanofluids,12 photocatalysis, gas sensors,13 biosensors,14 photodetectors,15 field emission displays,16 supercapacitors,17 energetic materials,18 and magnetic storage media.19 Furthermore, nanosized CuO can be used to prepare various organic–inorganic nanocomposites.20,21 Moreover, Cu2O and CuO have different colors and physical properties.22 For instance, Cu2O is reddish p-type semiconductor. In contrast, CuO has an iron-dark color and exhibits promising antiferromagnetic properties. CuO is more stable than Cu2O because Cu(II) ions are much more stable under ambient conditions, which makes it more important in potential catalytic applications.Alumina can also act as an important support for catalysts. For instance, alumina-supported copper oxide material has been widely investigated during the past decades due to its enhanced catalytic activity toward many reactions. CuO species supported on γ-Al2O3 were classified by Tikhov et al. as isolated ionic species, weakly magnetic, two- and three-dimensional clusters, and CuO domains.23 This hybrid material is also an effective catalyst for oxidation of CO, NO, and volatile organic chemicals such as methanol and ethene.24–27 Zhu et al. reported that copper catalysts supported on mesoporous Al2O3 nanofibers are excellent catalysts for the conversion of N2O to N2. It was shown that the activity of these catalysts depends on the type of structure and dispersion of copper species on the alumina support. Several copper species such as surface spinel, bulk CuAl2O4, and CuO present in the alumina-supported catalysts have been widely investigated by Zhu and co-workers.28 Some reports showed that CuO is the most promising catalyst for simultaneous removal of SO2 and NOx.29 In addition, the alumina-supported copper species can be used for catalytic dehydrogenation of cyclohexanol,30 microwave-enhanced degradation of phenol,31 defluoridation of drinking water,32 and removal of As(III) and As(V) from contaminated water.33
Generally, there are two major ways of synthesizing MA: nanocasting and soft-templating. In the first case, ordered silica or carbon materials are used as hard templates.34,35 For instance, Zhao et al. utilized bimodal mesoporous carbon as a hard template to prepare MA with variable architectures.35 The other frequently used method is the sol–gel process employing triblock copolymers as soft templates.8,36–39 For instance, Niesz and co-workers prepared ordered mesoporous alumina in the presence of triblock copolymers under strictly controlled conditions.37 Yuan et al. reported evaporation-induced self-assembly (EISA) method for the synthesis of highly ordered and thermally stable mesoporous alumina,8 which seems to be one of the simplest and effective ways of preparing MA. Furthermore, Wang et al. obtained highly ordered mesoporous zirconia–alumina nanomaterials by improved EISA method.40
Extension of the self-assembly synthesis from mesoporous silica to alumina materials is more challenging. This is due to the differences in the hydrolysis and condensation rates of alumina precursors (especially alkoxides), and in the interactions between alumina precursors, structure directing agents and other synthesis components as compared to tetraethyl orthosilicate (silica precursor).41 Also, a strict control of the synthesis conditions (pH, solvents, additives to control hydrolysis rates, etc.) is often required in the synthesis of mesoporous materials. Moreover, many strategies used for the incorporation of metal species into silica and alumina mesostructures afford composites/hybrids with reduced pore widths and surface area41 due to the partial blockage or constriction of some pores, which can limit the utility of such materials as sorbents and catalysts. Among various strategies the one-pot synthesis involving co-assembly of metal species and silica and/or alumina precursors is promising because it is much less prone to the formation of constrictions and/or pore blocking. However, the structural properties of hybrid aluminum–metal oxides can differ substantially from those of pure alumina. Thus, the adjustment of chemical composition of these hybrid oxides can be successfully used to tune their surface and structural characteristics, and consequently their sorption and catalytic properties.41
Among many metal oxides, alumina and copper oxides-containing sorbents can be prepared with large pore volumes, highly interconnected mesoporosity, high BET surface area, and high mechanical stability. Note that aluminum oxide alone shows relatively good catalytic activity. Thus, it would be worthy to synthesize composite sorbents based on aluminum and copper oxides to enhance further their properties and applications. Additional benefits of these materials are good thermal stability and high resistance toward corrosion and degradation.
Here we report the synthesis of porous composite sorbents consisting of aluminum and copper oxides. A simple and feasible sol–gel synthesis combined with EISA was carried out under acidic conditions in the presence of Pluronic F127 triblock copolymer (soft template),40 which is inexpensive and biodegradable. This study shows the effect of copper content and calcination temperature on the structure, porosity, surface area, thermal stability, and acidic and basic properties of the resulting hybrid materials. Interestingly, the mesostructural ordering of the composite Al–Cu oxides was not considerably affected by increasing Cu content. To the best of our knowledge, this is the first effort to synthesize series of mesoporous alumina–copper oxide composites with interesting basic and acidic properties as evidenced by CO2 and NH3 sorption at elevated temperature (50 °C).
Experimental
Materials, characterization, and calculations
A detailed information including chemicals used in the synthesis, techniques employed for characterization of the hybrid Al and Cu oxides such as nitrogen adsorption, thermogravimetry (TG), X-ray diffraction (XRD), and XPS (X-ray photoelectron spectroscopy) are provided in the ESI.† Also, the relevant information related to the determination of the specific surface area, pore diameter, pore volume, and pore size distribution (PSD)42 and to the analysis of the CO2 and NH3 temperature programmed desorption (TPD) at 50 °C for the samples studied is provided in the ESI.†Preparation of porous composites
Alumina-supported copper oxide samples were obtained by adapting the existing procedures for the synthesis of alumina,8,36,38,40 as shown in Scheme 1. Specifically, Pluronic F127 (EO106PO70EO106; 3.2 g) was dissolved in 99.5% anhydrous ethanol (20 mL) containing citric acid (0.8 g) and 37 wt% HCl (1.6 g). The resultant mixture was further stirred for 4 h at room temperature. Next, 98% aluminum isopropoxide (3.26 g = 16 mmol) and the specified amount of Cu(NO3)2 (0.16, 0.32, 0.80, 1.60, 2.40, 3.20 mmol) were simultaneously and slowly added to the above solution. The resultant mixture was stirred further at room temperature for 24 h. The final solution mixture was transferred to a Petri-dish to evaporate solvent at 60 °C for 48 h followed by additional thermal treatment at 100 °C for 24 h. The polymeric template was removed by heating in a horizontal quartz tube furnace at 400, 550, and 700 °C for 5 h in flowing air with a heating rate of 1 °C min−1. The samples were labelled as Al-XCu-T, where X denotes the molar ratio of Cu to Al, T refers to the temperature of thermal treatment, and Al and Cu refer to alumina and copper oxide originating from aluminum isopropoxide and copper nitrate precursors, respectively. For instance, Al-0.15Cu-550 refers to the sample containing one mmol of alumina from aluminum isopropoxide precursor and 0.15 mmol of copper (X = 0.15) from copper nitrate precursor, which was thermally treated at 550 °C for 5 h in flowing air. The as-synthesized samples of alumina and alumina with X moles of copper species are denoted by Al and Al-XCu, respectively.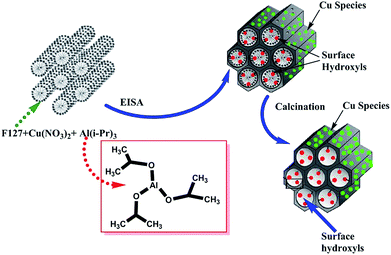 | ||
| Scheme 1 Illustration of the formation of mesoporous alumina with incorporated Cu species via EISA in the presence of Pluronic F127 block copolymer. | ||
Results and discussion
Properties of ordered mesoporous aluminum–copper oxides
 | ||
| Fig. 1 (a) TG and (b) DTG profiles recorded for the as-synthesized Al and Al-XCu (X = 0.01 to 0.20) samples. | ||
Note that the DTG profiles recorded for all as-synthesized samples exhibit peak in the temperature range of 40–130 °C related to the removal of adsorbed water. Two additional peaks observed on these profiles in the temperature ranges of 230–340 °C and 350–450 °C, respectively, correspond to the decomposition of Pluronic F127 triblock copolymer (used as a soft template) and to the dehydration of hydrated alumina–copper oxide hybrid samples. The remaining portion of the template after extraction was removed by heating (calcination) at a temperature of 400 °C or higher (550, 700 °C). The disappearance of the template decomposition peak in the temperature range of 230–340 °C (not shown here) was monitored to confirm the completeness of the removal of block copolymer from as-synthesized samples. The DTG profiles obtained for Al-550, Al-0.01Cu-550, Al-0.02Cu-550, Al-0.05Cu-550, Al-0.10Cu-550, Al-0.15Cu-550, and Al-0.20Cu-550 samples display two main peaks at the temperature ranges of 140–230 °C and 400–500 °C (not shown here). The first one reflects desorption of adsorbed water, while the second one at 450 °C refers to the dehydration of hydrated alumina–copper oxide samples. As can be seen from Fig. 1b, the decomposition temperature of Pluronic F127 in the as-synthesized Cu–Al mesostructures is shifted to lower temperatures with increasing Cu loading; Pluronic F127 [H(C2H4O)70(C3H6O)106(C2H4O)70OH] is a triblock copolymer possessing hydrophilic–hydrophobic–hydrophilic segments. During the self-assembly process these segments interact with metal oxide precursors and play a crucial role in forming micelles that assemble into ordered mesostructures. It is possible that Cu species facilitate thermal degradation of F127, which results in the observed shift of the decomposition temperature.
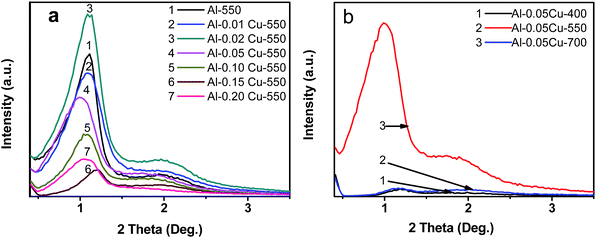 | ||
| Fig. 2 Small angle XRD patterns of (a) Al-XCu-550 (X = 0.01 to 0.20) and (b) Al-0.05Cu-T (T = 400, 550, 700) samples. | ||
However, as can be seen from Fig. 2b and S1 (top and bottom panels) and S2 in ESI,† calcination of the as-synthesized hybrid samples at different temperatures (400, 550, 700 °C) altered significantly the XRD spectra, indicating that the structural ordering depends strongly on the calcination temperature. For instance, the XRD patterns obtained for Al-0.05Cu-550 and Al-0.05Cu-700 samples exhibit one broad less intensive peak in comparison to that recorded for Al-0.05Cu-400 in the range of 2θ from 0.85 to 1.20°, indicating a noticeable deterioration of the ordered mesostructure with increasing calcination temperature (Fig. 2b). Regardless of the calcination temperature, higher Cu loading tends to shift the d100 peak toward larger values of 2θ angle and causes a broadening of the XRD spectrum. This might be due to the contraction of micelles with increasing Cu loading.
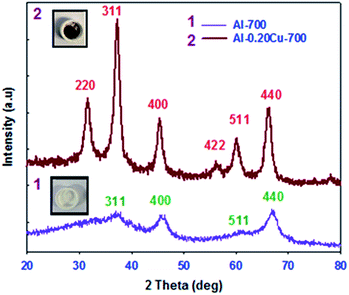
Wide angle powder X-ray diffraction patterns were also recorded for the samples studied to determine their crystalline properties. Fig. 3 shows the XRD patterns for the selected Al-700 and Al-0.02Cu-700 samples. Four XRD peaks were recorded for Al-700 sample at 2θ equal to 37.6, 45.79, 60.89, and 66.76° with the corresponding d-spacing values of 2.39, 1.98, 1.52, and 1.40 Å, respectively. Peaks observed at 2θ equal to 37.6, 45.79, 60.89, and 66.76° can be identified as 311, 400, 511, and 440, respectively.44 In addition, Al-0.2Cu-700 sample shows 6 peaks at 2θ equal to 31.52, 37.39, 45.30, 56.02, 60.04, and 66.50° with the corresponding d-spacing values of 2.85, 2.43, 2.01, 1.64, 1.55, and 1.42 Å, respectively. Six peaks of the Al-0.2Cu-700 sample at 2θ equal to 31.52, 37.39, 45.30, 56.02, 60.04, and 66.50° can be identified as 220, 311, 400, 422, 511, and 440, respectively.44 The wide angle XRD peaks of Al-700 and Al-0.2Cu-700 refer to copper aluminate spinel (CuAl2O4) and γ-alumina (Al2O3) phases, respectively. Note that CuAl2O4 and γ-alumina (Al2O3) are, respectively, brown and white in color. However all samples calcined at 400 and 550 °C do not show any visible peaks in the wide angle range, which indicates their amorphous nature. These findings confirm that the crystalline phases can be observed for the samples calcined at higher temperatures (600 °C or higher).44 The mean size of crystalline domains in the Al-0.2Cu-700 sample estimated on the basis of Scherer equation was in the range of 9–12 nm. The XPS (X-ray Photoelectron Spectroscopy) pattern was also recorded for the Al-0.01Cu-550 sample to determine the valence state of copper and to check the information about elemental composition of the samples studied. About 10% of carbon was found in Al-0.01Cu-550, which can be attributed to the incompletely removed triblock copolymer (Fig. 4).
 | ||
| Fig. 5 N2 adsorption isotherms (a) and the corresponding PSD curves (b) for the Al-550 and Al-XCu-550 (X = 0.01 to 0.20) samples. | ||
| Sample | SBET [m2 g−1] | Pore width Wmax [nm] | Pore volume [cm3 g−1] |
|---|---|---|---|
| a SBET – specific surface area calculated from adsorption data in relative pressure range 0.05–0.20; Wmax – pore width calculated at the maximum of PSD obtained by the improved KJS method. Pore volume – total volume of pores. | |||
| Al-400 | 339 | 4.9 | 0.56 |
| Al-0.01Cu-400 | 457 | 5.5 | 0.69 |
| Al-0.02Cu-400 | 408 | 5.5 | 0.67 |
| Al-0.05Cu-400 | 338 | 5.7 | 0.56 |
| Al-0.10Cu-400 | 283 | 6.9 | 0.66 |
| Al-0.15Cu-400 | 410 | 4.8 | 0.57 |
| Al-0.20Cu-400 | 333 | 5.1 | 0.53 |
| Al-550 | 232 | 5.4 | 0.39 |
| Al-0.01Cu-550 | 373 | 5.2 | 0.58 |
| Al-0.02Cu-550 | 334 | 5.1 | 0.53 |
| Al-0.05Cu-550 | 363 | 6.3 | 0.67 |
| Al-0.10Cu-550 | 328 | 5.8 | 0.60 |
| Al-0.15Cu-550 | 283 | 5.7 | 0.56 |
| Al-0.20Cu-550 | 249 | 5.9 | 0.52 |
| Al-700 | 266 | 4.8 | 0.39 |
| Al-0.01Cu-700 | 388 | 4.7 | 0.60 |
| Al-0.02Cu-700 | 328 | 5.2 | 0.54 |
| Al-0.05Cu-700 | 304 | 4.9 | 0.62 |
| Al-0.10Cu-700 | 276 | 5.7 | 0.52 |
| Al-0.15Cu-700 | 129 | 8.5 | 0.37 |
| Al-0.20Cu-700 | 134 | 6.1 | 0.39 |
Among all the samples calcined at 550 °C, the capillary condensation step observed for Al-550 shows the smallest steepness, whereas the sharpest step is visible for Al-0.01Cu-550. The samples with the Cu loadings higher than 0.05 feature a gradual shift of the condensation step toward higher relative pressures, associated with gradual increase of the mesopore diameter. Another exciting feature of the samples studied is the absence of any significant alternation of the isotherm shape due to the incorporation of higher amount of copper species (X ≥ 0.01).
Adsorption and desorption branches of the hysteresis loops for Al-XCu-550 (X = 0.01 to 0.20) are steep and parallel to each other, which is observed for uniform mesopores including ordered channel-like mesopores. This observation is confirmed by the powder XRD pattern having three peaks indexed as 100, 110, and 200 characteristic for hexagonally ordered mesopores. Thus, a change in the XRD pattern (Fig. 2 a) affects the position of the capillary condensation step of the corresponding nitrogen adsorption isotherm shown in Fig. 5. Namely, a shift of 100 peak in direction of small angles reflects an increase in the pore width, manifested by a shift of the capillary condensation step toward higher relative pressures.
The specific surface area and single-point pore volume obtained for Al-0.01Cu-550 (sample with smallest content of Cu species) are higher than the corresponding values Al-550 (sample without Cu species). Namely, the specific surface area changes from 232 m2 g−1 for Al-550 to 373 m2 g−1 for Al-0.01Cu-550, and the pore volume changes from 0.39 cm3 g−1 for Al-550 to 0.58 cm3 g−1 for Al-0.01Cu-550. However, note that the pore width decreases from 5.4 for Al-550 to 5.2 nm for Al-0.01Cu-550 (see Table 1). A small contraction of the micelles size after adding a small amount of Cu species might be an explanation for the observed change in the pore size. However, the relation between the surface area and pore volume seems to be more complex. An initial increase in the Cu content seems to significantly improve the structural properties of the hybrid mesostructures as evidenced by higher values of the surface area and pore volume, however further increase in the Cu content reduces the values of these parameters (see Table 1). Thus, while a small content of Cu promotes the formation of hybrid mesostructures, its further increase causes a gradual deterioration of their structural properties. This deterioration can be caused by partial blocking of pores and/or formation of some constrictions inside pores, which is reflected by gradual reduction in the specific surface area and pore volume. Overall, the BET surface area for all the samples studied is in the range of 230–460 m2 g−1 except for the Al-0.15Cu-700 and Al-0.20Cu-700 samples, and the pore volume is in the range of 0.39–0.69 cm3 g−1.
The pore size distributions are shown in Fig. 5b; these distributions were calculated by the KJS (Kruk, Jaroniec, Sayari) method.42,45 The pore size distributions for the Al-550 and Al-XCu-550; (0.01 ≤ X ≤ 0.15) samples are narrow, indicating the uniform nature of mesopores (Fig. 5). As can be seen from Fig. 5b, the shape of the PSD curves changes slightly with increasing molar fraction of Cu in the samples studied from Al-550 to Al-0.15Cu-550.
Nitrogen adsorption–desorption isotherms were also measured for the selected samples calcined at different temperatures (Al-0.05Cu-T; T = 400, 550, and 700 °C). As shown in Fig. 6a, the adsorption–desorption isotherms measured for these samples exhibit type IV isotherms with sharp capillary condensation–evaporation steps and narrow H1 hysteresis loops starting at relative pressure of about 0.55–0.70. Adsorption–desorption isotherms measured for the samples with other copper loadings and calcined at different temperatures are displayed in Fig. S3† (top panel for the Al-T samples; bottom panel for the Al-0.01Cu-T samples), S4† (top panel for the Al-0.02Cu-T samples; bottom panel for the Al-0.10Cu-T samples), and S5† (top panel for the Al-0.15Cu-T samples; bottom panel for the Al-0.20Cu-T samples) in the ESI.† As can be seen from these figures, there is no significant deviation in the shape of the adsorption isotherms and hysteresis loops, suggesting that the quality of materials did not change significantly during thermal treatment in nitrogen at 550 and 700 °C. Moreover, the hysteresis loops observed for all samples (Al-XCu-T; X = 0.01, 0.02, 0.05, 0.10, and T = 400, 550, & 700 °C) except Al-0.15Cu-T and Al-0.20Cu-T (T = 400, 550, 700 °C) are in the relatively narrow range of relative pressures from 0.55 to 0.75, indicating the presence of uniform mesopores and similar pore widths (Wmax). However, in the case of the Al-0.15Cu-T and Al-0.20Cu-T samples, the hysteresis loops for the samples calcined at high temperatures shift to low relative pressures, which is associated with a decrease in the pore widths of these samples; for instance, see the isotherm curves for Al-0.15Cu-400 and Al-0.15Cu-700 in Fig. S5† (top panel). The corresponding pore size distributions calculated from nitrogen adsorption isotherms shown in Fig. 6a, S3–S5 (ESI†) are displayed, respectively, in Fig. 6b, S6–S8† (top and bottom panels) in the ESI.† These PSD curves are narrow indicating the presence of uniform mesopores even for the samples calcined at higher temperatures. Except a few cases, the large surface areas and pore volumes are observed for all the samples calcined at 400 and 550 °C as compared to the corresponding values obtained for the samples calcined at 700 °C. For instance, the specific surface area changes from 408 m2 g−1 for Al-0.02Cu-400 to 334 m2 g−1 for Al-0.02Cu-550 and 328 m2 g−1 for Al-0.02Cu-700, and the pore volumes for these samples are in the range of 0.53–0.67 cm3 g−1.
Sorption of ammonia
The strength and the amount of basic and acidic sites of the samples studied were investigated at 50 °C by temperature programmed desorption (TPD) of CO2 and NH3, respectively. Note that the TPD technique has been widely used to study the sorption properties of various porous materials toward acidic (CO2, SO2, NO2) and basic (NH3) gases. During NH3 and CO2 TPD experiments, all calcined samples were subjected to the pretreatment with inert He gas up to the selected temperature T (T = 400, 550, 750 °C) followed by preferential pulse of suitable gas at 50 °C (NH3 and CO2 sorption), and consequently desorption by ramping temperature up to the specified value.Shown in Fig. 7 are the NH3-TPD profiles obtained for Al-550 and selected Al-XCu-550 (X = 0.10, 0.15, 0.20) samples and the total amounts of sorbed NH3 (indicator of acidity of a given sample) obtained on the basis of the aforementioned profiles (Table 2). As can be seen from Fig. 7, all NH3-TPD profiles show broad band in the temperature range of 140–350 °C reflecting the presence of acidic sites on the surface of the samples studied. Regardless of different desorption temperatures of NH3, the area of the desorption peaks shown in Fig. 7 increases with increasing amount of copper in the samples studied. For instance, the NH3 uptake changes from 0.84 mmol g−1 for Al-0.01Cu-550 to a very high value of 16.7 mmol g−1 for Al-0.20Cu-550 at 50 °C (see Table 2). The lowest and highest uptakes of NH3 of 0.76 and 16.19 mmol g−1 at 50 °C are listed for Al-550 and Al-0.20Cu-550, respectively, indicating a significant increase in the sample acidity after incorporating 20% (molar) of copper in relation to aluminum.
 | ||
| Fig. 7 NH3 TPD profiles recorded for the selected Al-550 and Al-XCu-550 samples studied (X = 0.10, 0.15, and 0.20; * refers to another batch of Al-0.20Cu-550); NH3 pulse at 50 °C. | ||
| Sample | NH3 sorption (mmol g−1) | SBET [m2 g−1] |
|---|---|---|
| a The value in bracket refers to another batch of Al-0.20Cu-550. | ||
| Al-550 | 0.76 | 232 |
| Al-0.01Cu-550 | 0.84 | 373 |
| Al-0.02Cu-550 | 3.34 | 334 |
| Al-0.05Cu-550 | 3.94 | 363 |
| Al-0.10Cu-550 | 7.21 | 329 |
| Al-0.15Cu-550 | 10.7 | 283 |
| Al-0.20Cu-550 | 16.7 (16.2) | 249 |
Sorption of carbon dioxide
One of the additional aims of this work was also to identify the basic properties of the samples studied depending on the copper loading by recording the TPD profiles for CO2 sorption at 50 °C. Shown in Fig. 8 are the CO2-TPD profiles obtained for Al-550 and Al-XCu-550 (X = 0.01 to 0.20) samples. As can be seen from this figure, the most of captured CO2 at 50 °C by the samples studied was desorbed in the range of 535–550 °C. The peaks centered at that range are much broader upon CO2 sorption, which can be possibly attributed to the formation of carbonate species upon binding CO2 to the surface of hybrid oxides. As can be seen from Table 3, no significant change in the CO2 sorption capacity was observed with increasing the percentage of Cu in the samples. The Al-550 sample showed the highest CO2 uptake of about 1.39 mmol g−1, whereas Al-0.2Cu-550 showed the lowest CO2 uptake of about 0.59 mmol g−1 at 50 °C. The CO2 uptake values for all the samples calcined at 550 °C are in the range of 0.59 and 1.39 mmol g−1. All the samples studied show smaller CO2 uptake as compared to the corresponding NH3 uptake. Thus, it can be concluded that the alumina–copper oxide samples studied exhibit somewhat similar basic properties toward CO2. Moreover, there is no significant change in the CO2 uptake for the samples calcined at different temperatures (see Fig. 9 and Table 3). | ||
| Fig. 8 CO2 TPD profiles recorded for the Al-550 and Al-XCu-550 (X = 0.01 to 0.20) samples studied; CO2 pulse at 50 °C. | ||
| Sample | CO2 sorption (mmol g−1) | SBET [m2 g−1] |
|---|---|---|
| Al-550 | 1.39 | 232 |
| Al-0.05Cu-550 | 1.11 | 363 |
| Al-0.10Cu-550 | 0.68 | 329 |
| Al-0.15Cu-550 | 0.69 | 283 |
| Al-0.20 Cu-550 | 0.59 | 249 |
| Al-0.05Cu-400 | 1.09 | 338 |
| Al-0.05Cu-550 | 1.04 | 363 |
| Al-0.05Cu-700 | 1.02 | 304 |
Interactions of NH3 and CO2 with sorbents
It is worthy to comment on possible interactions between NH3 molecules and the hybrid mesostructures studied. According to the existing literature there are at least three types of possible sorption sites available on the surface of these mesostructures at elevated temperatures (>50 °C). Firstly, terminal hydroxyl groups attached to Al and Cu (Cu–OH and Al–OH) (see Scheme S1 and the left panel of Scheme S2, ESI†). Cu–OH sites can interact with ammonia via acid/base interactions (see Scheme S1, ESI†). Secondly, Cu2+ and Al3+ species distributed on the pore walls can facilitate NH3 binding via Lewis acid/base interactions. Finally, the acid–base (Cu2+–O2−) and (Al3+–O2−) pair sites present in the hybrid matrix can attract NH3 molecules too. Some of these interactions are already well documented in literature.46–54 For instance, a shift in the OH frequencies has been observed on the FTIR spectra upon NH3 chemisorption, which suggests the possibility of forming strong hydrogen bond between NH3 and OH groups.54 This mechanism has been confirmed by recording the FTIR spectra of a given sample before and after exposure to NH3. In the current study, the area under the NH3-TPD profile increases with increasing Cu content in the hybrid samples studied from Al-0.05Cu-550 to Al-0.20Cu-550. Note that this area correlates with the amount of acid sites, while the temperature corresponding to the peak maximum reflects the strength of the sites toward releasing NH3 molecules. Since Cu species show higher affinity toward NH3 molecules than Al sites, the hybrid Cu and Al oxides with higher Cu content can bind more ammonia. Thus, Al-0.20Cu-550 shows a very high NH3 uptake (16.7 mmol g−1) as compared to that of Al-0.01Cu-550 (0.84 mmol g−1). Thus, hybrid Al and Cu oxides with high Cu content can be effectively used as NH3 sorbents.In addition, the OH-terminated surface Al species can interact with CO2 molecules to form hydrogen carbonate and bidentate carbonate structures as illustrated in Scheme S2, ESI.†55
Potential applications of aluminum–copper oxides
The hybrid oxides of Al and Cu can be used for various applications as sorbents and catalysts. Their performance depends on the surface properties, which can be controlled by chemical composition of the synthesis mixture (metal oxide precursors and surfactant/block copolymer template) and experimental conditions (calcination temperature, hydrothermal treatment temperature and time). Note that gaseous NH3 is considered as one of pollutants that significantly affects human health. Various anthropogenic activities continue to increase the NH3 level, which according to the U. S. Occupational Safety and Health Administration should not exceed 35 ppm.51,53 Thus, the development of high affinity sorbents toward ammonia is of great significance for its removal from contaminated environment. This study demonstrates that the hybrid oxides of Cu and Al show high NH3 sorption capacity, which increases with increasing Cu loading. Namely, the Al-0.20Cu-550 sample possessing the highest Cu content captured over 16 mmol g−1 of ammonia at 50 °C. Also, these hybrid oxides can be used for removal of toxic NOx species from automotive gas emissions via catalytic reduction in the presence of NH3.50,53,56 Among various catalysts the supported copper oxides saturated with NH3 showed high activity toward catalytic reduction of NOx over a wide range of conditions. In this process the adsorbed NH3 acts as reducing agent as indicated by the following reaction:50,53,56| 4NO + 4NH3 + O2 ⇄ 4N2 + 6H2O |
Conclusions
Several series of alumina mesostructures with incorporated copper species were prepared and calcined at different temperatures to examine the effect of copper loading and calcination temperature on the structural and sorption properties such as mesostructural ordering, crystallinity, porosity, surface area, thermal stability, and sorption affinity toward basic and acidic probe molecules (NH3 and CO2). The soft templating strategy used for the synthesis of the samples was shown to be very effective and feasible for the incorporation of copper species into mesoporous alumina, providing abundant options for tailoring properties of these materials such as the shape, width and volume of mesopores, the specific surface area and the surface acidity and basicity. The XRD and nitrogen adsorption data indicate high uniformity of mesopores, which is not significantly affected by increasing loading of copper. It is shown that all composite samples exhibit much higher NH3 sorption capacity as compared to the corresponding CO2 uptake. High NH3 uptake at 50 °C was observed for the samples with high copper loading indicating a substantial increase in the affinity of copper-containing samples toward basic NH3. The importance of copper species for enhancing NH3 sorption at elevated temperatures is evidenced by pulse NH3 chemisorption and TPD measurements. High NH3 sorption capacity at elevated temperatures, preferential selectivity, and low cost of the alumina–copper oxide mesostructured sorbents along with their chemical and mechanical stability shows their potential for capture of NH3 and related sorption and catalytic applications.Acknowledgements
This work was financially supported by the NSFC project (No. 21203246) and the State Scholarship Fund Program administered by the China Scholarship Council.References
- X. S. Fang, C. H Ye, X. X. Xu, T. Xie, Y. C. Wu and L. D. Zhang, J. Phys.: Condens. Matter, 2004, 16, 4157 CrossRef CAS.
- S. Kurien, J. Mathew, S. Sebastian, S. N. Potty and K. C. George, Mater. Chem. Phys., 2006, 98, 470 CrossRef CAS.
- M. Trueba and S. P. Trasatti, Eur. J. Inorg. Chem., 2005, 17, 3393 CrossRef.
- C. Marquez-Alvarez, N. Zilkova, J. Perez-Pariente and J. Cejka, Catal. Rev.: Sci. Eng., 2008, 50, 222 CAS.
- J. Cejka, Appl. Catal., A, 2003, 254, 327 CrossRef CAS.
- S. Iftekar, J. Grins, G. Svensson, J. Loof, T. Jarmar, G. A. Botton, C. M. Andrei and H. J. Engqvist, J. Eur. Ceram. Soc., 2008, 28, 747 CrossRef.
- S. E. Kim, J. H. Lim, S. C. Lee, S. C. Nam, H. G. Kang and J. Choi, Electrochim. Acta, 2008, 53, 4846 CrossRef CAS.
- Q. Yuan, A. X. Yin, C. Luo, L. D. Sun, Y. W. Zhang, W. T. Duan, H. C. Liu and C. H. Yan, J. Am. Chem. Soc., 2008, 130, 3465 CrossRef CAS PubMed.
- Q. B. Zhang, D. G. Xu, G. C. Yang, H. Huang, F. Nie, F. Liu and S. H. Yang, Prog. Mater. Sci., 2014, 60, 208 CrossRef CAS.
- I. Ali, Chem. Rev., 2012, 112, 5073 CrossRef CAS PubMed.
- X. Y. Yu, R. X. Xu, C. Gao, T. Luo, Y. Jia and J. H. Liu, ACS Appl. Mater. Interfaces, 2012, 4, 1954 CAS.
- L. P. Zhou, B. X. Wang, X. F. Peng, X. Z. Du and Y. P. Yang, Adv. Mech. Eng., 2010, 2, 1 CrossRef.
- K. J. Choi and H. W. Jang, Sensors, 2010, 10, 4083 CrossRef CAS PubMed.
- M. M. Rahman, A. J. Saleh, J. H. Jin, S. J. Ahn and J. J. Lee, Sensors, 2010, 10, 4855 CrossRef CAS PubMed.
- S. B. Wang, C. H. Hsiao, S. J. Chang, K. T. Lam, K. H. Wen and S. C. Hung, Sens. Actuators, A, 2011, 171, 207 CrossRef CAS.
- Y. W. Zhu, T. Yu, F. C. Cheong, X. J. Xu, C. T. Lim and V. B. C. Tan, Nanotechnology, 2005, 16, 88 CrossRef CAS.
- X. Zhang, W. Shi, J. Zhu, D. Kharistal, W. Zhao and B. Lalia, ACS Nano, 2011, 5, 2013 CrossRef CAS PubMed.
- C. Rossi, K. Zhang, D. Esteve, P. Alphonse, P. Tailhades and C. Vahlas, J. Microelectromech. Syst., 2007, 16, 919 CrossRef CAS.
- R. Kumar, Y. Diamant and A. Gedanken, Chem. Mater., 2000, 12, 2301 CrossRef CAS.
- M. Vaseem, A. Umar, S. H. Kim and Y.-B. Hahn, J. Phys. Chem. C, 2008, 112, 5729 CAS.
- R. Kumar, R. Elgamiel, Y. Diamant, A. Gedanken and J. Norwig, Langmuir, 2001, 17, 1406 CrossRef CAS.
- W. Ching, Y. Xu and K. Wong, Phys. Rev. B: Condens. Matter Mater. Phys., 1989, 40, 7684 CrossRef CAS.
- S. F. Tikhov, V. A. Sadykov, G. N. Kryukova and E. A. Paukshtis, J. Catal., 1992, 134, 506 CrossRef CAS.
- L. Xu, S. Sithambaram, Y. Zhang and C.-h. Chen, Chem. Mater., 2009, 21, 1253 CrossRef CAS.
- K. Zhou and Y. Li, Angew. Chem., Int. Ed., 2012, 51, 602 CrossRef CAS PubMed.
- K. Zhou, R. Wang, B. Xu and Y. Li, Nanotechnology, 2006, 17, 3939 CrossRef CAS.
- M. Jayamurthy, J. Catal., 2014, 309, 11 CrossRef.
- Z. H. Zhu, H. Y. Zhu, S. B. Wang and G. Q. Lu, Catal. Lett., 2003, 91, 73 CrossRef CAS.
- H. W. Pennline and J. S. Hoffman, Fuel Process. Technol., 2013, 114, 109 CrossRef CAS.
- N. P. Tangale, Appl. Catal., A, 2013, 467, 421 CrossRef CAS.
- B. Y. Jibril, J. Ind. Eng. Chem., 2013, 19, 1800 CrossRef CAS.
- A. Bansiwal, P. Pillewan, B. Biniwale and S. Rayalu, Microporous Mesoporous Mater., 2010, 129, 54 CrossRef CAS.
- P. Pillewana, S. Mukherjeea, T. Roychowdhuryb and S. Dasc, J. Hazard. Mater., 2011, 186, 367 CrossRef PubMed.
- Q. Liu, A. Wang and X. Wang, Chem. Mater., 2006, 18, 5153 CrossRef CAS.
- Z. Wu, Q. Li, D. Feng, P. A. Webley and D. Zhao, J. Am. Chem. Soc., 2010, 132, 12042 CrossRef CAS PubMed.
- S. M. Grant and M. Jaroniec, J. Mater. Chem., 2012, 22, 86 RSC.
- K. Niesz, P. Yang and G. A. Somorjai, Chem. Commun., 2005, 1986 RSC.
- W. Cai, J. Yu, C. Anand, A. Vinu and M. Jaroniec, Chem. Mater., 2011, 23, 1147 CrossRef CAS.
- X. Jiang, H. Oveisi, Y. Nemoto, N. K. Suzuki, C. W. Wu and Y. Yamauchi, Dalton Trans., 2011, 40, 10851 RSC.
- X. Y. Wang, D. H. Pan, M. Guo and M. He, Mater. Lett., 2013, 97, 27 CrossRef CAS.
- D. Lutic, M. Sanati, A. L. Spetz, Y. Xu, W. A. Shelton and W. F. Schneider, in Synthesis, properties and application of oxide nanomaterials, ed. J. A. Rodriquez and M. Fernandez-Garcia, John Wiley & Sons, Inc., Hoboken, NJ, 2007, ch. 15 and 20 Search PubMed.
- M. Kruk and M. Jaroniec, Chem. Mater., 2001, 13, 3169 CrossRef CAS.
- F. Rashidi, A. N. Kharat, A. M. Rashidi and E. Lima, Eur. J. Inorg. Chem., 2010, 2010, 1544 CrossRef.
- C. Y. Hua, K. Shihb and J. O. Leckiec, J. Hazard. Mater., 2010, 181, 399 CrossRef PubMed.
- M. Jaroniec and L. Solovyov, Langmuir, 2006, 22, 6757 CrossRef CAS PubMed.
- I. L. Gonzalez, U. Deka, B. Arstad, A. V. Y. D. Deyne, K. Hemelsoet, M. Waroquier, V. V. Speybroeck, B. M. Weckhuysen and A. M. Beale, Phys. Chem. Chem. Phys., 2014, 16, 1639 RSC.
- H. Zhu, J. H. Kwak, C. H. F. Peden and J. Szanyi, Catal. Today, 2013, 205, 16 CrossRef CAS.
- I. L. Gonzalez, D. S. Wragg, W. A. Slawinski, K. Hemelsoet, A. V. Y. D. Deyne, M. Waroquier, V. V. Speybroeck and A. M. Beale, J. Phys. Chem. C, 2015, 119, 24393 Search PubMed.
- A. M. B. Furtado, J. Liu, Y. Wang and M. D. LeVan, J. Mater. Chem., 2011, 21, 6698 RSC.
- F. Giordanino, E. Borfecchia, K. A. Lomachenko, A. Lazzarini, G. Agostini, E. Gallo, A. V. Soldatov, P. Beato, S. Bordiga and C. Lamberti, J. Phys. Chem. Lett., 2014, 5, 1552 CrossRef CAS PubMed.
- C. Petit and T. J. Bandosz, Adv. Funct. Mater., 2011, 21, 2108 CrossRef CAS.
- E. Liu, B. Sarkar, L. Wang and R. Naidu, Appl. Clay Sci., 2016, 121, 154 CrossRef.
- M. Trombetta, G. Ramis, G. Busca, B. Montanari and A. Vaccari, Langmuir, 1997, 13, 4628 CrossRef CAS.
- D. Stoilova, K. Cheshkova and R. Nickolov, React. Kinet. Catal. Lett., 1999, 68, 331–337 CrossRef CAS.
- C. Gunathilake and M. Jaroniec, J. Mater. Chem. A, 2015, 3, 2707 CAS.
- J. M. G. Amores, V. S. Escribano, G. Ramis and G. Busca, Appl. Catal., B, 1997, 13, 45 CrossRef.
Footnotes |
| † Electronic supplementary information (ESI) available: 8 figures showing XRD patterns, N2 adsorption–desorption isotherms, and pore size distributions and 2 schemes showing NH3 and CO2 binding models on the sorbents studied. See DOI: 10.1039/c6ra01019a |
| ‡ Authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2016 |