A new complex alkali metal aluminium amide borohydride, Li2Al(ND2)4BH4: synthesis, thermal analysis and crystal structure†
S. Hinoa,
T. Ichikawab,
Y. Kojimac,
M. H. Sørby*a and
B. C. Haubacka
aPhysics Department, Institute for Energy Technology, P. O. Box 40, NO-2027 Kjeller, Norway. E-mail: magnuss@ife.no; Fax: +47 6381 0920; Tel: +47 6380 6000
bGraduate School of Integrated Arts and Sciences, Hiroshima University, 1-3-1 Kagamiyama, Higashi-Hiroshima, 739-8530, Japan
cInstitute for Advanced Materials Research, Hiroshima University, 1-3-1 Kagamiyama, Higashi-Hiroshima, 739-8530, Japan
First published on 14th March 2016
Abstract
The mixtures of alkali metal aluminium amide MAl(ND2)4 and alkali metal borohydride MBH4 were synthesized using ball-milling. Thermal analysis and phase identifications of the MAl(ND2)4–MBH4 system were performed by differential scanning calorimetry-thermogravimetric analysis, temperature programmed desorption-residual gas analysis, in situ synchrotron radiation powder X-ray diffraction and Fourier transform infrared spectroscopy. A previously unknown phase, Li2Al(ND2)4BH4, was formed in the samples of LiAl(ND2)4 + 2LiBH4 and NaAl(ND2)4 + 2LiBH4. Its crystal structure was solved and refined in the monoclinic space group P21/c, with a = 6.8582(3) Å, b = 11.3128(5) Å, c = 11.2117(4) Å, β = 124.186(5)°. Li2Al(ND2)4BH4 is the first reported compound containing both [Al(ND2)4]− and BH4− anions. The ball-milling of NaAl(ND2)4 + 2NaBH4 and KAl(ND2)4 + 2KBH4 gave mixtures of the starting materials. The synthesized samples desorbed ammonia in the temperature range between 50 and 200 °C and hydrogen above ∼300 °C.
Introduction
Complex hydrides, e.g., metal alanates (MAlH4), borohydrides (MBH4) and amides (MNH2), are ionic compounds of metal cations and covalently bonded complex anions (AlH4−, BH4−, NH2−). They have been studied as hydrogen storage materials possessing high gravimetric and volumetric hydrogen densities.1 Research has been focused on tailoring the properties of the system to have favourable kinetics and thermodynamics for hydrogen desorption and absorption at moderate temperature.2,3Lithium amide (LiNH2) desorbs ammonia on thermal decomposition above ∼300 °C, while the mixture with LiH desorbs hydrogen at lower temperature (>200 °C).4 The dehydrogenated product is Li2NH and higher temperature (>320 °C) is necessary for complete dehydrogenation into Li3N.5 The reversible hydrogen desorption/absorption reactions are described as follows:
| LiNH2 + 2LiH ↔ Li2NH + LiH + H2 ↔ Li3N + 2H2. |
In the LiNH2–LiBH4 mixture it has been assumed that Li3BN2 is the hydrogen-free dehydrogenated state:6,7
| 2LiNH2 + LiBH4 → Li3BN2 + 4H2. |
As a result of the stabilization of the dehydrogenated state, less positive enthalpy (23 kJ per mol H2) for the dehydrogenation reaction would be required compared to pure LiBH4 (75 kJ per mol H2) according to DFT calculations.7 This results in release of ∼10 wt% hydrogen at relatively low temperature around 250 °C.6 It is also reported that an intermediate phase (Li4BN3H10) is formed after ball milling of the mixture of LiNH2 and LiBH4.8
Metal aluminium amide (MAl(NH2)4), are compounds where H− in metal alanate are substituted by NH2− ions. They desorbs ammonia at around 100 °C for M = Li, Na and K.9,10 In analogy to the LiNH2–LiH system, the LiAl(NH2)4–LiH system has been studied. The ball-milled mixture of LiAl(NH2)4 + 4LiH desorbed >5.0 wt% of H2 at 130 °C.11 In the present study, the mixtures of alkali metal aluminium amides and borohydrides were prepared and thermal analysis and phase identification of the mixtures were performed.
Experimental
LiBH4 (≥95%), NaBH4 (≥99%) and KBH4 (≥98%) were purchased from Sigma-Aldrich and LiAl(ND2)4, NaAl(ND2)4 and KAl(ND2)4 were synthesized as described elsewhere.12 Mixtures of LiAl(ND2)4–LiBH4 (denoted as S1), NaAl(ND2)4–LiBH4 (S2), NaAl(ND2)4–NaBH4 (S3) and KAl(ND2)4–KBH4 (S4) in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratios were ball-milled for 1 h (15 min milling + 15 min rest, 4 cycles) under 0.1 MPa Ar atmosphere using a Pulverisette 7 (Fritsch). The samples studied in this paper are shown in Table 1. Sample handling was carried out in a MBraun Unilab glovebox filled with purified Ar (<1 ppm O2 and H2O).
2 molar ratios were ball-milled for 1 h (15 min milling + 15 min rest, 4 cycles) under 0.1 MPa Ar atmosphere using a Pulverisette 7 (Fritsch). The samples studied in this paper are shown in Table 1. Sample handling was carried out in a MBraun Unilab glovebox filled with purified Ar (<1 ppm O2 and H2O).
Powder X-ray diffraction (PXD) patterns were collected using Cu Kα radiation in a Bruker-AXS D8 Advance diffractometer equipped with a Göbel mirror and a LynxEye 1D strip detector. The samples were contained in rotating boron glass capillaries (0.8 mm ϕ) filled and sealed under Ar atmosphere.
Fourier transform infrared (FTIR) spectrometer (ALPHA, Bruker) equipped with a 45° diamond attenuated total reflectance (ATR) accessory was used to collect IR spectra at room temperature (RT). The spectra were obtained in the range of 4000–400 cm−1 with a resolution of 2 cm−1 without any dilution of the samples.
Temperature programmed desorption (TPD) was performed under dynamic vacuum up to 500 °C using an in-house built setup. The samples were heated from RT to 500 °C with a ramp rate of 2 °C min−1. The desorbed gas was analyzed with a MKS Microvision-IP residual gas analyzer (RGA).
Combined differential scanning calorimetry-thermogravimetric analysis (DSC-TGA) was carried out using a Netzsch STA 449 F3 Jupiter instrument with an Al sample crucible covered by a lid with a small hole. The samples were heated from RT to 500 °C with a ramp rate of 5 °C min−1 under a 50 mL min−1 Ar flow.
In situ synchrotron radiation (SR) PXD measurements (λ = 0.694118 Å) were carried out using a pixel area detector (Pilatus 2M, DECTRIS) at the Swiss-Norwegian Beamlines (SNBL, BM01A) at the European Synchrotron Radiation Facility (ESRF), Grenoble, France. The samples were mounted in boron glass capillaries (0.5 mm ϕ) fixed in a Swagelok fitting and kept under dynamic vacuum. A diffraction pattern was collected every 40 s (exposure time 30 s). The capillary was rotated 30° during exposure to improve the powder averaging. Measurements were carried out between RT and 500 °C with a ramp rate of 5 °C min−1. The two-dimensional data were integrated to one-dimensional diffraction patterns using the Fit2D program.13
The Dicvol14 and Chekcell15 programs were used for unit cell and space group determination. The global optimization approach with parallel tempering as implemented in the FOX program16,17 was used for crystal structure determination. The structure refinements were carried out using the GSAS18 software package with the EXPGUI19 user interface based on the Rietveld method.20,21 The backgrounds were fitted by a 36-term Chebyshev polynomial. A pseudo-Voigt function22 with asymmetry correction23 was used to model the peak profile. Isotropic displacement parameters were refined with a common Uiso value for D- and H-atoms, respectively. The crystal structures were illustrated using the VESTA program.24
Results and discussion
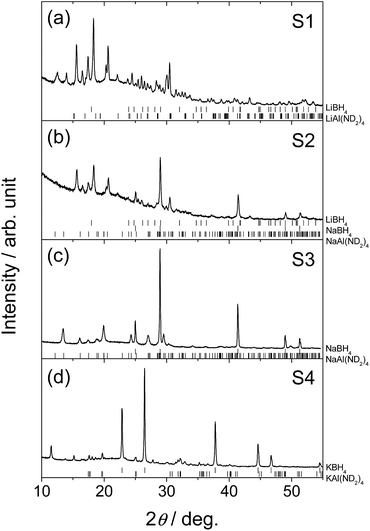
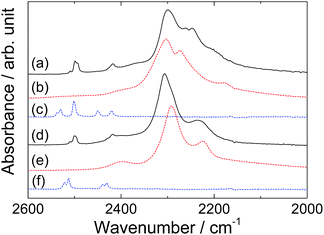
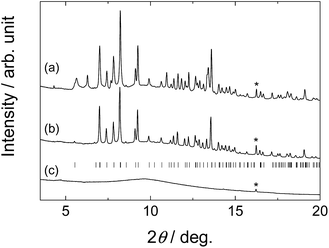
Fig. 1 shows PXD patterns of the samples after ball-milling. For sample S1, Bragg peaks from new phase(s) were observed, while no peaks remained from the starting materials. This indicates a complete chemical reaction. Similar for S2, the starting materials were not observed, and peaks from a new phase as well as NaBH4 were found. The unidentified peaks in S2 are a subset of the unidentified peaks in the S1 sample. This indicates the possibilities that S1 contain the new phase observed in S2 in addition to other unidentified phase(s) or that the crystal structure of the phase in S1 has a superstructure of the one in S2. To our knowledge, the unidentified peaks from S1 and S2 do not match the reported structure of the Li–Al–B–N–H related compounds, such as LiNH2BH3,25 LiBH4NH3,26 Li4BH4(NH2)3,8 Li2BH4NH2,27 Al(BH4)3,28 Al(NH3)6Li2(BH4)5,29 Al(BH4)3NH3 (ref. 30) and Al3Li4(BH4)13.31 For S3 only peaks from the starting materials were observed. For S4 the main peaks are also from the starting material, but in addition a small amount of a partially decomposed product9 of KAl(ND2)4, i.e., KAl(ND2)2ND is observed. This indicates that no chemical reaction occurred between the reactants during ball-milling.ATR-IR spectra of S1, S2 and the starting materials of each sample as references are shown in Fig. 2. The peak positions are summarized in Table 2. Peaks corresponding to N–D and B–H stretching modes were observed in the IR spectra of both S1 and S2. Isotopic exchange between ND2 and BH4 groups was negligible since no peaks of N–H or B–D stretching mode were observed. The peak profile of B–H stretching mode in S1 was different from pure LiBH4 (Fig. 2b). Two main peaks were observed at 2303 and 2273 cm−1 in LiBH4, while three peaks (2299, 2261 and 2247 cm−1) were present in S1. Peaks corresponding to asymmetric stretching of ND2 at 2537, 2530 and 2450 cm−1 in LiAl(ND2)4 (Fig. 2c) were not apparent in S1 but the peaks at 2509 and 2426 (shoulder) might be due to ND2 stretching. Peak splitting (2499 and 2493 cm−1) and slight red-shift were observed for symmetric stretching mode of ND2 in S1. For sample S2, two main peaks in B–H region were observed at 2306 and 2236 cm−1, respectively, and the most intense peak in NaBH4 (2291 cm−1) was present as a shoulder. Peaks corresponding to N–D stretching modes were almost the same as those in S1 and different from NaAl(ND2)4. Therefore, the unidentified phase(s) in S2 could contain both BH4 and ND2 ions. Here, we assume the composition of the phase to be Li2Al(ND2)4BH4. From the PXD-data (Fig. 1), it is suggested that the following reaction occur during preparation of S2:
| NaAl(ND2)4 + 2LiBH4 → Li2Al(ND2)4BH4 + NaBH4 |
| S1 | LiBH4 | Modea | LiAl(ND2)4 | Modeb | S2 | NaBH4 | Modea | NaAl(ND2)4 | Modeb |
|---|---|---|---|---|---|---|---|---|---|
| a From ref. 32.b From ref. 33. The subscript a and s denotes asymmetric and symmetric vibration, respectively. The description (a) and (b) corresponds to two types of different ND2 groups. | |||||||||
| 2537 | νaND2(a) | ||||||||
| 2509 | 2530 | 2509 | 2522 | νaND2 | |||||
| 2499 | 2501 | νsND2(a) | 2499 | 2512 | |||||
| 2493 | 2496 | ||||||||
| 2426sh | 2450 | νaND2(b) | 2427sh | 2438 | νsND2 | ||||
| 2417 | 2420 | νsND2(b) | 2418 | 2430 | |||||
| 2397 | ν2 + ν4(A′′)/2ν(A′) | 2396 | ν2 + ν4 | ||||||
| 2299 | 2303 | ν3(A′) | 2306 | 2291 | ν3 | ||||
| 2261 | 2273 | ||||||||
| 2247 | |||||||||
| 2177 | 2ν4(A′′) | 2236 | 2222 | 2ν4 | |||||
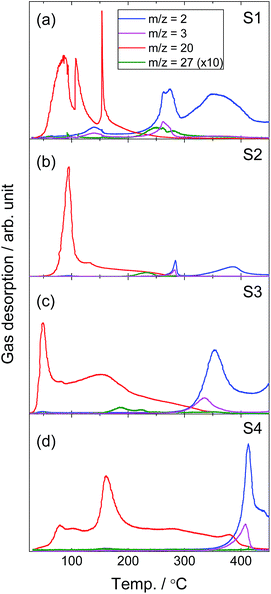
Gas desorption properties of the samples were investigated using TPD-RGA measurement (Fig. 3). By analogy with LiNH2–LiBH4 and LiAl(NH2)4–LiH systems,6,11 dominant release of hydrogen is expected, however, ND3 (m/q = 20) was desorbed from all the samples above 50 °C. H2 (m/q = 2) desorption was observed only above 250 °C in S1 and S2, 300 °C in S3 and 350 °C in S4. As described above from the IR data, H–D exchange between ND2 and BH4 groups was not apparent after ball-milling. HD (m/q = 3) would be released as a result of direct interaction of hydrogen atoms in the amide (ND2−) and borohydride (BH4−) ions. However, HD signals were synchronized with H2 signals and less intense than H2. This suggests that HD was generated from H2 and D from ND3 in the RGA chamber during ionization as a fragment of H2 desorption. Small amounts of B2H6 (m/q = 27) were also released above 220 °C, 200 °C and 170 °C in S1, S2 and S3, respectively, while only a trace was detected from S4. The H2 desorption temperature of S3 is much lower compared to the ball-milled NaBH4 (>490 °C)34 and similar to the NaNH2–NaBH4 system (>300 °C).35 For MNH2–MBH4 it is proposed that an attraction between protic Hδ+ in NH2 and hydridic Hδ− in BH4 could accelerate the hydrogen desorption.36 For the MAl(ND2)4–MBH4 samples an attraction between Dδ+ in ND2 (or ND in metal aluminium imide MAl(ND)2) and hydridic Hδ− in BH4 could lower the hydrogen desorption temperature.
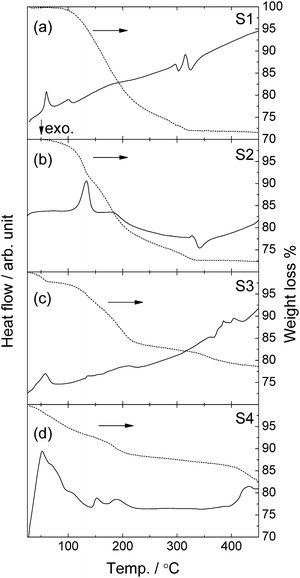
TGA-DSC profiles of the samples are shown in Fig. 4. Two endothermic events at 60 and 100 °C without weight change were observed in S1, which might be due to phase transition(s). Ammonia desorption reactions appear as endothermic events in S2 (135 °C), S3 (60 °C) and S4 (150 °C). Weight losses below 250 °C were 23.8, 23.2, 16.7 and 11.8 wt% in the S1–S4 samples, respectively. The theoretical weight loss for 2 mole of ND3 release which corresponds to decomposition of metal aluminium amides into metal aluminium imides (MAl(NH)2) are 26.8, 24.2, 20.2 and 16.2 wt% in S1–S4, respectively. Considering the low decomposition temperatures of NaAl(NH2)4 and KAl(NH2)4, part of the aluminium amides would be decomposed during ball-milling in S3 and S4. Actually, PXD data shows the formation of the partially decomposed product in S4. Moreover, the end points of ammonia desorption are not very well defined, and some desorption continues also above 250 °C. The total mass losses up to 450 °C, which include hydrogen desorption, are 28.5, 27.7, 21.4 and 17.4 wt% for S1–S4, respectively. These are close to values for desorption of 2H2 in addition to 2 ND3, which are 29.5, 26.6, 22.3 and 17.9 wt%. This thus matches hydrogen desorption from the BH4 unit in Li2Al(ND2)4BH4 in S1 and S2. It also indicate that a similar type of reaction takes place at elevated temperature in S3 and S4, since unreacted NaBH4 and KBH4 should not emit hydrogen in the investigated temperature range. The inconsistency of the temperatures for ammonia desorption between TGA-DSC and RGA resulted from different experimental conditions in these measurements. While the samples were under dynamic vacuum in the RGA measurements, an Al sample container was covered by a lid with a small hole in the TGA-DSC measurements. Thus, for TGA-DSC the decomposition can be suppressed by ammonia partial pressure around the sample. Moreover, faster heating rate was used in the TGA-DSC experiments (5 °C min−1) than in RGA (2 °C min−1).
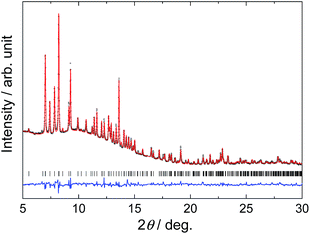
Selected diffraction patterns of in situ SR-PXD data for the S1 sample are shown in Fig. 5 (the rest of the in situ SR-PXD data for S1 and S2 are given as ESI†). The peaks that were not observed in S2 disappear around 50 °C on heating S1 under vacuum. The remaining peaks, thus corresponding to those observed from S2, were successfully indexed with a monoclinic unit cell, P21/c, a = 6.8582(3) Å, b = 11.3128(5) Å, c = 11.2117(4) Å, β = 124.186(5)° at 53 °C. Crystal structure determination and refinement was performed, confirming the suggested composition Li2Al(ND2)4BH4. For the structure refinement, ND2 and BH4 units were treated as rigid bodies and the positions of hydrogen atoms are uncertain. N–D and B–H distances of 1.01 and 1.25 Å, D–N–D and H–B–H angles of 98.19 and 109.47°, respectively, were applied for the rigid bodies. The fit obtained in the Rietveld refinement based on the in situ SR-PXD data at 53 °C for S1 is shown in Fig. 6. Table 3 summarizes the structural parameters of Li2Al(ND2)4BH4 and the structure model is visualized in Fig. 7. Al is tetrahedrally coordinated by ND2 groups and the average Al–N distance is 1.974 Å which is slightly longer than that in LiAl(ND2)4 (1.868 Å).12 The average Li–N distance (2.16 Å) of Li2Al(ND2)4BH4 is almost the same as that of LiAl(ND2)4 (2.100 Å). In LiBH4, BH4 group is tetrahedrally coordinated by Li atoms (average Li–B distance, 2.515 Å),37 while planar Li2B2 units are linked by corner sharing where each B is shared between 2 planar units in Li2Al(ND2)4BH4 (average Li–B distance, 2.63 Å), yielding a 4-coordination which is neither tetrahedral nor planar. The average distance between N in ND2 group and B in BH4 group is 3.955 Å, and ND2 and BH4 groups are well-separated (3.77 Å or more).
| Space group: P21/c (no. 14), Z = 4, T = 53 °C | |||||
|---|---|---|---|---|---|
| a = 6.8582(3) Å, b = 11.3128(5) Å, c = 11.2117(4) Å, β = 124.186(5)° | |||||
| Atom | Site | x | y | Z | Uiso/Å2 |
| Li1 | 4e | 0.935(5) | 0.477(2) | 0.354(3) | 0.10(1) |
| Al | 4e | 0.1187(8) | 0.3074(4) | 0.8821(4) | 0.060(2) |
| N1 | 4e | 0.0269(11) | 0.1716(7) | 0.7549(5) | 0.024(3) |
| D1 | 4e | 0.0314(11) | 0.1063(7) | 0.6951(5) | 0.80(10) |
| D2 | 4e | 0.1837(11) | 0.1554(7) | 0.8481(5) | 0.80(10) |
| N2 | 4e | 0.8126(14) | 0.3829(5) | 0.8405(7) | 0.060(3) |
| D3 | 4e | 0.8421(14) | 0.3610(5) | 0.9367(7) | 0.80(10) |
| D4 | 4e | 0.8913(14) | 0.3125(5) | 0.8290(7) | 0.80(10) |
| N3 | 4e | 0.3151(15) | 0.2609(6) | 0.0817(10) | 0.064(4) |
| D5 | 4e | 0.4121(15) | 0.2181(6) | 0.1770(10) | 0.80(10) |
| D6 | 4e | 0.4112(15) | 0.3360(6) | 0.1104(10) | 0.80(10) |
| N4 | 4e | 0.2822(13) | 0.4280(6) | 0.8437(7) | 0.047(3) |
| D7 | 4e | 0.3745(13) | 0.4957(6) | 0.8398(7) | 0.80(10) |
| D8 | 4e | 0.1236(13) | 0.4469(6) | 0.7530(7) | 0.80(10) |
| B | 4e | 0.6969(17) | 0.0906(7) | 0.9505(8) | 0.013(4) |
| H1 | 4e | 0.5227(17) | 0.0230(7) | 0.8924(8) | 0.10(2) |
| H2 | 4e | 0.6572(17) | 0.1782(7) | 0.0018(8) | 0.10(2) |
| H3 | 4e | 0.7334(17) | 0.1244(7) | 0.8585(8) | 0.10(2) |
| H4 | 4e | 0.8743(17) | 0.0369(7) | 0.0492(8) | 0.10(2) |
| Li2 | 4e | 0.560(5) | 0.124(2) | 0.120(3) | 0.15(1) |
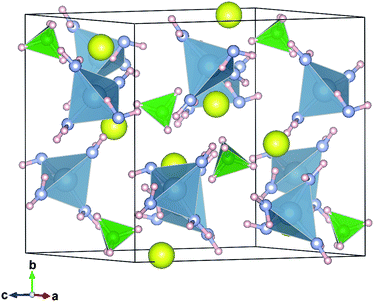 | ||
| Fig. 7 Visualization of the crystal structure of Li2Al(ND2)4BH4. Li: yellow spheres, Al: in centers of blue tetrahedra, N: gray spheres, B: in centers of green tetrahedra, H(D): pink spheres. | ||
After heating up to ∼80 °C in Fig. 5c, the Bragg peaks of the monoclinic Li2Al(ND2)4BH4 disappeared as a result of amorphization and/or decomposition. It should be noted that metallic Li was observed in the PXD profile of S1 while it was found neither in the starting material LiAl(ND2)4 nor in S2. This indicates that metallic Li is formed in a redox process in conjunction with formation of the unknown phase which is only observed in S1.
Conclusions
Thermal and phase analyses of the MAl(ND2)4–MBH4 systems were carried out by means of TGA-DSC, RGA, PXD and IR measurements. Ball-milled mixtures of LiAl(ND2)4 + 2LiBH4 (S1), NaAl(ND2)4 + 2LiBH4 (S2), NaAl(ND2)4 + 2NaBH4 (S3) and KAl(ND2)4 + 2KBH4 (S4) desorbed ammonia in the temperature range between 50 and 200 °C and hydrogen above ∼300 °C. From the PXD results, mixture of the starting materials were found in the sample of NaAl(ND2)4 + 2NaBH4 (S3) and KAl(ND2)4 + 2KBH4 (S4), while previously undescribed phases were observed in the sample of LiAl(ND2)4 + 2LiBH4 (S1) and NaAl(ND2)4 + 2LiBH4 (S2). Structure determination of the novel phase Li2Al(ND2)4BH4, found in both S1 and S2, was performed and it is revealed that the phase has the monoclinic unit cell (P21/c) which contains [Al(ND2)4]− and BH4− anions.Acknowledgements
The EU FP7 Marie Curie Action Incoming International Fellowship project MATERHY (FP7-PEOPLE-2009-IIF, no. 253863) is acknowledged for financial support. The authors acknowledge the skillful assistance from the staff of the Swiss-Norwegian Beamline, at the European Synchrotron Radiation Facility, Grenoble, France.References
- S. Orimo, Y. Nakamori, J. R. Eliseo, A. Zuettel and C. M. Jensen, Chem. Rev., 2007, 107, 4111–4132 CrossRef CAS PubMed.
- J. Wang, H.-W. Li and P. Chen, MRS Bull., 2013, 38, 480–487 CrossRef CAS.
- L. H. Jepsen, M. B. Ley, Y.-S. Lee, Y. W. Cho, M. Dornheim, J. O. Jensen, Y. Filinchuk, J. E. Jorgensen, F. Besenbacher and T. R. Jensen, Mater. Today, 2014, 17, 129–135 CrossRef CAS.
- T. Ichikawa, S. Isobe, N. Hanada and H. Fujii, J. Alloys Compd., 2004, 365, 271–276 CrossRef CAS.
- P. Chen, Z. T. Xiong, J. Z. Luo, J. Y. Lin and K. L. Tan, Nature, 2002, 420, 302–304 CrossRef CAS PubMed.
- F. E. Pinkerton, G. P. Meisner, M. S. Meyer, M. P. Balogh and M. D. Kundrat, J. Phys. Chem. B, 2005, 109, 6–8 CrossRef CAS PubMed.
- M. Aoki, K. Miwa, T. Noritake, G. Kitahara, Y. Nakamori, S. Orimo and S. Towata, Appl. Phys. A: Mater. Sci. Process., 2005, 80, 1409–1412 CrossRef CAS.
- Y. E. Filinchuk, K. Yvon, G. P. Meisner, F. E. Pinkerton and M. P. Balogh, Inorg. Chem., 2006, 45, 1433–1435 CrossRef CAS PubMed.
- R. Brec and J. Rouxel, Bull. Soc. Chim. Fr., 1968, 2721–2726 CAS.
- T. Ono, K. Shimoda, M. Tsubota, S. Hino, K. Kojima, T. Ichikawa and Y. Kojima, J. Alloys Compd., 2010, 506, 297–301 CrossRef CAS.
- R. Janot, J. B. Eymery and J. M. Tarascon, J. Phys. Chem. C, 2007, 111, 2335–2340 CAS.
- S. Hino, H. Grove, T. Ichikawa, Y. Kojima, M. H. Sørby and B. C. Hauback, Int. J. Hydrogen Energy, 2015, 40, 16938–16947 CrossRef CAS.
- A. P. Hammersley, FIT2D: An Introduction and Overview, 1997 Search PubMed.
- A. Boultif and D. Louer, J. Appl. Crystallogr., 1991, 24, 987–993 CrossRef CAS.
- B. L. Bochu and J. Laugier, Suite of Programs for the Interpretation of X-ray Experiments, http://www.ccp14.ac.uk/tutorial/lmgp Search PubMed.
- V. Favre-Nicolin and R. Cerny, J. Appl. Crystallogr., 2002, 35, 734–743 CrossRef CAS.
- V. Favre-Nicolin and R. Cerny, Z. Kristallogr., 2004, 219, 847–856 CAS.
- A. C. Larson and R. B. Von Dreele, General Structure Analysis System (GSAS), 1994 Search PubMed.
- B. H. Toby, J. Appl. Crystallogr., 2001, 34, 210–213 CrossRef CAS.
- H. M. Rietveld, Acta Crystallogr., 1967, 22, 151–152 CrossRef CAS.
- H. M. Rietveld, J. Appl. Crystallogr., 1969, 2, 65–71 CrossRef CAS.
- P. Thompson, D. E. Cox and J. B. Hastings, J. Appl. Crystallogr., 1987, 20, 79–83 CrossRef CAS.
- L. W. Finger, D. E. Cox and A. P. Jephcoat, J. Appl. Crystallogr., 1994, 27, 892–900 CrossRef CAS.
- K. Momma and F. Izumi, J. Appl. Crystallogr., 2011, 44, 1272–1276 CrossRef CAS.
- H. Wu, W. Zhou and T. Yildirim, J. Am. Chem. Soc., 2008, 130, 14834–14839 CrossRef CAS PubMed.
- S. R. Johnson, W. I. F. David, D. M. Royse, M. Sommariva, C. Y. Tang, F. P. A. Fabbiani, M. O. Jones and P. P. Edwards, Chem.–Asian. J., 2009, 4, 849–854 CrossRef CAS PubMed.
- P. A. Chater, W. I. F. David and P. A. Anderson, Chem. Commun., 2007, 4770–4772 RSC.
- K. Miwa, N. Ohba, S. Towata, Y. Nakamori, A. Zuettel and S. Orimo, J. Alloys Compd., 2007, 446, 310–314 CrossRef.
- Y. Guo, H. Wu, W. Zhou and X. Yu, J. Am. Chem. Soc., 2011, 133, 4690–4693 CrossRef CAS PubMed.
- É. B. Lobkovskii, V. B. Polyakova, S. P. Shilkin and K. N. Semenenko, J. Struct. Chem., 1975, 16, 66–72 CrossRef.
- I. Lindemann, R. Domenech Ferrer, L. Dunsch, Y. Filinchuk, R. Cerny, H. Hagemann, V. D'Anna, L. M. L. Daku, L. Schultz and O. Gutfleisch, Chem.–Eur. J., 2010, 16, 8707–8712 CrossRef CAS PubMed.
- O. Zavorotynska, M. Corno, A. Damin, G. Spoto, P. Ugliengo and M. Baricco, J. Phys. Chem. C, 2011, 115, 18890–18900 CAS.
- R. Brec, A. Novak and J. Rouxel, Bull. Soc. Chim. Fr., 1967, 2432–2435 CAS.
- J. Mao, Z. Guo, X. Yu and H. Liu, J. Phys. Chem. C, 2011, 115, 9283–9290 CAS.
- P. A. Chater, P. A. Anderson, J. W. Prendergast, A. Walton, V. S. J. Mann, D. Book, W. I. F. David, S. R. Johnson and P. P. Edwards, J. Alloys Compd., 2007, 446, 350–354 CrossRef.
- Y. Zhang and Q. F. Tian, Int. J. Hydrogen Energy, 2011, 36, 9733–9742 CrossRef CAS.
- J. P. Soulié, G. Renaudin, R. Černý and K. Yvon, J. Alloys Compd., 2002, 346, 200–205 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available: In situ SR-PXD data for ball-milled samples of LiAl(ND2)4 + 2LiBH4 and NaAl(ND2)4 + 2LiBH4. See DOI: 10.1039/c6ra00931j |
| This journal is © The Royal Society of Chemistry 2016 |