Epoxidised soybean oil–Cu/Cu2O bio-nanocomposite material: synthesis and characterization with antibacterial activity†
Abstract
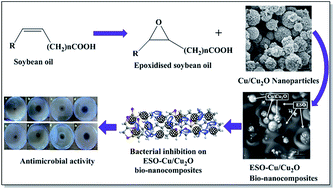
A facile approach for the synthesis of a novel epoxidised soybean oil–Cu/Cu2O (ESO–Cu/Cu2O) bio-nanocomposite material via ultrasound irradiation was investigated. The synthesis was carried out using epoxidised soybean oil (ESO) and Cu/Cu2O nanoparticles (NPs) in presence of 4-dimethylaminopyridine (DMAP). In this composite, Cu/Cu2O NPs homogeneously anchored onto epoxidised soybean oil. The morphology and elemental compositions of the bio-nanocomposite material were determined with the help of scanning electron microscopy, energy dispersive X-ray spectroscopy, and FTIR spectroscopy analysis. Investigation of plant-derived antibacterial materials as alternatives to synthetic antibacterial agents has generated great interest recently. In that direction, the antibacterial activity of resulting bio-nanocomposite material against Staphylococcus aureus as well as Escherichia coli bacteria was investigated. The synthesized ESO–Cu/Cu2O bio-nanocomposite material showed better antibacterial activity as compared to ESO, DMAP, and Cu/Cu2O NPs alone for the first time. This study endorses the bio-nanocomposite as a promising antibacterial material in a wide range of biological applications.

 Please wait while we load your content...
Please wait while we load your content...