Phosphine-catalyzed domino reactions of alkynyl ketones with sulfonylhydrazones: construction of diverse pyrazoloquinazoline derivatives†
Abstract
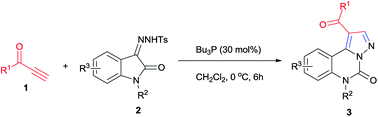
An efficient method for the synthesis of C-1 position acyl substituted pyrazoloquinazoline derivatives via a PBu3-promoted regioselective domino process of sulfonylhydrazones and alkynyl ketones was developed, which may be potentially useful for drug discovery.

 Please wait while we load your content...
Please wait while we load your content...