In situ synthesis of Cu/Ni alloy nanoparticles embedded in thin polymer layers†
Ryo Shimizu,
Takaya Kawakami,
Yohei Takashima,
Takaaki Tsuruoka and
Kensuke Akamatsu*
Department of Nanobiochemistry, Frontiers of Innovative Research in Science and Technology (FIRST), Konan University, 7-1-20 Minatojimaminami, Chuo-ku, Kobe 650-0047, Japan. E-mail: akamatsu@center.konan-u.ac.jp
First published on 9th February 2016
Abstract
In situ formation of Cu–Ni alloy nanoparticles with controlled composition in thin polyimide layers was investigated. The ferromagnetic properties of the nanoparticles were found to depend on the composition and the interparticle distance between the nanoparticles.
Nanocomposites consisting of polymer matrices containing metal nanoparticles are expected to be useful in next-generation optical, electrical and biological applications.1–5 The unique characteristics of nanocomposites are largely dependent on several structural variables, including particle size, concentration and composition, as well as on the physical properties of the surrounding polymer matrix.6,7 These nanocomposite structures exhibit properties that are often different from those of the same metal nanoparticles in solution. As an example, thin polymer films with high concentrations of embedded metal nanoparticles synthesized from ion-doped polymer precursors exhibit unique physical characteristics that vary with the interparticle distance between the embedded nanoparticles.8–11 In a previous study, we reported the synthesis of nanocomposite films with embedded metal nanoparticles by the in situ reduction of initially-doped metallic ions in polymer matrices, as well as the control of the above structural variables.12,13 In this in situ approach, molecular hydrogen reduces doped metallic ions to form metal nanoparticles via the diffusion of metal atoms in polymer matrices. The uniform and slow growth rate of the metal nanoparticles in the solid state by reaction with hydrogen molecules, as opposed to conventional nanoparticle growth in solution using chemical reducing agents, results in both homogeneous dispersion and monodispersity of the resulting metal nanoparticles. In addition, the interparticle distance between embedded nanoparticles can be independently controlled by decomposition of the polymer matrix through hydrogenation of the polymer molecules, a process that is catalyzed by the metal nanoparticles themselves.14 These metal/polymer nanocomposites with controlled microstructures allow the study of the fundamental properties of metal nanoparticle structures, especially the analysis of the effects of the size and interparticle distance of the embedded metal nanoparticles. Despite numerous studies into the synthesis of monometallic nanoparticles in polymer matrices, the controlled synthesis of bimetallic (alloy) nanoparticles in these nanocomposite systems has not been reported. The development of effective methodology for the synthesis of alloy nanoparticles with tunable compositions in nanocomposite systems is of importance, both to increase our fundamental understanding of the physicochemical properties of the nanoparticles and to allow the fabrication of nanocomposite devices with desired properties.
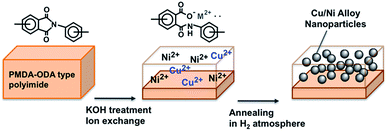
Herein, we report the first example of the synthesis and systematic composition control of alloy nanoparticles in thin polymer layers, using copper and nickel as doping ions. In this work, hydrolyzed polyimide films containing these ions, doped via a simultaneous ion exchange reaction, were subjected to heat treatment under a hydrogen atmosphere, resulting in the formation of monodispersed copper–nickel alloy nanoparticles (Scheme 1). These nanoparticles have various potential applications, such as in catalysis and in the production of magnetic materials, and exhibit properties that are dependent on their chemical composition.15–19 To the best of our knowledge, this is the first example of the synthesis of Cu/Ni alloy nanoparticles in polyimide films that show both suitable thermal stability and dielectric properties, based on an in situ technique using ion-doped precursors.
To synthesize the ion-codoped precursor films, hydrolyzed polyimide films (thickness of hydrolyzed layer: 5 μm) were reacted with an aqueous solution of both copper chloride and nickel chloride at room temperature (see ESI† for details). Although quantitative analysis of the product by inductively coupled plasma (ICP) spectroscopy revealed that the concentrations of doping ions varied with the initial concentrations in the aqueous reaction solution (Fig. 1), the doping ion concentrations in the polymer were not strictly proportional to the initial concentration in solution (but the total amount is constant: 1600 nmol cm−2). For example, films immersed in a solution containing 80 mol% Ni ions contains 20 mol% Ni ions amongst the total quantity of doping ions in the film. This may be attributed to the higher stability of copper ions in the presence of the carboxylate anions in the hydrolyzed polyimide layers compared to nickel ions, as observed in ion-exchange membrane.20 Therefore, it was possible to systematically control the relative amounts of doping ions in the precursor films by careful adjustment of the initial concentrations of ions in the aqueous solution.
The resulting metal nanoparticle/polyimide composites, each differing in the concentration of doping ions, were subsequently subjected to heat treatment at 400 °C (the film was stable up to 400 °C, Fig. S1†) for 30 min under a hydrogen atmosphere (20/80 H2/N2 v/v) and then analyzed by transmission electron microscopy (TEM). The largest nanoparticles (17.8 nm) were obtained from the precursor films containing only Cu ions (Fig. 2A), and were much larger than the pure Ni nanoparticles (8.9 nm, Fig. 2E). In the case of those samples with 70% Cu ions, the nanoparticles showed a bimodal distribution, with diameters of 6.5 and 16.5 nm (Fig. 2B). The nanoparticle size was found to slightly decrease as the mole fraction of Ni ions was increased above 50% (Fig. 2C and D).
X-ray diffraction (XRD) analyses of all samples indicated the formation of a cubic fcc phase (Fig. 3A). A sharp reflection peak attributed to monometallic Cu was observed in the case of the sample with only Cu ions, as expected from the larger nanoparticle size. Phase segregation into Cu and Ni phases was determined from the relatively intense peak at 43.2° and the weaker peak at 44.6° (indicated by arrows in Fig. 3A) generated by the sample with 70% Cu, since these peaks can be assigned to monometallic Cu and Ni phases, respectively. The samples with more than 50% Ni produced peaks partway between those expected from bulk Cu and Ni phases in their XRD patterns. The positions of the peaks associated with the (111) and (200) planes shifted to higher angles as the fraction of Ni was increased above 50%, indicative of the formation of alloy nanoparticles with differing compositions. Interestingly, the changes in the lattice parameters of the alloy nanoparticles did not obey the classical Vegard's law; the lattice parameters were slightly larger than those predicted by Vegard's equation (Fig. 3B).
We suggest that there are two effects that likely contribute to the observed microstructural behavior, based on the TEM and XRD results. First, according to the literature, reduced Cu metal atoms have weaker interactions with polyimide molecules than is the case with Ni, leading to greater diffusion lengths of approximately 100 nm for Cu atoms at 400 °C (compared to only 20 nm for Ni atoms).21 This effect causes Cu nanoparticles to be larger than Ni nanoparticles (Fig. 2). Second, although Cu and Ni ions in the polyimide are reduced at 200–270 °C and 280–360 °C, respectively, both ions are reduced within the same temperature range (200–320 °C) applied to the films (Fig. S2†). Therefore, it is considered that, in the samples with both Cu and Ni ions, Cu nuclei are formed at an earlier stage of the reduction process, following which Ni ions are reduced through catalytic reactions with the reduced Cu and the resulting Ni atoms are thus trapped in the Cu nuclei. This is in agreement with previous reports regarding the Cu-assisted reduction of Ni ions by hydrogen gas in the incipient wetness impregnation method.16 Therefore, the formation of the alloy nanoparticles can be kinetically controlled through the reduction of ions and the diffusion of metal atoms and/or clusters (representing diffusion-controlled aggregative growth). The samples with higher concentrations of Cu ions are able to generate large numbers of smaller Cu nuclei, while the quantity of Ni ions being reduced is minimal. Conversely, larger Cu nuclei tend to form in the samples with lower amounts of Cu ions and higher concentrations of Ni ions. This effect increases the sizes of the alloy nanoparticles as the concentration of Ni ions is increased in the precursor layer. Thus, the nanoparticle size appears to be determined by a delicate balance between the above two effects. Namely, smaller Cu nuclei (formed in Cu-rich samples) grow to form larger nanoparticles with the addition of large amounts of Cu (associated with greater diffusion lengths) and smaller amounts of Ni (associated with smaller diffusion lengths), while larger Cu nuclei (formed in Ni-rich samples) react with smaller amounts of Cu but larger amounts of Ni. We note that, in the case of the samples with the highest concentration of Cu (70%) in the co-doped film, relatively large, isolated Cu nanoparticles were initially formed. Cu atoms were presumably trapped in these larger nanoparticles and therefore may not have contributed to the alloy formation because the large nanoparticles would not have been as mobile in the glassy polyimide matrix (the Tg of polyimide is above 400 °C). This effect would lead to segregation and the formation of a monometallic phase with large Cu and small Ni nanoparticles, giving a bimodal size distribution (Fig. 2B).
The slightly larger lattice parameter observed for alloy nanoparticles was presumably caused by the formation of the nanoparticles in the solid polymer matrix, due to surface passivation of the alloy nanoparticles by polyimide molecules or by the inclusion of polymer molecules in the nanoparticles. Surface passivation and/or inclusion of polyimide molecules will generate tensile stress and/or elastic strain (or structural defects) in the nanoparticles, respectively, leading to increased lattice parameters, although further experimental study is necessary to confirm this consideration.
To investigate the effect of the composition of alloy nanoparticles on the magnetic properties of the film, we employed ferromagnetic resonance (FMR) spectroscopy (Fig. 4). Although no FMR signal was evident in samples having alloy nanoparticles with a 50% Ni content, signals appeared at an external magnetic field of 3000–3500 Oe and shifted to higher magnetic fields as the Ni fraction in the alloy nanoparticles increased (indicated by arrows in the inset to Fig. 4). Since the particle sizes in this composition range were similar (8.9 to 9.3 nm, Fig. 2), the shift can be attributed to increasingly strong magnetic moments in the alloy nanoparticles with increasing ferromagnetic Ni content.22 Mary et al. reported that Cu–Ni alloy nanoparticles with higher Ni concentrations (above 60%) prepared by microwave combustion and 20 to 35 nm in size exhibit ferromagnetic properties, whereas nanoparticles with lower Ni content (less than 50%) are superparamagnetic.17 Our own results are in agreement with this report. The FMR measurements clearly show that control over the composition, and therefore the magnetic characteristics, of Cu–Ni alloy nanoparticles (when using higher Ni proportions) can be achieved via the present solid state, in situ synthetic method. Additionally, the interparticle distances between embedded alloy nanoparticles can be controlled through the catalytic decomposition process (Fig. S3†), by which the polyimide loses mass through a decomposition reaction catalyzed by the alloy nanoparticles.14 The resulting nanocomposite layers, having different interparticle distances but the same nanoparticle sizes, exhibit an angular dependence of the resonant field, such that the layers change from non-interacting to interacting magnetic composite films. This effect suggests further possibilities for controlling the magnetic behavior by varying the magnetic interactions between adjacent nanoparticles.
Conclusions
In conclusion, we have investigated the effects of the proportions of metallic ions doped into surface-modified polyimide on the in situ formation of monometallic and alloy nanoparticles. The phase of the binary system was found to depend on the initial proportions of the added metallic ions, and the composition of the resulting alloy nanoparticles could be systematically controlled when employing higher concentrations of Ni ions, thus varying the ferromagnetic behavior of the nanocomposite layers. This in situ approach provides the ability to systematically tune aspects of the nanocomposite microstructures, including composition, size and interparticle distance. Extending the methodology reported herein to the many other types of alloy nanoparticles is predicted to have an impact in various areas of nanoscience, including catalysis and controlled optical and magnetic interactions, and to enhance our fundamental understanding of nanoscale phase changes in alloy systems.Notes and references
- S. Ehlert, C. Stegelmeier, D. Pirner and S. Föster, Macromolecules, 2015, 15, 5323–5327 CrossRef.
- N. P. B. Tan, C. H. Lee, L. Chen, K. M. Ho, Y. Lu, M. Ballauff and P. Li, Polymer, 2015, 76, 271–279 CrossRef CAS.
- M. Signoretto, I. Suárez, V. S. Chirvony, R. Abargues, P. J. Rodrigues-Cantó and J. Martinez-Pastor, Nanotechnology, 2015, 26, 475201 CrossRef CAS PubMed.
- J. Song, H. Kang, C. Lee, S. H. Hwang and J. Jang, ACS Appl. Mater. Interfaces, 2012, 1, 460–465 Search PubMed.
- K. Y. Chun, Y. Oh, J. Rho, J. H. Ahn, Y. J. Kim, H. R. Choiand and S. Baik, Nat. Nanotechnol., 2010, 5, 853–857 CrossRef CAS PubMed.
- G. V. Ramesh, S. Porel and T. P. Radhakrishman, Chem. Soc. Rev., 2009, 38, 2646–2656 RSC.
- D. H. Cole, K. R. Shull, P. Baldo and L. Rehn, Macromolecules, 1999, 32, 771–779 CrossRef CAS.
- V. K. Rao and T. P. Radhakrishnan, ACS Appl. Mater. Interfaces, 2015, 7, 12767–12773 CAS.
- G. M. Neelgund, E. Hrehorova, M. Joyce and V. Bliznyuk, Polym. Int., 2008, 57, 1083–1089 CrossRef CAS.
- L. Balan, M. Jin, J. P. Malval, H. Chaumeil, A. Defoin and L. Vidal, Macromolecules, 2008, 23, 9359–9365 CrossRef.
- M. Sakamoto, T. Tachikawa, M. Fujitsuka and T. Majima, Adv. Funct. Mater., 2007, 17, 857–862 CrossRef CAS.
- I. Toda, T. Tsuruoka, J. Matsui, T. Murashima, H. Nawafune and K. Akamatsu, RSC Adv., 2014, 4, 4723–4726 RSC.
- K. Akamatsu, S. Adachi, T. Tsuruoka, S. Ikeda, S. Tomita and H. Nawafune, Chem. Mater., 2008, 20, 3042–3047 CrossRef CAS.
- K. Akamatsu, M. Fujii, T. Tsuruoka, S. Nakano, T. Murashima and H. Nawafune, J. Phys. Chem. C, 2012, 116, 17947–17954 CAS.
- G. Guisbiers, S. Khanal, F. R. Zepeda, J. R. Puente and M. J. Yacaman, Nanoscale, 2014, 6, 14630–14635 RSC.
- Q. Wu, L. D. L. Duchstein, G. L. Chiarello, J. M. Christensen, C. D. Damsgaard, C. F. Elkjar, J. B. Wagner, B. Temel, J. D. Grunwaldt and A. D. Jensen, ChemCatChem, 2014, 6, 301–310 CrossRef CAS.
- J. A. Mary, A. Manikandan, L. J. Kennedy, M. Bououdina, R. Sundaram and J. J. Vijaya, Trans. Nonferrous Met. Soc. China, 2014, 24, 1467–1473 CrossRef CAS.
- S. Pramanik, S. Pal, S. Bysakh and G. De, J. Nanopart. Res., 2011, 13, 321–329 CrossRef CAS.
- C. Damle and M. Sastry, J. Mater. Chem., 2002, 12, 1860–1864 RSC.
- G. Suresh, Y. M. Scindia, A. K. Pandey and A. Goswami, J. Phys. Chem. B, 2004, 108, 4104–4110 CrossRef CAS.
- F. Faupel, R. Willecke and A. Thran, Mater. Sci. Eng., R, 1998, 22, 1–55 CrossRef.
- S. Tomita, P. E. Jonsson, K. Akamatsu, H. Nawafune and H. Takayama, Phys. Rev. B: Condens. Matter Mater. Phys., 2007, 76, 174432 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental procedure; amount of unreduced metal ions as a function of temperature; FMR signal of obtained samples. See DOI: 10.1039/c6ra00240d |
| This journal is © The Royal Society of Chemistry 2016 |