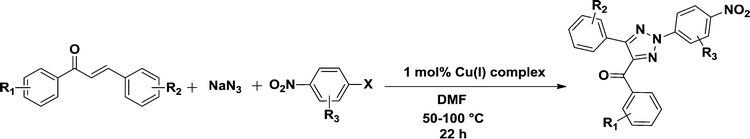
A binuclear Cu(I) complex as a novel catalyst towards the direct synthesis of N-2-aryl-substituted-1,2,3-triazoles from chalcones†
Divya Pratap Singh,
Bharat Kumar Allam,
Rahul Singh,
Krishna Nand Singh and
Vinod Prasad Singh*
Department of Chemistry, Institute of Science, Banaras Hindu University, Varanasi 221005, India. E-mail: singvp@yahoo.co.in; Tel: +91 9450145060
First published on 29th January 2016
Abstract
A binuclear Cu(I) complex containing a N′,N′-bis{(1H-indol-3-yl)methylene}oxalohydrazide (H2bioh) ligand has been synthesized and characterized. The molecular structures of the synthesized compounds have been determined by single crystal X-ray diffraction. The crystal structures are stabilized by inter- and intra-molecular π–π stacking and C–H⋯π interactions. The Cu(I)-complex has successfully been employed as an efficient catalyst for one-pot operation involving the azide–chalcone click reaction and subsequent arylation has been developed. The complex exhibits excellent catalytic activity with significantly low catalyst loading. The overall process is insensitive to air and moisture and can be manipulated under ambient temperature with operational ease.
1. Introduction
Click chemistry is the most straightforward and atom-economical approach for the synthesis of 1,2,3-triazoles.1 Owing to its fidelity and compatibility, this strategy has been widely used in chemical biology, materials science, and medicinal chemistry.2 Most of the reported click reactions are two-component, employing organic azides and alkynes. In this system, the organic azides need to be synthesized in advance, and the potential hazards of organic azides, especially in isolation or purification processing, can be problematic. Thus, it is desirable to develop efficient methods that use sodium azide for direct cycloaddition.The synthesis and functionalization of triazole derivatives is an attractive area of research. Recently described methods for the catalytic direct functionalization of triazole and its derivatives have also been found attractive but are mainly limited to the N-1 position.3 One important group of triazole derivatives is the N-2-aryl/alkyl triazoles. In N-2-aryl triazoles the two aromatic rings adopt coplanar conformation and thus posses a typical electron density distribution, and exhibit unique photonic properties.4 It has been well documented that the N-2 substitution is a major challenge for triazoles due to the lower relative electron density at the internal nitrogen atom (N-2) as compared to the two terminal nitrogen atoms (N-1 and N-3). The efforts have been made by Sharpless and others to further advance this difficult N-2 functionalization of triazoles.5 Despite of these achievements, a facile preparation of N-2-substituted triazoles with low copper loading under mild conditions remains challenging.
The Cu(I) species necessary for the click reaction is often generated in situ either by employing Cu(II) in conjunction with a reducing agent such as ascorbate,6 or via the comproportionation of Cu(II) and Cu(0). Additionally, 1,2,3-triazoles and other compounds have been employed as stabilizing ligands for Cu(I), inhibiting the oxidation and disproportionation of the ion while retaining its catalytic activity.7 Various genuine Cu(I) catalysts that do not require a sacrificial reductant are also known. The advantage of ligated Cu(I), particularly with nitrogen ligands, being the rate accelerator, for instance with the efficient polytriazoles8 and tris(2-aminoethyl)amine derivatives.9 The nitrogen ligands allow the use of Cu(I) catalysts in reduced amounts (most often of the order of 1%) compared to the original, simple, and practical catalyst CuSO4 + sodium ascorbate that is still the most commonly utilized catalyst but in much larger quantities.10 Although Cu(I) may be obtained directly from the utilization of Cu(I) salts and coordination complexes,11 the thermodynamically unstable Cu(I) either disproportionates to Cu(II) and Cu(0) or oxidizes to Cu(II). Moreover, when Cu(I) halides are employed as azide–alkyne cycloaddition catalysts in organic solvents, significant amounts of undesired side-products are generated.12 Owing to the cytotoxicity associated with Cu(I), several groups have endeavored to minimize the copper concentration while maintaining high reaction rates.13
Heterocyclic Schiff bases contain a number of coordination sites in addition to ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–, forming more stable bi-, tri- or poly-nuclear metal complexes. It has also been observed that the binuclear complexes of transition metal ions show better catalytic efficiency than those of corresponding mononuclear ones due to the synergic effect of two metal ions.14 In view of the potential catalytic applications offered by the binuclear metal complexes of acyldihydrazone Schiff bases, and as a part of our recent investigations on the catalytic application of transition metals15 and their complexes;16 we have synthesized and characterized a novel binuclear copper(I) complex with H2bioh ligand. The catalytic efficiency of the above complex has successfully been explored for N-2-aryl-1,2,3-triazole synthesis.
N–, forming more stable bi-, tri- or poly-nuclear metal complexes. It has also been observed that the binuclear complexes of transition metal ions show better catalytic efficiency than those of corresponding mononuclear ones due to the synergic effect of two metal ions.14 In view of the potential catalytic applications offered by the binuclear metal complexes of acyldihydrazone Schiff bases, and as a part of our recent investigations on the catalytic application of transition metals15 and their complexes;16 we have synthesized and characterized a novel binuclear copper(I) complex with H2bioh ligand. The catalytic efficiency of the above complex has successfully been explored for N-2-aryl-1,2,3-triazole synthesis.
2. Experimental
2.1 Synthesis of ligand
The ligand, N′,N′-bis{(1H-indol-3-yl)methylene}oxalo hydrazide (H2bioh) was synthesized by reacting 50 mL aqueous solution of oxalic acid dihydrazide (10 mmol, 1.18 g) with 50 mL methanolic solution of indole-3-carboxaldehyde (20 mmol, 2.90 g) in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio in a round bottom flask. A light yellow colored product was obtained by stirring the reaction mixture for 5 h at room temperature. The product was filtered on a Buckner funnel and washed several times with water followed by methanol, and dried in a desiccator over anhydrous calcium chloride at room temperature. Single crystal of the ligand was obtained by slow evaporation of its solution in a mixture of dichloromethane and DMSO solvents at room temperature. Yield (85%). Mp > 300 °C. Anal. calc. for C20H16N6O2 (372): C, 64.51; H, 4.33; N, 22.57. Found: C, 64.35; H, 4.35; N, 22.45%. IR (ν cm−1, KBr): ν(NH) 3263b; ν(C
2 molar ratio in a round bottom flask. A light yellow colored product was obtained by stirring the reaction mixture for 5 h at room temperature. The product was filtered on a Buckner funnel and washed several times with water followed by methanol, and dried in a desiccator over anhydrous calcium chloride at room temperature. Single crystal of the ligand was obtained by slow evaporation of its solution in a mixture of dichloromethane and DMSO solvents at room temperature. Yield (85%). Mp > 300 °C. Anal. calc. for C20H16N6O2 (372): C, 64.51; H, 4.33; N, 22.57. Found: C, 64.35; H, 4.35; N, 22.45%. IR (ν cm−1, KBr): ν(NH) 3263b; ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) 1667s; ν(C
O) 1667s; ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) 1578m; ν(N–N) 989w. 1H NMR (DMSO-d6; δ ppm): 11.90 (s, NH); 11.65 (s, NH, indole moiety); 8.73 (s, CH); 8.25–7.13 (Ar-H). 13C NMR (DMSO-d6; δ ppm): 166.62 (
N) 1578m; ν(N–N) 989w. 1H NMR (DMSO-d6; δ ppm): 11.90 (s, NH); 11.65 (s, NH, indole moiety); 8.73 (s, CH); 8.25–7.13 (Ar-H). 13C NMR (DMSO-d6; δ ppm): 166.62 (![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); 158.33 (
O); 158.33 (![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N); 156.12–111.57 (aromatic carbons).
N); 156.12–111.57 (aromatic carbons).
2.2 Synthesis of the Cu(I) complex
The Cu(I) complex of N′,N′-bis{(1H-indol-3-yl)methylene} oxalohydrazide (H2bioh) was synthesized by reacting 50 mL methanolic solution of the ligand H2bioh (1 mmol, 0.334 g) with 50 mL solution of bis-(triphenylphosphine)copper(I) nitrate (2 mmol, 1.3 g) in dichloromethane in 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (M
1 (M![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) L) molar ratio. The reaction mixture was continuously stirred on a magnetic stirrer for 24 h at room temperature. The product was crystallized from the above solution by evaporating the solvent slowly at room temperature. Yellow crystal, yield (70%). Mp 185 °C. Anal. calc. for C94H84Cu2N8O10P4 (1736.67): Cu, 7.31; C, 64.95; H, 4.84; N, 6.45. Found: Cu, 7.28; C, 64.76; H, 4.85; N, 6.41%. IR (ν cm−1, KBr): ν(NH) 3258b; ν(C
L) molar ratio. The reaction mixture was continuously stirred on a magnetic stirrer for 24 h at room temperature. The product was crystallized from the above solution by evaporating the solvent slowly at room temperature. Yellow crystal, yield (70%). Mp 185 °C. Anal. calc. for C94H84Cu2N8O10P4 (1736.67): Cu, 7.31; C, 64.95; H, 4.84; N, 6.45. Found: Cu, 7.28; C, 64.76; H, 4.85; N, 6.41%. IR (ν cm−1, KBr): ν(NH) 3258b; ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) 1634m; ν(C
O) 1634m; ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) 1553m; ν(N–N) 1020w. 1H NMR (DMSO-d6; δ ppm): 11.95 (s, NH); 11.67 (s, NH, indole moiety); 8.76 (s, CH); 8.24–7.14 (m, Ar-H). 13C NMR (DMSO-d6; δ ppm): 155.97 (C
N) 1553m; ν(N–N) 1020w. 1H NMR (DMSO-d6; δ ppm): 11.95 (s, NH); 11.67 (s, NH, indole moiety); 8.76 (s, CH); 8.24–7.14 (m, Ar-H). 13C NMR (DMSO-d6; δ ppm): 155.97 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); 147.90 (C
O); 147.90 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N); 137.05–111.34 (aromatic carbons); 31P NMR (DMSO-d6; δ ppm): 10.71.
N); 137.05–111.34 (aromatic carbons); 31P NMR (DMSO-d6; δ ppm): 10.71.
2.3 General procedure for the synthesis of N-2-aryl-substituted-1,2,3-triazoles
The chalcones (1 mmol), sodium azide (1 mmol), Cu(I) complex (1 mol%) and 3 mL DMF were taken in a 25 mL round bottom flask equipped with a stirrer. The reaction mixture was stirred at 50 °C for 10 h, and then 2,4-dinitrochlorobenzene/4-fluoronitro benzene (1 mmol) was added to the mixture. The reaction was continued further for 12 h at 100 °C. After completion of the reaction, brine water (10 mL) was added and extracted with ethyl acetate (3 × 10 mL). The combined organic phases were dried over anhydrous Na2SO4 and concentrated under reduced pressure in a rotary evaporator. The crude product thus obtained, was purified using column chromatography (ethylacetate/hexane). All the products have been characterized based upon reported literature characteristics.3. Results and discussion
It appears from the analytical data that reaction between the precursor bis-(triphenylphosphine)copper(I) nitrate and ligand H2bioh occurs in 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (M
1 (M![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) L) molar ratio to give a binuclear Cu(I) complex (Scheme 1).
L) molar ratio to give a binuclear Cu(I) complex (Scheme 1).
The complex is highly soluble in water, dichloromethane, ethanol, methanol, chloroform, chloroform, DMF and DMSO. It melts with decomposition at 185 °C and shows molar conductance value of 59.35 Ω−1 mol−1 cm2 at room temperature in the range of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 electrolyte for 10−3 M solution in DMSO.
1 electrolyte for 10−3 M solution in DMSO.
3.1 Spectral characterization
The IR spectral bands observed at 3263, 1667, 1578 and 989 cm−1 in the free ligand are assigned to ν(N–H), ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), ν(C
O), ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) and ν(N–N), respectively.16 In the complex, ν(C
N) and ν(N–N), respectively.16 In the complex, ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) and ν(C
O) and ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) occur at significantly lower wave numbers than the free ligand, indicating coordination of the
N) occur at significantly lower wave numbers than the free ligand, indicating coordination of the ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and
O and ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N groups to metal.17 A considerable shift of ν(N–N) to higher wave number in the complex indicates the involvement of one of the nitrogen atoms of
N groups to metal.17 A considerable shift of ν(N–N) to higher wave number in the complex indicates the involvement of one of the nitrogen atoms of ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) N–N
N–N![[double bond splayed right]](https://www.rsc.org/images/entities/char_e00a.gif) moiety in bonding.18 The presence of ν(N–H) nearly at the same position in the complex as in the ligand shows non-participation of N–H group in bonding.
moiety in bonding.18 The presence of ν(N–H) nearly at the same position in the complex as in the ligand shows non-participation of N–H group in bonding.
The 1H NMR spectrum of H2bioh in DMSO exhibits signals at 11.90 and 11.65 ppm due to ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) NH protons of –CONH and indole–NH, respectively (Fig. S1†). In the Cu(I) complex, these protons occur almost at the same positions as in the ligand, suggesting non-participation of
NH protons of –CONH and indole–NH, respectively (Fig. S1†). In the Cu(I) complex, these protons occur almost at the same positions as in the ligand, suggesting non-participation of ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) NH group in bonding (Fig. S2†). The –CH
NH group in bonding (Fig. S2†). The –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N proton observed at 8.73 ppm in the ligand, shows downfield shift in the complex due to participation of –CH
N proton observed at 8.73 ppm in the ligand, shows downfield shift in the complex due to participation of –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– group in bonding. The signals due to aromatic ring protons are observed in the region 8.24–7.14 ppm as multiplets in the ligand and complex.19 The 13C NMR spectrum exhibits a cluster of peaks between 156.13–111.57 ppm in the ligand and 137.05–111.34 ppm in the complex corresponding to carbons of the aromatic rings (Fig. S3†). The signals observed at 158.33 and 166.62 ppm in the ligand are assigned to
N– group in bonding. The signals due to aromatic ring protons are observed in the region 8.24–7.14 ppm as multiplets in the ligand and complex.19 The 13C NMR spectrum exhibits a cluster of peaks between 156.13–111.57 ppm in the ligand and 137.05–111.34 ppm in the complex corresponding to carbons of the aromatic rings (Fig. S3†). The signals observed at 158.33 and 166.62 ppm in the ligand are assigned to ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– and
N– and ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O carbons, respectively. These signals show a considerable high field shift in the Cu(I) complex, suggesting the bonding of
O carbons, respectively. These signals show a considerable high field shift in the Cu(I) complex, suggesting the bonding of ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– and
N– and ![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups with metal (Fig. S4†). The 31P NMR spectrum of the complex shows a peak at 10.71 ppm due to PPh3.
O groups with metal (Fig. S4†). The 31P NMR spectrum of the complex shows a peak at 10.71 ppm due to PPh3.
3.2 Crystal structure of H2bioh ligand
The crystallographic data, structural refinement details, selected bond lengths, bond angles and hydrogen bonding parameters of H2bioh and its Cu(I) complex are given in Tables S1–S3,† respectively. Fig. 1 shows ORTEP diagram of the ligand with atomic numbering scheme. The molecule displays an E configuration about the![[double bond splayed left]](https://www.rsc.org/images/entities/char_e009.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond.16 The C(10)–O(1) and C(9)–N(2) display bond distances of 1.236 (2) and 1.285 (3) Å, respectively as reported for double bonds.16 The N(2)–N(3) bond distance is 1.390 Å, which is slightly shorter than the single bond distance (1.411 Å), indicating some double bond character.20 All the C–C bond length (1.390 Å) and C–C–C bond angle (116.62°) of the indole ring as well as the C–N bond length (1.361 Å) and C–N–C angle (109.48°) are in accordance with a typical indole moiety.21
N bond.16 The C(10)–O(1) and C(9)–N(2) display bond distances of 1.236 (2) and 1.285 (3) Å, respectively as reported for double bonds.16 The N(2)–N(3) bond distance is 1.390 Å, which is slightly shorter than the single bond distance (1.411 Å), indicating some double bond character.20 All the C–C bond length (1.390 Å) and C–C–C bond angle (116.62°) of the indole ring as well as the C–N bond length (1.361 Å) and C–N–C angle (109.48°) are in accordance with a typical indole moiety.21
The molecular structure of the ligand is stabilized by inter-molecular H-bonding as well as inter-molecular C–H⋯π interactions. The inter-molecular H-bonding interactions N(1)–H(1A)⋯O(1) (2.10 Å), N(3)–H(3A)⋯O(1) (2.09 Å) and N(3)–H(3A)⋯O(1) (2.34 Å) form supra-molecular architectures on axis ‘a’ and ‘c’ (Fig. S5†). The inter-molecular C–H⋯π interactions occur between the centroid of indole/phenyl ring and phenyl hydrogen with the contact distances of 2.974 and 3.398 Å (Fig. 2).16
 | ||
| Fig. 2 Diagram showing intermolecular hydrogen bonding and intra-molecular C–H⋯π interactions along ‘c’ axis in H2bioh. | ||
3.3 Crystal structure of Cu(I) complex
Fig. 3 shows ORTEP diagram of the Cu(I) complex with atomic numbering scheme. In the binuclear complex, each metal centre is coordinated with NOP2 core by using azomethine–nitrogen and a carbonyl–oxygen of H2bioh ligand, and two phosphorous atoms of triphenylphosphine ligands. The two metal atoms in the complex are arranged in a trans manner to the coordinating sites of ligands. The average bond lengths in the complex are: C(9)–N(2) = 1.294(3), C(10)–O(1) = 1.228 (3) and N(2)–N(3) = 1.398(3) Å, respectively, which are longer or shorter than those of the corresponding distances in the ligand. The Cu–O(1) and Cu–N(2) bond distances are 2.197 (18) Å and 2.076 (2) Å, which fall in the range reported for Cu(I) tetrahedral complexes.16 The C(9)–N(2) distance (1.294 Å) in the complex is significantly longer than the free ligand (1.285 Å) due to coordination of azomethine–N to the metal ion. | ||
| Fig. 3 ORTEP diagram of [Cu2(H2bioh)(PPh3)4](NO3)2·2CH3OH showing atomic numbering scheme with ellipsoids of 30% probability (phenyl rings, methanol and nitrate ions are omitted for clarity). | ||
The molecular structure of Cu(I)-complex is stabilized by a number of inter-molecular hydrogen bonding and intra-molecular C–H⋯π interactions (Fig. 4). The inter-molecular hydrogen bonding interactions form a supra-molecular architecture on axis ‘c’ (Fig. S6†). The intra-molecular C–H⋯π interactions occur between a phenyl proton and the centroid of the phenyl ring with contact distances of 3.168, 3.289 and 3.386 Å. The C–H⋯π interactions also take place when phenyl protons approach the centroid of the chelate rings with contact distances of 2.968 and 3.183 Å (Fig. 4).16
3.4 Catalytic properties
The synthesized Cu(I)-complex has successfully been applied as a catalyst for the synthesis of N-2-aryl-1,2,3-triazoles through azide–chalcone oxidative cycloaddition and post-triazole arylation. The initial experiment was performed by using a model reaction between chalcone and sodium azide using 0.5 mol% of Cu(I)-complex under air for 10 h first, then 2,4-(NO2)2-C6H3Cl was added to the mixture. The reaction was continued further for 12 h to deliver the desired product 4a with 67% yield (Table 1, entry 1). Further optimization of the reaction conditions resulted 84% product yield when 1 mol% Cu(I) complex was used as a catalyst (entry 2). Various solvents were also probed for their effect on the reaction yield and found DMF as the most suitable solvent (entries 4 and 5). The Cu(I)-precursor was also tested for its efficiency and found lower yield (entry 6). The ligand H2bioh was failed to promote the reaction (entry 7).| Entry | Catalyst mol (%) | Solvent | Yieldb (%) |
|---|---|---|---|
| a Using chalcone (1 mmol), sodium azide (1 mmol, 65 mg), DMF (4 mL), Cu(I) complex (1 mol%), DMF (4 mL), at 50 °C under air for 10 h first, then 2,4-(NO2)2-C6H3Cl/4-NO2C6H4F (1 mmol) and DBU (1 equiv.) was added to the mixture, and the reaction continued further for 12 h at 100 °C.b Isolated yields. | |||
| 1 | Cu(I) complex (0.5) | DMF | 67 |
| 2 | Cu(I) complex (1) | DMF | 84 |
| 3 | Cu(I) complex (2) | DMF | 84 |
| 4 | Cu(I) complex (1) | CH3CN | 69 |
| 5 | Cu(I) complex (1) | H2O | n.r |
| 6 | [Cu(PPh3)2]NO3 (5) | DMF | 45 |
| 7 | H2bioh (5) | DMF | n.r |
To test the versatility of the optimized reaction conditions, the reactions of a variety of chalcones were studied with sodium azide and 2,4-(NO2)2-C6H3Cl/4-NO2C6H4F (Table 2). Chalcones having various substituents viz. (E)-3-(phenyl)-1-phenylprop-2-en-1-one (3a), (E)-3-(4-chlorophenyl)-1-phenylprop-2-en-1-one (3b), (E)-3-(4-chlorophenyl)-1-(4-methoxyphenyl)prop-2-en-1-one (3c), (E)-1-phenyl-3-(4-methoxy)prop-2-en-1-one (3d), (E)-1-(4-methoxyphenyl)-3-(4-nitrophenyl)prop-2-en-1-one (3e) and aryl fluorides/chlorides having electron-withdrawing groups were participated well in these reactions. The presence of an electron-withdrawing group on the aryl halide counterpart is necessary driving force for a successful post arylation. Functional groups appended to chalcones and aryl halides were also well tolerated in these reactions.
| Entry | Aryl halide | Chalcone | Product | Yieldb (%) | |||
|---|---|---|---|---|---|---|---|
| a Reaction conditions: using chalcone (1 mmol), sodium azide (1 mmol, 65 mg), Cu(I) complex (1 mol%), DMF (4 mL), at 50 °C under air for 10 h first, then 2,4-(NO2)2-C6H3Cl/4-NO2C6H4F (1 mmol) and DBU (1 equiv.) was added to the mixture, and the reaction continued further for 12 h at 100 °C.b Isolated yields. | |||||||
| 1 | 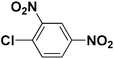 |
1a | 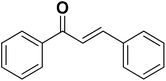 |
3a | 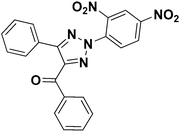 |
4a | 84 |
| 2 |  |
1b |  |
3a | 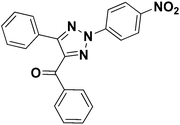 |
4b | 81 |
| 3 | 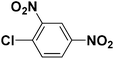 |
1a |  |
3b |  |
4c | 75 |
| 4 |  |
1b | 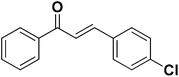 |
3b | 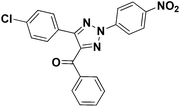 |
4d | 73 |
| 5 |  |
1a |  |
3c |  |
4e | 82 |
| 6 |  |
1b |  |
3c |  |
4f | 79 |
| 7 |  |
1a |  |
3d |  |
4g | 74 |
| 8 |  |
1b |  |
3d |  |
4h | 72 |
| 9 |  |
1a |  |
3e |  |
4i | 82 |
| 10 |  |
1b |  |
3e |  |
4j | 85 |
4. Conclusions
The present work describes the synthesis of a ligand N′,N′-bis{(1H-indol-3-yl)methylene}oxalohydrazide (H2bioh) and its Cu(I) complex and their characterization by various spectroscopic techniques. The molecular structures of the ligand and its Cu(I) complex have been determined by X-ray crystallography. The intermolecular π–π and C–H⋯π interactions are observed in the ligand as well as in its Cu(I) complex. The complex has a binuclear structure with a 4-coordinate tetrahedral geometry around each metal ion. The catalytic activity of the complex was successfully explored for the N-2-aryl-substituted-1,2,3-triazoles synthesis.Acknowledgements
The authors D. P. Singh and B. K. Allam are thankful to CSIR, New Delhi for the award of Senior Research Fellowships (SRF), V. P. Singh is thankful to UGC, New Delhi for financial assistance.Notes and references
- (a) H. C. Kolb, M. G. Finn and K. B. Sharpless, Angew. Chem., Int. Ed., 2001, 40, 2004–2021 CrossRef CAS; (b) C. Barner-Kowollik, F. E. Du Prez, P. Espeel, C. J. Hawker, T. Junkers, H. Schlaad and W. Van Camp, Angew. Chem., Int. Ed., 2011, 5, 60–62 CrossRef PubMed.
- (a) M. C. Kozlowski, Reactive Intermediates in Chemistry, Biology. Copper–Oxygen Chemistry, ed. E. R. Steven, D. K. Kenneth and I. Shinobu, Wiley, 2011 Search PubMed; (b) A. E. Wendlandt, A. M. Suess and S. S. Stahl, Angew. Chem., Int. Ed., 2011, 50, 11062–11087 CrossRef CAS PubMed; (c) Y.-C. Wang, Y.-Y. Xie, H.-E. Qu, H.-S. Wang, Y.-M. Pan and F.-P. Huang, J. Org. Chem., 2014, 79, 4463–4469 CrossRef CAS PubMed; (d) D. Sahu, S. Dey, T. Pathak and B. Ganguly, Org. Lett., 2014, 16, 2100–2103 CrossRef CAS PubMed; (e) S. Ding, G. Jia and J. Sun, Angew. Chem., Int. Ed., 2014, 53, 1877–1880 CrossRef CAS PubMed; (f) L. Hong, W. Lin, F. Zhang, R. Liu and X. Zhou, Chem. Commun., 2013, 49, 5589–5591 RSC; (g) W. Xi, T. F. Scott, C. J. Kloxin and C. N. Bowman, Adv. Funct. Mater., 2014, 24, 2572–2590 CrossRef CAS.
- (a) C. W. Tornøe, C. Christensen and M. J. Meldal, J. Org. Chem., 2002, 67, 3057–3064 CrossRef; (b) V. Aucagne and D. A. Leigh, Org. Lett., 2006, 8, 4505–4507 CrossRef CAS PubMed; (c) M. M. Majireck and S. J. Weinreb, J. Org. Chem., 2006, 71, 8680–8683 CrossRef CAS PubMed; (d) D. Yang, N. Fu, Z. Liu, Y. Li and B. Chen, Synlett, 2007, 278–282 CAS; (e) B. Sreedhar, P. S. Reddy and V. R. Krishna, Tetrahedron Lett., 2007, 48, 5831–5834 CrossRef CAS; (f) F. Shi, J. P. Waldo, Y. Chen and R. C. Larock, Org. Lett., 2008, 10, 2409–2412 CrossRef CAS PubMed; (g) W. Qian, D. Winternheimer and J. Allen, Org. Lett., 2011, 13, 1682–1685 CrossRef CAS PubMed.
- (a) Y. Zhang, X. Ye, J. L. Petersen, M. Li and X. Shi, J. Org. Chem., 2015, 80, 3664–3669 CrossRef CAS PubMed; (b) J. Cheon and J. H. Lee, Acc. Chem. Res., 2008, 41, 1630–1640 CrossRef CAS PubMed.
- (a) J. Kalisiak, K. B. Sharpless and V. V. Fokin, Org. Lett., 2008, 10, 3171–3174 CrossRef CAS PubMed; (b) S. Kamijo, T. Jin, Z. Huo and Y. Yamamoto, J. Am. Chem. Soc., 2002, 125, 7786–7787 CrossRef PubMed; (c) Y. Chen, Y. Liu, J. L. Petersen and X. Shi, Chem. Commun., 2008, 3254–3256 RSC; (d) Y. Liu, W. Yan, Y. Chen, J. L. Petersen and X. Shi, Org. Lett., 2008, 10, 5389–5392 CrossRef CAS PubMed; (e) X. Wang, L. Zhang, H. Lee, N. Haddad, D. Krishnamurthy and C. H. Senanayake, Org. Lett., 2009, 11, 5026–5028 CrossRef CAS PubMed; (f) Y. Zhang, X. Li, J. Li, J. Chen, X. Meng, M. Zhao and B. Chen, Org. Lett., 2012, 14, 26–29 CrossRef CAS PubMed.
- V. V. Rostovtsev, L. G. Green, V. V. Fokin and K. B. Sharpless, Angew. Chem., Int. Ed., 2002, 41, 2596–2599 CrossRef CAS.
- Q. Wang, T. R. Chan, R. Hilgraf, V. V. Fokin, K. B. Sharpless and M. G. Finn, J. Am. Chem. Soc., 2003, 125, 3192–3193 CrossRef CAS PubMed.
- (a) T. R. Chan, R. Hilgraf, K. B. Sharpless and V. V. Fokin, Org. Lett., 2004, 6, 2853–2855 CrossRef CAS PubMed; (b) V. Hong, S. I. Presolski, C. Ma and M. G. Finn, Angew. Chem., Int. Ed., 2009, 48, 9879–9883 CrossRef CAS PubMed.
- (a) P. L. Golas, N. V. Tsarevsky, B. S. Sumerlin and K. Matyjaszewski, Macromolecules, 2006, 39, 6451–6457 CrossRef CAS; (b) P. L. Golas, N. V. Tsarevsky and K. Matyjaszewski, Macromol. Rapid Commun., 2008, 29, 1167–1171 CrossRef CAS; (c) L. Liang, J. Ruiz and D. Astruc, Adv. Synth. Catal., 2011, 353, 3434–3450 CrossRef CAS; (d) N. Candelon, D. Lastécouère, A. K. Diallo, J. Ruiz, D. Astruc and J.-M. Vincent, Chem. Commun., 2008, 741–743 RSC.
- (a) C. Ornelas, J. Ruiz, E. Cloutet, S. Alves and D. Astruc, Angew. Chem., Int. Ed., 2007, 46, 872–877 CrossRef CAS PubMed; (b) A. K. Diallo, C. Ornelas, L. Salmon, J. Ruiz and D. Astruc, Angew. Chem., Int. Ed. Engl., 2007, 46, 8644–8648 CrossRef CAS PubMed; (c) D. Astruc, L. Liang, A. Rapakousiou and J. Ruiz, Acc. Chem. Res., 2012, 45, 630–640 CrossRef CAS PubMed.
- (a) J. E. Hein and V. V. Fokin, Chem. Soc. Rev., 2010, 39, 1302–1315 RSC; (b) M. Meldal and C. W. Tornoe, Chem. Rev., 2008, 108, 2952–3015 CrossRef CAS PubMed.
- P. Siemsen, R. C. Livingston and F. Diederich, Angew. Chem., Int. Ed., 2000, 39, 2633–2657 CrossRef.
- (a) Y. M. A. Yamada, S. M. Sarkar and Y. Uozumi, J. Am. Chem. Soc., 2012, 134, 9285–9290 CrossRef CAS PubMed; (b) C. Deraedt, N. Pinaud and D. Astruc, J. Am. Chem. Soc., 2014, 136, 12092–12098 CrossRef CAS PubMed.
- K. C. Gupta and A. K. Sutar, Coord. Chem. Rev., 2008, 252, 1420–1450 CrossRef CAS.
- (a) R. Singh, B. K. Allam, N. Singh, K. Kumari, S. K. Singh and K. N. Singh, Adv. Synth. Catal., 2015, 357, 1181–1186 CrossRef CAS; (b) N. Singh, R. Singh, D. S. Raghuvanshi and K. N. Singh, Org. Lett., 2013, 15, 5874–5877 CrossRef CAS PubMed; (c) D. S. Raghuvanshi, A. K. Gupta and K. N. Singh, Org. Lett., 2012, 14, 4326–4329 CrossRef CAS PubMed; (d) B. K. Allam and K. N. Singh, Synthesis, 2011, 1125–1131 CAS.
- (a) D. P. Singh, B. K. Allam, K. N. Singh and V. P. Singh, J. Mol. Catal. A: Chem., 2015, 398, 158–163 CrossRef CAS; (b) D. P. Singh, B. K. Allam, K. N. Singh and V. P. Singh, RSC Adv., 2014, 4, 1155–1158 RSC; (c) D. P. Singh, D. S. Raghuvanshi, K. N. Singh and V. P. Singh, J. Mol. Catal. A: Chem., 2013, 379, 21–29 CrossRef CAS.
- P. Singh, D. P. Singh and V. P. Singh, Polyhedron, 2014, 81, 56–65 CrossRef CAS.
- V. P. Singh and D. P. Singh, Macromol. Res., 2013, 21, 757–766 CrossRef CAS.
- (a) M. R. Maurya, S. Agrawal, C. Bader and D. Rehder, Eur. J. Inorg. Chem., 2005, 147–157 CrossRef CAS; (b) M. R. Maurya, S. Khurana, C. Schulzke and D. Rehder, Eur. J. Inorg. Chem., 2001, 779–788 CrossRef CAS.
- M. A. Ali, A. H. Mirza, R. J. Butcher, M. T. H. Tarafder and M. A. Ali, Inorg. Chim. Acta, 2001, 320, 1–6 CrossRef.
- K. K. W. Lo, K. H. K. Tsang, W. K. Hui and N. Zhu, Inorg. Chem., 2005, 44, 6100–6110 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental details and spectral data for ligand. CCDC 984536 and 984537 contain the crystallographic data for the ligand H2bioh and Cu(I) complex, respectively. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c5ra27907k |
| This journal is © The Royal Society of Chemistry 2016 |