A polymer/metal core–shell nanofiber membrane by electrospinning with an electric field, and its application for catalyst support
Chenglin Jiang,
Jun Nie and
Guiping Ma*
State Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, North Third Ring Road 15, Chaoyang District, Beijing 100029, PR China. E-mail: magp@mail.buct.edu.cn
First published on 23rd February 2016
Abstract
In the present study, PVP/Ag nanofibers with a core–shell structure have been successfully prepared by using the electrospinning technique under the action of electric field induction. PVP (polyvinyl pyrrolidone), as the functional template during electrospinning, plays an important role both as the reducing agent and as the capping agent. The structure and properties of the thus-obtained nanofibers have been investigated thoroughly through scanning electron microscopy (SEM), transmission electron microscopy (TEM), X-ray diffraction (XRD), and thermogravimetric analysis (TGA). Attenuated total reflection Fourier transform infrared spectroscopy (ATR-FTIR), X-ray photoelectron spectroscopy (XPS) and Auger electron spectroscopy (AES) have also been employed to analyze the coordination interactions and chemical states of the core–shell nanofiber surface. Moreover, a static simulation of an electric-field-induced experiment has been carried out and energy-dispersive X-ray analysis (EDS) has been performed to demonstrate the field-induced mechanism. The results prove the fact that the electric field plays an important role on the induction of silver migration and formation of core–shell nanofibers. On the other hand, UV-Vis spectrophotometry has been used to test the catalytic properties of the samples for the reduction of methylene blue (MB) by NaBH4, it shows that PVP/Ag core–shell nanofibers hold great potential in the field of catalysis.
Introduction
Electrospinning is a simple and well-established technique for the preparation of ultrafine one-dimensional fibers with diameters ranging from the nano- to micrometric scale.1 Compared with the other nanofiber fabrication methods, electrospinning has gained a large amount of attention in the past decade as this technique is more versatile, productive, cost-effective and less demanding for processing conditions.2,3 Variety of materials such as polymers, polymer blends, sol–gels, and emulsions are the most widely used precursors for nanofiber formation.4–7 As the electrospun nanofibers and their nanofiber mats have remarkable characteristics including a very high specific surface area, high porosity within the nanoscale and good mechanical strength with light weight,8 such distinctive properties can offer good opportunities for developing advanced materials and devices.9 Moreover, multifunctional electrospun nanofibers can be feasibly obtained through the control of the fiber surface morphology, fiber orientation, and cross-sectional configuration or designing flexibility for physical/chemical modification. Based on their excellent properties, it has been researched that these nanofibers/mats have potentials for various applications in the field of bio/gas sensing,8,10 catalysis,11 sensitive substrates for SERS,12,13 antimicrobial wound dressing,14 and environment,15,16 etc.Recently, there has been increasing attention directed toward the synthesis and assembly of functional polymer–inorganic hybrid nanostructures.17,18 The electrospinning method provides operational flexibility for nanocomposite formation with different species incorporated into nanofibers. Large amounts of metallic and inorganic nanoparticles have been incorporated into electrospun nanofibers for achieving desired functional properties which combine the advantages of polymers such as structural flexibility and lightweight with the outstanding characteristics of inorganic materials such as excellent electrical, optical, magnetic, and catalytical properties,19–21 etc. Furthermore, inorganic nanoparticles, when they are in solution, have a tendency to form aggregates for minimizing their surface energy, which diminishes their intriguing properties. To overcome this drawback, inorganic nanoparticles can be formed and immobilized onto nanofibers which provide high surface area, high porosity, and flexibility.14,22,23 Through electrospinning, nanoparticles of predetermined size can be obtained and stably exist in the fiber against aggregation with a homogeneous dispersion. In recent years, immobilization of metal nanoparticles onto electrospun polymeric nanofibers has been widely concerned. Metallic nanoparticles and their corresponding metallic oxides, for instance, silver,24 gold, copper,25 and TiO2,26 ZnO,27 have found wide applications based on their outstanding physical, chemical, and biological properties. Among them, silver nanoparticles have received extensive interest due to their natural advantages of excellent antimicrobial activity, relatively low toxicity to humans, and good performance on catalytic application.22,23,28,29 The formation of Ag nanoparticles were usually prepared by reducing AgNO3 with various reducing agents prior to electrospinning30 or by post-processing of the electrospun polymer/AgNO3 composite nanofibers using heat, UV radiation or chemical reduction.31,32 Morphological characteristics of the polymer–inorganic composite nanofibers play an important role in presenting their properties. For example, core–shell nanofibers are quite compelling since their morphology could combine the advantages of the core and the shell materials and enhance the material properties. Various methods have been employed to produce core–shell nanofibers. Inorganic–inorganic coaxial nanofibers have been fabricated by co-electrospinning of two different sol–gel system.33 On the other hand, polymer–polymer coaxial nanofibers can be obtained by using coaxial electrospinning setup or they can also be prepared by single spinneret electrospinning of blends of the two types of polymers with different surface energy.34,35 However, the fabrication of polymer–organic core–shell nanofibers is now somewhat facing difficulties and challenges as a result of the incompatibility between organic and inorganic materials.
Recently, there have been several publications which investigated hybrid electrospun nanofibers with silver nanoparticles loaded in the polymer matrix. Jin et al., have prepared PVP nanofibers containing silver nanoparticles via a simple one-step method using DMF.36 The average size of the Ag nanoparticles was about 4 nm. DMF was used as a solvent for the PVP as well as a reducing agent for the Ag+ ions in the PVP solutions. Wang et al., have presented a convenient route to PVP/Ag nanocomposite by electrospinning.37 Silver nanoparticles were prepared through reduction of AgNO3 by ethanol under refluxing condition. Jin et al., have employed heat treatment of various compositions of AgNO3-doped PVP composite nanofibers fabricated by electrospinning to produce two kinds of silver species: (i) Ag nanoparticles dispersed in PVP nanofibers, when the loading of AgNO3 was 5 wt%, and a net-like silver nanofibers film when the loading of AgNO3 was five times greater than that of PVP in the composite nanofibers.38 Dong et al. have reported uniform Ag nanoparticle-embedded PVA and PVP nanofibers prepared directly by electrospinning, which exhibited excellent antibacterial activity.39 Yu et al. have successfully developed a modified coaxial electrospinning process to prepare a new type of antibacterial nanofiber in which AgNPs were distributed only on the surface of PAN nanofibers to improve their antibacterial capability.40 Zhang et al. have reported a facile technique for macroscopic scale alignment of AgNWs in an arranged polymer nanofiber mat using the magnetic-field-assisted electrospinning technique.41 With this method, AgNWs can be assembled in the same polymer nanofiber and arranged parallel to each other, while the polymer nanofibers can also be arranged by the magnetic-assisted-field electrospinning technique.
In this study, we have successfully fabricated PVP/Ag nanofibers with core–shell structure by electrospinning of PVP/AgNO3 blend solutions. Based on the research reported previously, the ratio of AgNO3 relative to the polymer is usually very small in the electrospun solution, which results in the relatively low silver content in the fibers.42 What's more, a certain amount of the silver nanoparticles are embedded in the fibers, which decreases the amount of accessible sites and greatly limits their applications. To work around this problem and have the surface of the polymer adequately covered with Ag nanoparticles, a large ratio of AgNO3 was incorporated into the polymer solution in our experiments. Meanwhile, a horizontal positive electric field was added via a silver filament inserted into the electrospun solution to induce the silver ions to move outward and generate metallic silver on the surface of fibers. PVP as a chemically functional template was chose as the polymeric core material in our work. For the preparation of PVP/Ag nanofibers, PVP played an important role both as the reducing agent and as the capping agent during the electrospinning process. When silver ions were attached to PVP, electron transfer took place via partial donation of oxygen loan pair electrons of PVP to the vacant orbitals of the silver surface, which resulted in the reduction of silver ions. The formation of the core–shell structure took full advantages of the binding capability of pyrrolidyl groups to silver ions and silver nanoparticles. In our experiments, neither additional reducing agents nor any toxic reagents were employed for Ag nanoparticles generation.
Experimental
Materials
Silver nitrate (AgNO3, >99.5%), methylene blue (MB), ethanol (EtOH) and glutaraldehyde (GA) were purchased from Sigma Aldrich. Polyvinylpyrrolidone (PVP K90, MW = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300
300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) was supplied by Shanghai Chemical Reagent Co., China. All materials were used directly without further purification. Distilled water was used throughout the experiment.
000) was supplied by Shanghai Chemical Reagent Co., China. All materials were used directly without further purification. Distilled water was used throughout the experiment.
Electrospun solutions
The PVP solution was prepared by dissolving PVP powder in a mixed solvent of distilled water and ethanol with the volume ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 and stirring at room temperature for 12 h. After that, a suitable AgNO3 was added to the PVP solution and then stirred for another 1 h in dark room to ensure complete dissolution of the mixture and obtain a homogeneous mixed solution. The PVP and AgNO3 were blended with different weight ratios, but the total polymer concentration was kept at 15% (w/w) in all solutions.
8 and stirring at room temperature for 12 h. After that, a suitable AgNO3 was added to the PVP solution and then stirred for another 1 h in dark room to ensure complete dissolution of the mixture and obtain a homogeneous mixed solution. The PVP and AgNO3 were blended with different weight ratios, but the total polymer concentration was kept at 15% (w/w) in all solutions.
Electrospinning of PVP/Ag nanofibers
A modified electrospinning apparatus was introduced to prepare nanofibers, as shown in Fig. 1. The glass syringe was fitted with a silver filament inside which was used to generate a transverse built-in electric field. As silver ions are positively charged, when electrospinning, silver ions would migrate outward and aggregate on the surface of fibers under the force of the transverse built-in electric field. Then the formation, nucleation, and growth of silver nanoparticles occurred and a continuous shell structure was further achieved.In our experiments, the syringe was fitted on a syringe pump (WSZ-50FZ, Zhejiang University Medical Instrument Co. Ltd) with a solution flow rate of 1 mL h−1 during electrospinning. A power supply (BGG4-21, BMEI CO., Ltd) was used to provide a high voltage of 20 kV between the tip of the silver filament and a metal collector. The electrospun fibers were collected on an aluminum foil. The tip-to-collector distance was set at 20 cm for the electrospinning of the prepared solutions. All of the electrospinning processes were carried out at 25 °C. The obtained nanofibers were dried in a vacuum oven for 24 h at 30 °C for characterizations.
Characterization
The morphology, uniformity and dimensions of the core–shell PVP/Ag nanofibers were performed by using scanning electron microscope (SEM) (Hitachi S-4700, Japan). The samples were sputter-coated with a thin layer of gold before SEM experiments. In order to determine the average fiber diameter form SEM images, around 50 fibers were analyzed. Transmission electron microscope (TEM) (S-800 Hitachi, Tecnai G2 20 S-TWIN FEI, JEM-100CX JEOL) was used for the detailed morphological investigation of the core–shell PVP/Ag nanofibers, as well as measurement of core and shell thicknesses. XRD (Rigaku D/Max2500VB+/PC) patterns of the samples were recorded over a diffraction angle (2θ) from 5 to 90° using a wide-angle X-ray diffraction analyzer (WAXD, D/Max 2500 VB2t/PC, 40 kV, 100 mA) equipped with a monochromic Cu Kα (λ = 0.154 nm) X-ray source. The surface compositions of the core–shell PVP/Ag nanofibers were determined by using X-ray photoelectron spectroscopy (XPS, Thermo Electron Corporation, Escalab 250, Germany). Also, energy dispersive X-ray spectroscopy (EDS) analysis was also used to analyze to elemental composition. Thermo gravity (TG) was conducted on a Seiko EXSTAR 6000 thermal analyzer at the scanning rate of 10 °C min−1 in a nitrogen flow. ATR-IR spectra of the PVP/Ag nanofiber mats were carried out with a Nicolet 5700 FTIR spectrometer (Thermo Electron Corporation, Waltham, MA, USA), which operated from 4000 to 400 cm−1. Optical absorption spectra were measured at different time on an SP752-PC ultraviolet-visible (UV-vis) spectrophotometer over the wavelength range from 300 to 800 nm.Catalytic properties of PVP/Ag nanofiber mats
To investigate the catalytic properties of the PVP/Ag nanofiber mats, a suitable 2 cm × 2 cm size of mats and a mixed solution of dye MB (1 × 10−3 mol L−1) and NaBH4 (0.5 mol L−1) with volume ratio of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 were prepared for catalytic examinations. After adding the mats to the mixed solution, the color of the solution became pale gradually. The catalytic performance was investigated by monitoring the variation in optical absorption of the dye with a UV-vis spectrometer.
3 were prepared for catalytic examinations. After adding the mats to the mixed solution, the color of the solution became pale gradually. The catalytic performance was investigated by monitoring the variation in optical absorption of the dye with a UV-vis spectrometer.
Static simulation of electric-field induced experiment
The static simulation of electric-field induced experiment was carried out by inserting vertically the silver filament into a glass bottle that was fitted with the electrospun solution. We used a high voltage DC generator (BGG4-21, BMEI CO., Ltd) to generate a constant positive high voltage (20 kV) on the silver filament for 10 min. Then liquid nitrogen was used to freeze the mixed solution instantly. At last, the sample was transferred into a freeze-drying vessel for at least 48 h to be lyophilized. EDS analysis was used to determine the elemental composition at different positions of the lyophilized PVP/AgNO3 composites.Results and discussion
Reduction mechanisms of silver by PVP
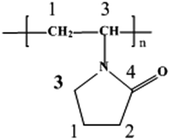
PVP has a structure of a polyvinyl skeleton with polar pyrrolidyl groups. The nitrogen and oxygen atoms in the polar groups of the PVP molecules both have lone unpaired electrons, which may be donated into sp hybrid orbitals of Ag+ to form coordination compounds. As an sp hybrid orbital usually construct a linear coordinative bond, two possible styles of bonding may take place when Ag+ interact with PVP molecules43–45 [see eqn (1) in Scheme 1]. The two kinds of coordination bond (PVP molecular intra- and inter-chain interactions) can significantly decrease the chemical potential and further promote the PVP-attached Ag+ to be reduced into metallic Ag more easily [see eqn (2) in Scheme 1]. During the electrospinning process, it was found that the color of the PVP/Ag nanofiber mats changed gradually from white, to pale yellow, and to gray with the growth of time, which implies the nucleation and growth of silver nanoparticles on the surface of fibers.Morphological observations of PVP/Ag nanofiber mats
Fig. 2 shows SEM images of the PVP/Ag nanofiber mats. It can be clearly seen that individual silver nanoparticles distribute uniformly and in high coverage density on the surface of the PVP nanofibers. This indicates that some silver nanoparticles with a diameter of 60–80 nm are spontaneously generated on the PVP nanofibers. As is typical for electrospun nanofibers, there is a distribution of fiber diameters ranging from 400 to 600 nm, and the fibers are randomly oriented. | ||
| Fig. 2 SEM images of electrospun PVP/Ag nanofibers with different weight ratio: (a) PVP/AgNO3 = 8/2, (b) PVP/AgNO3 = 7/3, (c) PVP/AgNO3 = 6/4, (d) PVP/AgNO3 = 5/5. | ||
In our experiments, it is found that the color of the PVP/Ag nanofiber mats changed from white to light yellow during the electrospinning process, indicating the reduction of silver ions and formation of silver nanoparticles on the PVP nanofibers. As mentioned above, the silver ions cannot be reduced completely by PVP, which depends on the amount of electron transfers. As the density of the carbonyl groups is critical for the reduction of silver ions to metallic silver nanoparticles, a limited number of the carbonyl groups exposed to the nanofibers could result in the incomplete reduction of silver ions. Fig. 3 shows the SEM images of PVP/Ag nanofiber mats without any post-processing (a) and treated by glutaraldehyde vapor (b). Comparing Fig. 3a and b, it can be clearly observed that the density of silver nanoparticles becomes higher on the surface of PVP nanofibers with continuous uniform coverage after the post-processing of glutaraldehyde vapor. The result proves the uncomplete reduction of silver ions on the surface of PVP nanofibers.
 | ||
| Fig. 3 SEM images of PVP/Ag nanofibers (PVP/AgNO3 = 6/4) without any post-processing (a), and treated by glutaraldehyde vapor (b). | ||
To clearly assess the core–shell structure of the PVP/Ag nanofibers, the composite fibers were directly electrospun onto TEM grids coated with lacey support films. Fig. 4 shows the TEM images of a fiber sample coated with silver nanoparticles with an average wall thickness of 20–30 nm. The coating appears to have covered the fiber completely, with some spherical tin silver particles having formed and distributing homogeneously on the fiber surface and next to the fiber. These spherical silver particles are considered to be the result of the nucleation processes from the coating reaction. We believe that the coating reaction first creates nucleation sites of metallic silver on the surface of fiber. As the coating time increased and the reduction reaction continued, the nucleation sites grew together to form a continuous coating of silver shell.
 | ||
| Fig. 4 TEM micrographs of core–shell structure PVP/Ag nanofibers with different weight ratio: (a) PVP/AgNO3 = 8/2, (b) PVP/AgNO3 = 7/3, (c) PVP/AgNO3 = 6/4, (d) PVP/AgNO3 = 5/5. | ||
Structure of PVP/Ag nanofiber mats
In order to obtain the structure information of the element silver in our nanofiber mats, XRD detection was conducted. In other words, we want to know whether silver ions have been reduced to metallic silver by PVP which plays a role both as the reducing agent and as the capping agent during the electrospinning process. Fig. 5 shows the X-ray diffraction pattern of PVP/Ag nanofiber mats. The diffraction peaks at 2θ = 38.1°, 44.3°, 64.4°, 77.4° are corresponding to the (111), (200), (220), (311) lattice planes of metallic silver respectively, which are in agreement with the face-centered cubic (fcc) structure of Ag (Joint Committee on Powder Diffraction Standards file (JCPDS) no. 87-0597). In experiments, we notice that no diffraction peaks of Ag2O can be seen from Fig. 1, which suggests that the existence of PVP can prevent the oxidation of silver nanoparticles. Furthermore, the XRD pattern confirms that silver nanoparticles exist on the surface of the nanofiber mats, which indicates the reduction of silver ions by PVP at the same time of fiber formation.Optical properties of PVP/Ag nanofiber mats
UV-Vis spectroscopy is one of the most widely used characterization methods for optical response of nanoparticle. Fig. 6 shows the UV-Vis absorption spectra of the PVP/Ag nanofiber mats with different weight ratio. In experiments, PVP/Ag nanofiber mats were firstly cut into 2 cm × 2 cm in size and then the prepared mats were dissolved into deionized water for UV-Vis spectra detection. All the mats were required the same weight. It can be seen that the appearance of characteristic peak at 420 nm clearly indicates the formation of Ag nanoparticle, which proves the reduction of silver ions by PVP during electrospinning process. With the increase of silver nitrate content in the precursor solution, more silver nanoparticles form on the surface of PVP in fiber mats. As a result, the intensity of the absorption peak of Ag also increases with the same trend, as shown in Fig. 6. In addition, no obvious shift of the absorption peak is observed, which suggests that the size of the Ag nanoparticle is uniform and not affected by the content of silver ions. This can be attributed to the structure of PVP, including steric effect and chemical coordination, which provides an obstacle to silver diffusion and may decrease the silver grain growth. | ||
| Fig. 6 UV-Vis spectra of PVP/Ag nanofiber mats with different weight ratio: (a) PVP/AgNO3 = 8/2, (b) PVP/AgNO3 = 7/3, (c) PVP/AgNO3 = 6/4, (d) PVP/AgNO3 = 5/5. | ||
ATR-FTIR spectra of PVP/Ag nanofiber mats
To further acquire information on the interaction of PVP with the silver ions, ATR-FTIR spectra characterizations of PVP/Ag nanofiber mats are performed. Fig. 7 shows the ATR-FTIR spectra of pure PVP and PVP/Ag nanofiber mats with different weight ratio. As seen in curve (a), for pure PVP, the band at 1659 cm−1 is ascribed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration, the band at 1421 cm−1 is due to the deformation vibration of CH2 in the skeleton chain of PVP, another typical characteristic absorption peak at 1289 cm−1 is thought to be the C–N stretching vibration. It is obvious from other curves (b–d) that the ATR-FTIR of the PVP/Ag nanofiber mats is similar to that of pure PVP. This indicates the effect that PVP as a good substrate plays an important role in composite nanofiber mats. However, a few small changes can be observed when compared to curve (a). From curve (b) to curve (d), with the increase of silver content in composite nanofiber mats, the C
O stretching vibration, the band at 1421 cm−1 is due to the deformation vibration of CH2 in the skeleton chain of PVP, another typical characteristic absorption peak at 1289 cm−1 is thought to be the C–N stretching vibration. It is obvious from other curves (b–d) that the ATR-FTIR of the PVP/Ag nanofiber mats is similar to that of pure PVP. This indicates the effect that PVP as a good substrate plays an important role in composite nanofiber mats. However, a few small changes can be observed when compared to curve (a). From curve (b) to curve (d), with the increase of silver content in composite nanofiber mats, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O peak is found to shift to lower wavenumber (from 1659 to 1634 cm−1) compared with pure PVP. The decrease in wavenumber for C
O peak is found to shift to lower wavenumber (from 1659 to 1634 cm−1) compared with pure PVP. The decrease in wavenumber for C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O absorption may be caused by bond weakening via partial donation of oxygen loan pair electrons of PVP to the vacant orbitals of the silver surface.44 Based on the change of a red shift of C
O absorption may be caused by bond weakening via partial donation of oxygen loan pair electrons of PVP to the vacant orbitals of the silver surface.44 Based on the change of a red shift of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O absorption band, it can be implied that a coordination between silver ions or metallic silver and oxygen atoms of carbonyl in PVP molecules may occur. Nevertheless, there is not much difference of the band of C–N absorption vibration. All the results prove that the coordination between PVP and silver was mainly not on N, but on O, the chemical bonds of Ag:O hold the main form in the PVP/Ag nanofiber mats. Such results may arise from the steric effect that the pyrrolidyl has on the coordination between N and Ag+, which obstructs the electron donating from N to vacant orbitals of silver, though the electronegativity of N is lower than that of O. On the basis of the steric effect, the coordination between C
O absorption band, it can be implied that a coordination between silver ions or metallic silver and oxygen atoms of carbonyl in PVP molecules may occur. Nevertheless, there is not much difference of the band of C–N absorption vibration. All the results prove that the coordination between PVP and silver was mainly not on N, but on O, the chemical bonds of Ag:O hold the main form in the PVP/Ag nanofiber mats. Such results may arise from the steric effect that the pyrrolidyl has on the coordination between N and Ag+, which obstructs the electron donating from N to vacant orbitals of silver, though the electronegativity of N is lower than that of O. On the basis of the steric effect, the coordination between C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and Ag+ is more effortless than that between N and Ag+, and the chemical bonding between C
O and Ag+ is more effortless than that between N and Ag+, and the chemical bonding between C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and Ag is also stronger. So the reaction between N and silver is less important and the blue shift of absorption band can be only seen for C
O and Ag is also stronger. So the reaction between N and silver is less important and the blue shift of absorption band can be only seen for C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, not for C–N.
O, not for C–N.
 | ||
| Fig. 7 ATR-FTIR spectra of (a) pure PVP nanofiber mats, and different weight radio of composite nanofiber mats: (b) PVP/AgNO3 = 8/2, (c) PVP/AgNO3 = 6/4, (d) PVP/AgNO3 = 5/5. | ||
Thermal analysis of PVP/Ag nanofiber mats
Thermogravimetric analysis (TGA) is used to measure the weight percentage of silver in the composite nanofiber mats. Fig. 8 shows the TG and DTG curves of AgNO3 (a), pure PVP (b), and PVP/AgNO3 nanofiber mats with weight ratio of 6/4 (c) and 5/5 (d). It can be seen that AgNO3 begins to decompose already at a temperature of about 450 °C, and the complete decomposition is at about 520 °C, as is obvious from curve (a). The residual component should be silver, as the remaining mass is found to be 63.5% which is agreement to the silver content of silver nitrate. For pure PVP, as seen in curve (b), the sample presents two successive weight loss process from room temperature to 800 °C. The initial weight loss (7.2%) until 100 °C and the major weight loss (88.1%) between 400 and 480 °C are observed due to evaporation of absorbed water and thermal degradation of PVP, respectively. So the total weight loss of PVP is about 95.3%. The TGA studies of AgNO3 and pure PVP showed above are performed for comparison. From curve (c) and (d), we can find that the PVP/Ag nanofiber mats with different weight ratio have shown three-step weight loss at comparable temperature range. The first stage is from ambient temperature to 200 °C, which corresponds to loss of absorbed water and small molecules. In the second stage, it is noted that the TGA thermograms of PVP/Ag nanofiber mats decrease sharply at 200 °C, which is due to the decomposition of NO3− derived from AgNO3. For the samples PVP/AgNO3 = 6/4 (c) and PVP/AgNO3 = 5/5 (d), the NO3− content of AgNO3 amounts to about 36.5% so that the final mass of NO3− within the fiber mats should amount to about 14.6% and 18.2%, respectively. We notice that the weight loss of NO3− is 15.1% in curve (c) and 18.9% in curve (d), which is almost consistent with the theoretical values. The third process takes place from 300 °C to 500 °C and accompanies a mass loss of 35.3% (PVP/AgNO3 = 6/4) and 17.6% (PVP/AgNO3 = 5/5), which is attributed to the thermal degradation of PVP. However, the weight loss of PVP in composite nanofiber mats are much lower than the theoretical values (60% for sample PVP/AgNO3 = 6/4 and 50% for sample PVP/AgNO3 = 5/5). This may be because of the strong interaction, mainly the coordination and chemical bonding between C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and Ag. The more content of Ag in the nanofiber sample, the stronger the interaction between PVP and Ag. For this reason, we find that the addition of AgNO3 results in the incomplete decomposition of PVP, and this phenomenon is more obvious with the increasing amount of silver content, as seen in curve (c) and curve (d). So the residual components of PVP/AgNO3 nanofiber mats should be silver decomposed by AgNO3 and tar content produced by the incomplete thermal degradation of PVP. The remaining mass of sample PVP/AgNO3 = 6/4 is found to be 38.4% which is lower than that of sample PVP/AgNO3 = 5/5. One of the major factors is the difference of silver content, and on the other hand, PVP with less addition of AgNO3 can decompose more adequately and remain less tar content. Furthermore, comparing with the TG curve of pure PVP, it is also noted that the main decomposition temperature of PVP/AgNO3 nanofiber mats (decomposition onset at about 300 °C) is lower than that of pure PVP (decomposition onset at 400 °C), which is possibly due to the catalytic activity of Ag resulting in oxidative and earlier decomposition of PVP. A similar result was reported where the thermal degradation temperature of polymer was decreased with the presence of ZnO as inorganic filler.46 As the influence of electric field induction during the process of electrospinning, most of silver transfers and distributes on the surface of PVP nanofibers. Such inhomogeneous distribution may led to a broader temperature range (300–500 °C) of PVP degradation. Finally, no weight loss range of AgNO3 can be observed from the TGA curves from the PVP/AgNO3 nanofiber mats. Instead, NO3− appears alone at 200 °C with a sharp weight loss, indicating that Ag and NO3− is separated from each other in the composite nanofiber mats, not present in the form of AgNO3. The separation is considered to be caused by the electric field induction which promotes the reverse migration of Ag+ and NO3−. Then, along with the reduction of silver ions, the composite sample should be specifically called PVP/Ag nanofiber mats.
O and Ag. The more content of Ag in the nanofiber sample, the stronger the interaction between PVP and Ag. For this reason, we find that the addition of AgNO3 results in the incomplete decomposition of PVP, and this phenomenon is more obvious with the increasing amount of silver content, as seen in curve (c) and curve (d). So the residual components of PVP/AgNO3 nanofiber mats should be silver decomposed by AgNO3 and tar content produced by the incomplete thermal degradation of PVP. The remaining mass of sample PVP/AgNO3 = 6/4 is found to be 38.4% which is lower than that of sample PVP/AgNO3 = 5/5. One of the major factors is the difference of silver content, and on the other hand, PVP with less addition of AgNO3 can decompose more adequately and remain less tar content. Furthermore, comparing with the TG curve of pure PVP, it is also noted that the main decomposition temperature of PVP/AgNO3 nanofiber mats (decomposition onset at about 300 °C) is lower than that of pure PVP (decomposition onset at 400 °C), which is possibly due to the catalytic activity of Ag resulting in oxidative and earlier decomposition of PVP. A similar result was reported where the thermal degradation temperature of polymer was decreased with the presence of ZnO as inorganic filler.46 As the influence of electric field induction during the process of electrospinning, most of silver transfers and distributes on the surface of PVP nanofibers. Such inhomogeneous distribution may led to a broader temperature range (300–500 °C) of PVP degradation. Finally, no weight loss range of AgNO3 can be observed from the TGA curves from the PVP/AgNO3 nanofiber mats. Instead, NO3− appears alone at 200 °C with a sharp weight loss, indicating that Ag and NO3− is separated from each other in the composite nanofiber mats, not present in the form of AgNO3. The separation is considered to be caused by the electric field induction which promotes the reverse migration of Ag+ and NO3−. Then, along with the reduction of silver ions, the composite sample should be specifically called PVP/Ag nanofiber mats.
 | ||
| Fig. 8 TGA thermograms of (a) pure AgNO3, (b) pure PVP and different weight radio of PVP/Ag core–shell nanofiber mats: (c) PVP/AgNO3 = 6/4, (d) PVP/AgNO3 = 5/5. | ||
Surface analysis of the core–shell PVP/Ag nanofiber mats
To clarify the elemental and chemical states of the obtained PVP/Ag nanofiber mats, X-ray photoelectron spectroscopy technique is employed to analyze the composition of the composite fiber mats. In our work, we expect to get the core–shell nanofibers with structure of metal shell coated polymer nuclear layer. XPS is precisely used to probe the immobilization of Ag nanoparticles on the surface of PVP. Fig. 9a shows the typical XPS spectrum of the PVP/Ag nanofiber mats. The occurrence of carbon, nitrogen, and oxygen signals are mainly ascribed to PVP which constitutes the fiber substrate. Various Ag binding energy peaks are also observed from XPS full spectrum, which reveals that silver ions are reduced and immobilized on the surface of PVP nanofiber. In addition, part of the nitrogen and oxygen signals are derived from nitrate ions provided by AgNO3. All the results confirms that PVP/Ag core–shell nanofibers are composed of four elements of C, N, O and Ag, no other peaks can be found. Peak positions and respective assignments are gathered in Table 1. | ||
| Fig. 9 XPS spectra of the PVP/Ag (PVP/AgNO3 = 6/4) nanofiber mats and C 1s, O 1s, N 1s and Ag 3d regions. | ||
| Pure PVP | PVP/Ag | Assignment | |
|---|---|---|---|
| C 1s 1 | 285.0 | 285.0 | C–H &C–C |
| C 1s 2 | 285.4 | 285.4 | C2 showed in Scheme 2 |
| C 1s 3 | 286.2 | 286.2 | C–N |
| C 1s 4 | 288.0 | 288.0 | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O O |
| N 1s 1 | 399.6 | 399.68 | Nitrogen in pyrrolidyl |
| N 1s 2 | 406.6 | Nitrogen in nitrate (NO3−) | |
| O 1s 1 | 530.9 | 531.5 | C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O O |
| O 1s 2 | 532.9 | Oxygen in nitrate (NO3−) | |
| Ag 3d 1 | 368.2/368.8 | Ag 3d5/2 | |
| Ag0 (368.2) Ag+ (368.8) | |||
| Ag 3d 2 | 374.2/374.8 | Ag 3d3/2 | |
| Ag0 (374.2) Ag+ (374.8) |
The other panels in Fig. 9 show high-resolution XPS regions of various elements for the PVP/Ag nanofibers. For clarity, each of these elements have been fitted by Gaussian Lorentzian mixed function (20% Lorentzian and 80% Gaussian). Through curve fitting, it can be seen that C 1s (Fig. 9b) are fitted with a maximum of 4 peaks centered at 285.0, 285.4, 286.2, 288.0 eV, assigned to sp3 carbon, indicating C1 (C–C & C–H), C2, C3 (C–N) and C4 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), respectively. This is due to the different chemical environments of carbon atoms for PVP molecule43 (see Scheme 2). That C3a and C3b have the same chemical environment is just attributed to the same binding energy of C–C and C–H carbon atoms.
O), respectively. This is due to the different chemical environments of carbon atoms for PVP molecule43 (see Scheme 2). That C3a and C3b have the same chemical environment is just attributed to the same binding energy of C–C and C–H carbon atoms.
N 1s region (Fig. 9c) shows very distinctively a peak at 399.7 eV, which is ascribed to the nitrogen atoms of pyrrolidyl. The N 1s peak seems not be influenced by the silver shell, as no obvious shift is seen. This reveals that there is a limit to the coordination of silver ions and nitrogen atoms, and little electron transfer occurs between them. The explanation is similar to the ATR-FTIR result discussed above. Another visible peak at 406.6 eV arises from nitrogen atoms of nitrate ionic groups, which has a much higher binding energy than that of organic nitrogen. It is unambiguous that the nitrate ionic groups remain trapped in the PVP/Ag nanofiber mats accompanying by the electrospinning process, which is consistent with the TGA results.
In Fig. 9d, the O 1s profile is asymmetric and can be fitted into two symmetrical peaks centered at 531.5 and 532.9 eV. This implies the presence of two different kinds of O species in the composite fiber mats. The first one is associated with oxygen doubly bound to carbon, which indicates the main component of oxygen atom as carbonyl existing in pyrrolidyl groups. Comparing with pure PVP, it is worth mentioning herein that the O 1s peak of the carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) oxygen atom shifts to higher binding energy (from 530.9 to 531.5 eV), implying the decrease of electron density, due to the coordination between PVP and Ag nano-shell and electron transfer from carbonyl (C
O) oxygen atom shifts to higher binding energy (from 530.9 to 531.5 eV), implying the decrease of electron density, due to the coordination between PVP and Ag nano-shell and electron transfer from carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) oxygen atom to metallic Ag. The results prove the reduction reaction and chemical bonding between C
O) oxygen atom to metallic Ag. The results prove the reduction reaction and chemical bonding between C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and Ag. At higher binding energy, peak centered at 532.9 eV, as discussed by N 1s region, is expected to the oxygen component in nitrate groups.
O and Ag. At higher binding energy, peak centered at 532.9 eV, as discussed by N 1s region, is expected to the oxygen component in nitrate groups.
Ag 3d region (Fig. 9e) is a doublet with spin–orbit separation of 6.0 eV.47 The two peaks showed in the panel result from Ag 3d5/2 and Ag 3d3/2, respectively. It is obvious that the Ag regions of the XPS spectra are very sensitive to the chemical environment around the Ag nano-shell. Through curve fitting, it can be seen that the two peaks centered at 368.2 and 374.2 eV reveal that silver is present on the surface of the fiber mats and appears in its metallic form (Ag0). Another two peaks, centered at 368.8 and 374.8 eV, a little bit higher than that of metallic Ag, is regarded as the small contribution of nonreduced silver (Ag+) trapped in the fiber mats. The whole peaks positions are between those for metallic Ag (368.2 eV for Ag 3d5/2, 374.2 eV for Ag 3d3/2) and for Ag+ (368.8 eV for Ag 3d5/2, 374.8 eV for Ag 3d3/2), indicating the occurrence of electron transfer and a strong interaction between the carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) of PVP core and the Ag shell.
O) of PVP core and the Ag shell.
In the analysis of XPS, in spite of the curve fitting, it can be seen the chemical shift of elemental Ag is very small, which can not clearly express the chemical valence state of silver in the fiber mats. In order to know more information on the interaction between Ag and PVP, AES of the PVP/Ag composite fiber mats is further investigated. In experiments, the sample was irradiated with X-rays from a monochromated Al Ka source (hν = 1486.6 eV) operating at 150 W. Survey spectra is acquired and showed in Fig. 10. From the spectra, we can know the peak of AgMNN is located at 1131.5 eV (BE), namely 355.1 eV (KE). As the peak of Ag 3d5/2 is centered at 368.4 eV (Fig. 9e), the auger parameter can be calculated to be 723.5 eV (AP = 355.1 + 368.4). Table 2 lists the auger parameters and kinetic energy values of silver with several common valence (www.lasurface.com). Comparing with the data showed in Table 2, it can be seen that the auger parameter and kinetic energy of PVP/Ag are much smaller than that of pure Ag, while closed to that of AgNO3, indicating that parts of unreduced silver are contained in the sample with charge of plus one. As the silver in sample does not exist in isolation, there should be a degree of interaction between metallic silver or silver ions and PVP, which results in the obvious chemical shift showed in Table 2. As discussed above, a coordination between metallic silver and oxygen atoms of carbonyl in PVP molecules has been proved. On account of the higher electronegativity of oxygen, metallic silver may be considered as the existence with higher valence. On the other hand, a section of silver ions with charge of plus one has not been reduced and trapped in the composite fibers. It is for this reason that the auger parameter and kinetic energy of PVP/Ag are closed to that of pure AgNO3 and much smaller than that of pure silver.
| Al Ka | Ag | AgNO3 | Ag2O | PVP/Ag |
|---|---|---|---|---|
| KE-3d5/2, M4N45N45 | 358.2 | 354.2 | 356.6 | 355.1 |
| AP-3d5/2, M4N45N45 | 726.1 | 722.5 | 724.5 | 723.5 |
| Assignment | Ag0 | Ag+ | Ag–O | Ag0–O/Ag+–O |
Demonstration of field-induced mechanism
In order to demonstrate the field-induced mechanism on the formation of PVP/Ag core–shell nanofiber, a static simulation of electric-field induced experiment is carried out. After the process of lyophilization, the content of elements at different positions of PVP/AgNO3 composites is detected to illustrate the transportation of silver ions induced by electric field. Fig. 11 shows the EDS (energy-dispersive X-ray analysis) patterns of the selected regions (inside, middle, and outside of sample) in the same cross section of columnar PVP/AgNO3 composites. Elements carbon, nitrogen, oxygen, and silver are detected, and the corresponding weight and atomic percentages for the three positions are listed in Table 3. In our experiments, we mainly discuss the content variation of element nitrogen and silver, especially silver. In Fig. 10, the intense peaks related to elemental silver are observed between 2.5 keV and 3.5 keV, pertaining to the silver characteristic lines Lα and Lβ.48 From the EDS patterns, we can see the intensity of silver increases gradually from the interior to the exterior of PVP/AgNO3 composites, indicating the inhomogeneous transverse distribution of silver. As shown in Table 3, the weight and atomic percentages of silver increase consecutively from inside to outside of sample. Based on the great change in silver content (increase from 9.42 to 33.95 of wt% and from 1.30 to 6.12 of atom%), we can prove that the electric field induces the silver ions to migrate outwards. During the process of electrospinning, this effect results in the formation of PVP/Ag core–shell nanofiber. It can be explained that silver ions tend to move outwards under the influence of electric field, so more and more silver ions will aggregate on the outer surface of fibers and can be reduced to metallic silver by PVP, and finally we obtain the silver coated PVP nanofiber. In addition, from the content variation of element nitrogen showed in Table 3, it is obvious that NO3− has a tendency to move inwards, which causes the nitrogen content of inside is greater than that of outside. All the results above prove that electric field plays an important role on the induction of silver migration and formation of core–shell nanofibers. | ||
| Fig. 11 EDS patterns of the selected regions (inside, middle, and outside) of sample in the same cross section of columnar PVP/AgNO3 composites. | ||
| Sample | Inside | |||
|---|---|---|---|---|
| Element | C | N | O | Ag |
| wt% | 38.4 | 21.9 | 30.3 | 9.4 |
| Atom% | 47.5 | 23.2 | 28.1 | 1.3 |
| Sample | Middle | |||
|---|---|---|---|---|
| Element | C | N | O | Ag |
| wt% | 36.2 | 17.8 | 29.0 | 17.0 |
| Atom% | 48.2 | 20.3 | 29.0 | 2.5 |
| Sample | Outside | |||
|---|---|---|---|---|
| Element | C | N | O | Ag |
| wt% | 28.6 | 11.9 | 25.6 | 34.0 |
| Atom% | 46.3 | 16.5 | 31.1 | 6.1 |
Catalytic properties of PVP/Ag nanofiber mats
It has been long identified that Ag nanoparticles show excellent activity and selectivity on many catalytic systems. Herein, we investigate the catalytic properties of PVP/Ag nanofiber mats which accelerate the reducing of methylene blue (MB) by NaBH4. Fig. 12 shows UV-Vis spectra for the reduction of MB with time using PVP/Ag nanofiber mats as the catalyst. As is clear from the spectra showed in Fig. 12a, in the absence of catalyst PVP/Ag nanofiber mats, the absorbance at λmax (664 nm), namely the characteristic peak of the dye, has no obvious change over time. The result indicates that the redox reaction rate between MB and NaBH4 is very slow. The other, acting as a controlled trial, after the addition of the PVP/Ag nanofiber mats, the characteristic adsorption peak of MB at 664 nm rapidly decreases with respect to reaction time and completely disappears within 4 min, as seen in Fig. 12b. The reaction can also be monitored by the color change (from blue to pale yellow) of MB solution after the addition of the catalysts. All these results illustrate the reduction of MB by PVP/Ag nanofiber mats. Moreover, with the reaction time extending, we find that the characteristic absorption peaks of silver appear at 400 nm, accompanying with increasing intensity and occurrence of blue shift. This phenomenon suggests that new silver particles are generated in solution. The final pale yellow also proves the formation of silver colloid. The reason is that the PVP/Ag nanofiber mats contain part of silver ions which is not completely reduced by PVP during the electrospinning process. When added to the solution, the residual silver ions trapped in the mats will be reduced into silver by NaBH4. As more and more new silver particles form, the absorption intensity increases over time. The occurrence of blue shift is due to the decrease of the new generated silver particle size,49 which may be related to the coordination between Ag and PVP or MB in solution.The catalytic abilities of the PVP/Ag nanofiber mats could be explained as follows.50 After the addition of PVP/Ag nanofiber mats, Ag nanoparticles loaded on the fiber membranes dissolve in the solution to form silver colloid with pale yellow color. As is known to all, the bulk of the silver sol is positively charged, the silver particles will adsorb the negatively charged BH4− by electrostatic attraction. Meanwhile, because of the surface effect of nanoparticles, there are many unsaturated chemical active spots on the surface of Ag nanoparticles, which makes them easy to combine with other atoms for stabilization. So the MB molecules are also absorbed on the surface of Ag nanoparticles. In the process of catalytic reducing of MB by NaBH4, the Ag nanoparticles embedded in or fixed on the surface of the fibers serve as an electron relay in the oxidant–reductant system, and the electron transfer occurs via the metal particles. With respect to the silver nanoparticles, MB dyes are electrophilic and BH4− ions are nucleophilic. In the reaction, the nucleophile BH4− donates electrons to silver nanoparticles, and the electrophile MB dyes would get electrons from silver nanoparticles. Base on the important role silver nanoparticles have played, the reduction process of MB by NaBH4 can be greatly accelerated.
It is known that the stability of metal nanoparticles entrapped within fiber mats is a main advantage for their industrial applications. In our experiments, the Ag nanoparticles embedded in or fixed on the surface of the fibers are highly stable and do not show any signs of aggregation after storage for long time at room temperature. Such composite nanofiber mats, with the merits of good chemical stability as well as the catalytic activity, would have a potential application in the field of heterogeneous catalysis.
Conclusions
In summary, narrow and size-controllable PVP/Ag nanofibers with core–shell structure have been successfully prepared by using the electrospinning technique under the action of electric field induction. There was a distribution of nanofiber diameters ranging from 400 to 600 nm and the average wall thickness of the nanofibers was only 20–30 nm. The dense Ag shell layer has been synthesized by polyreduction in the presence of PVP. PVP has played an important role not only as the functional template but as the reducing agent and the capping agent. Neither additional reducing agents nor any toxic reagents have been employed for Ag nanoparticles generation, which is different from previous reports. The formation of silver nanoparticles is completely confirmed by XRD and UV-Vis characterization, and the SEM images have shown that silver nanoparticles distribute uniformly and in high coverage density on the surface of the PVP nanofibers. The TEM images clearly assess the core–shell structure and display a continuous coating of silver shell. Compared with pure PVP, the ATR-FTIR and XPS spectra of the PVP/Ag nanofiber mats indicate that the coordination between PVP and Ag was mainly not on N, but on O, the chemical bonds of Ag:O hold the main form in the PVP/Ag nanofiber mats. The thermos stability of PVP/Ag nanofibers is analyzed by TGA, it shows that the addition of Ag results in the incomplete decomposition of PVP, and this phenomenon is more obvious with the increasing amount of silver content. The EDS result demonstrate the field-induced mechanism, indicating that Ag and NO3− is separated from each other in the composite nanofibers. What's more, UV-Vis spectra show that PVP/Ag nanofibers accelerate the reducing of methylene blue (MB) by NaBH4, which identifies that Ag nanoparticles have excellent activity and selectivity on many catalytic systems. The prepared PVP/Ag nanofibers hold great potential in the field of catalysis.Acknowledgements
The author would like to thank the project supported by the National Natural Science Foundation of China (Grant No. 21304005) for its financial support. This study was also supported by Open Fund from State Key Laboratory of Chemical Resource Engineering.References and notes
- A. Greiner and J. H. Wendorff, Electrospinning: A Fascinating Method for the Preparation of Ultrathin Fibers, Angew. Chem., Int. Ed., 2007, 46, 5670–5703 CrossRef CAS PubMed.
- A. Greiner and J. H. Wendorff, Functional Self-assembled Nanofibers by Electrospinning, Adv. Polym. Sci., 2008, 219, 107–171 CAS.
- G. K. Chen, D. W. Fang, K. M. Wang, J. Nie and P. MaG, Core–shell structure PEO/CS nanofibers based on electric field induced phase separation via electrospinning and its application, J. Polym. Sci., Polym. Chem. Ed., 2015, 53, 2298–2311 CrossRef CAS.
- A. Celebioglu and T. Uyar, Electrospinning of Nanofibers from Non-Polymeric Systems: Polymer-Free Nanofibers from Cyclodextrin Derivatives, Nanoscale, 2012, 4, 621–631 RSC.
- W. E. Teo and S. Ramakrishna, Electrospun Nanofibers as a Platform for Multifunctional, Hierarchically Organized Nanocomposite, Compos. Sci. Technol., 2009, 69, 1804–1817 CrossRef CAS.
- C. Burger, B. S. Hsiao and B. Chu, Nanofibrous Materials and their Applications, Annu. Rev. Mater. Res., 2006, 36, 333–368 CrossRef CAS.
- Z. M. Huang, Y. Z. Zhang, M. Kotaki and S. A. Ramakrishna, Review on Polymer Nanofibers by Electrospinning and their Applications in Nanocomposites, Compos. Sci. Technol., 2003, 63, 2223–2253 CrossRef CAS.
- J. Wang, H. B. Yao, D. He, C. L. Zhang and S. H. Yu, Facile Fabrication of Gold Nanoparticles-Poly(vinyl alcohol) Electrospun Water-Stable Nanofibrous Mats: Efficient Substrate Materials for Biosensors, ACS Appl. Mater. Interfaces, 2012, 4, 1963–1971 CAS.
- X. Lu, C. Wang and Y. Wei, One-Dimensional Composite Nanomaterials: Synthesis by Electrospinning and their Applications, Small, 2009, 5, 2349–2370 CrossRef CAS PubMed.
- K. K. H. Wong, J. L. Hutter, M. Zinke-Allmang and W. Wan, Physical Properties of Ion Beam Treated Electrospun Poly(vinylalcohol) Nanofibers, Eur. Polym. J., 2009, 45, 1349–1358 CrossRef CAS.
- A. M. Signori, K. O. Santos, R. Eising, B. L. Albuquerque, F. C. Giacomelli and J. B. Domingos, Formation of Catalytic Silver Nanoparticles Supported on Branched Polyethyleneimine Derivatives, Langmuir, 2010, 26, 17772–17779 CrossRef CAS PubMed.
- L. Zhang, X. Gong, Y. Bao, Y. Zhao, M. Xi, C. Jiang and H. Fong, Electrospun Nanofibrous Membranes Surface-Decorated with Silver Nanoparticles as Flexible and Active/Sensitive Substrates for Surface-Enhanced Raman Scattering, Langmuir, 2012, 28, 14433–14440 CrossRef CAS PubMed.
- D. He, B. Hu, Q. F. Yao, K. Wang and S. H. Yu, Large-Scale Synthesis of Flexible Free-Standing SERS Substrates with High Sensitivity: Electrospun PVA Nanofibers Embedded with Controlled Alignment of Silver Nanoparticles, ACS Nano, 2009, 3, 3993–4002 CrossRef CAS PubMed.
- P.-O. Rujitanaroj, N. Pimpha and P. Supaphol, Wound-Dressing Materials with Antibacterial Activity from Electrospun Gelatin Fiber Mats Containing Silver Nanoparticles, Polymer, 2008, 49, 4723–4732 CrossRef CAS.
- S. L. Xiao, M. W. Shen, R. Guo, S. Y. Wang and X. Y. Shi, Immobilization of Zero-Valent Iron Nanoparticles into Electrospun Polymer Nanofibers: Synthesis, Characterization, and Potential Environmental Applications, J. Phys. Chem. C, 2009, 113, 18062–18068 CAS.
- H. Shahin and E. Mady, Nanocomposites Electrospun Nanofiber Membranes for Environmental Remediation, Materials, 2014, 7, 1017–1045 CrossRef.
- Y. N. Xia, P. D. Yang, Y. G. Sun, Y. Y. Wu, B. Mayers, B. Gates, Y. D. Yin, F. Kim and H. Q. Yan, One-Dimensional Nanostructures: Synthesis, Characterization, and Applications, Adv. Mater., 2003, 15, 353–389 CrossRef CAS.
- H. Zheng, Y. Li, H. Liu, X. Yin and Y. Li, Construction of Heterostructure Materials toward Functionality, Chem. Soc. Rev., 2011, 40, 4506–4524 RSC.
- D. Zhang, A. B. Karki, D. Rutman, D. P. Young and A. Wang, Electrospun Polyacrylonitrile Nanocomposite Fibers Reinforced with Fe3O4 Nanoparticles: Fabrication and Property Analysis, Polymer, 2009, 50, 4189–4198 CrossRef CAS.
- L. Machala, R. Zboril and A. Gedanken, Amorphous Iron(III) Oxide: A Review, J. Phys. Chem. B, 2007, 111, 4003–4018 CrossRef CAS PubMed.
- C. L. Zhang and S. H. Yu, Nanoparticles Meet Electrospinning: Recent Advances and Future Prospects, Chem. Soc. Rev., 2014, 43, 4423–4448 RSC.
- Z. W. Deng, H. B. Zhu, B. Peng, H. Chen, Y. F. Sun, X. D. Gang, P. J. Jin and J. L. Wang, Synthesis of PS/Ag Nanocomposite Spheres with Catalytic and Antibacterial Activities, ACS Appl. Mater. Interfaces, 2012, 4, 5625–5632 CAS.
- A. Kumar, P. K. Vemula, P. M. Ajayan and G. John, Silver-Nanoparticle-Embedded Antimicrobial Paints Based on Vegetable Oil, Nat. Mater., 2008, 7, 236–241 CrossRef CAS PubMed.
- Y. Zhang, H. Peng, W. Huang, Y. Zhou and D. Yan, Facile Preparation and Characterization of Highly Antimicrobial Colloid Ag or Au Nanoparticles, J. Colloid Interface Sci., 2008, 325, 371–376 CrossRef CAS PubMed.
- G. B. Bagihalli, P. G. Avaji, S. A. Patil and P. S. Badami, Synthesis, Spectral Characterization, in vitro Antibacterial, Antifungal and Cytotoxic Activities of Co(II), Ni(II) and Cu(II) Complexes with 1,2,4-Triazole Schiff Bases, Eur. J. Med. Chem., 2008, 43, 2639–2649 CrossRef CAS PubMed.
- M. Heinlaan, A. Ivask, I. Blinova, H. C. Dubourguier and A. Kahru, Toxicity of Nanosized and Bulk ZnO, CuO and TiO2 to Bacteria Vibrio fischeri and Crustaceans Daphnia magna and Thamnocephalus platyurus, Chemosphere, 2008, 71, 1308–1316 CrossRef CAS PubMed.
- K. Ghule, A. V. Ghule, B. J. Chen and Y. C. Ling, Preparation and Characterization of ZnO Nanoparticles Coated Paper and its Antibacterial Activity Study, Green Chem., 2006, 8, 1034–1041 RSC.
- Y. Liao, Y. Wang, X. Feng, W. Wang, F. Xu and L. Zhang, Antibacterial Surfaces through Dopamine Functionalization and Silver Nanoparticle Immobilization, Mater. Chem. Phys., 2010, 121, 534–540 CrossRef CAS.
- M. Sureshkumar, D. Y. Siswanto and C. K. Lee, Magnetic Antimicrobial Nanocomposite Based on Bacterial Cellulose and Silver Nanoparticles, J. Mater. Chem., 2010, 20, 6948–6955 RSC.
- Q. B. Yang, D. M. Li, Y. L. Hong, Z. Y. Li, C. Wang, S. L. Qiu and Y. Wei, Preparation and Characterization of a PAN Nanofiber Containing Ag Nanoparticles via Electrospinning, Synth. Met., 2003, 137, 973–974 CrossRef CAS.
- X. Y. Xu, Q. B. Yang, Y. Z. Wang, H. J. Yu, X. S. Chen and X. B. Jing, Biodegradable Electrospun Poly(L-lactide) Fibers Containing Antibacterial Silver Nanoparticles, Eur. Polym. J., 2006, 42, 2081–2087 CrossRef CAS.
- K. H. Hong, J. L. Park, I. H. Sul, J. H. Youk and T. J. Kang, Preparation of Antimicrobial Poly(vinyl alcohol) Nanofibers Containing Silver Nanoparticles, J. Polym. Sci., Part B: Polym. Phys., 2006, 44, 2468–2474 CrossRef CAS.
- X. Zhou, C. Shang, L. Gu, S. Dong, X. Chen, P. Han, L. Li, J. Yao, Z. Liu and H. Xu, Mesoporous Coaxial Titanium Nitride–Vanadium Nitride Fibers of Core Shell Structures for High-Performance Supercapacitors, ACS Appl. Mater. Interfaces, 2011, 3, 3058–3063 CAS.
- Z. C. Sun, E. Zussman, A. L. Yarin, J. H. Wendorff and A. Greiner, Compound Core–Shell Polymer Nanofibers by Co-Electrospinning, Adv. Mater., 2003, 15, 1929–1932 CrossRef CAS.
- Y. Zhang, Z. M. Huang, X. Xu, C. T. Lim and S. Ramakrishna, Preparation of Core–Shell Structured PCL-r-Gelatin Bi-Component Nanofibers by Coaxial Electrospinning, Chem. Mater., 2004, 16, 3406–3409 CrossRef CAS.
- W. J. Jin, H. K. Lee, E. H. Jeong, W. H. Park and J. H. Youk, Preparation of Polymer Nanofibers Containing Silver Nanoparticles by Using Poly(N-vinylpyrrolidone), Macromol. Rapid Commun., 2005, 26, 1903–1907 CrossRef CAS.
- Y. Z. Wang, Y. X. Li, S. T. Yang, G. L. Zhang, D. M. An and C. Wang, A Convenient Route to Polyvinyl Pyrrolidone/Silver Nanocomposite by Electrospinning, Nanotechnology, 2006, 17, 3304–3307 CrossRef CAS.
- M. Jin, X. T. Zhang, S. Nishimoto, Z. Y. Liu, D. A. Tryk, T. Murakami and A. Fujishima, Large-Scale Fabrication of Ag Nanoparticles in PVP Nanofibers and Net-Like Silver Nanofiber Films by Electrospinning, Nanotechnology, 2007, 18, 075605 CrossRef PubMed.
- G. P. Dong, X. D. Xiao, X. F. Liu, B. Qian, Z. J. Ma, S. Ye, D. P. Chen and J. R. Qiu, Preparation and Characterization of Ag Nanoparticle Embedded Polymer Electrospun Nanofibers, J. Nanopart. Res., 2010, 12, 1319–1329 CrossRef CAS.
- D. G. Yu, J. Zhou, N. P. Chatterton, Y. Li, J. Huang and X. Wang, Polyacrylonitrile Nanofibers Coated with Silver Nanoparticles Using a Modified Coaxial Electrospinning Process, Int. J. Nanomed., 2012, 7, 5725–5732 CrossRef CAS PubMed.
- C. L. Zhang, K. P. Lv, N. Y. Hu, L. Yu, X. F. Ren, S. L. Liu and S. H. Yu, Macroscopic-Scale Alignment of Ultralong Ag Nanowires in Polymer Nanofiber Mat and Their Hierarchical Structures by Magnetic-Field-Assisted Electrospinning, Small, 2012, 8, 2936–2940 CrossRef CAS PubMed.
- W. J. Jin, H. J. Jeon, J. H. Kim and J. H. Youk, A Study on the Preparation of Poly(vinyl alcohol) Nanofibers Containing Silver Nanoparticles, Synth. Met., 2007, 157, 454–459 CrossRef CAS.
- P. Jiang, S. Y. Li, S. S. Xie, Y. Gao and L. Song, Machinable Long PVP-Stabilized Silver Nanowires, Chem.–Eur. J., 2004, 10, 4817–4821 CrossRef CAS PubMed.
- Z. T. Zhang, B. Zhao and L. M. Hu, PVP Protective Mechanism of Ultrafine Silver Powder Synthesized by Chemical Reduction Processes, J. Solid State Chem., 1996, 121, 105–110 CrossRef CAS.
- H. S. Wang, X. L. Qiao, J. G. Chen, X. J. Wang and S. Y. Ding, Mechanisms of PVP in the Preparation of Silver Nanoparticles, Mater. Chem. Phys., 2005, 94, 449–453 CrossRef CAS.
- K. Fatma, O.-A. Cagla, D. Inci, B. Necmi and U. Tamer, Polymer-Inorganic Core–Shell Nanofibers by Electrospinning and Atomic Layer Deposition: Flexible Nylon–ZnO Core–Shell Nanofiber Mats and Their Photocatalytic Activity, ACS Appl. Mater. Interfaces, 2012, 4, 6185–6194 Search PubMed.
- A. M. Ferraria, S. Boufi, N. Battaglini and M. ReiVilar, Hybrid Systems of Silver Nanoparticles Generated on Cellulose Surfaces, Langmuir, 2010, 26, 1996–2001 CrossRef CAS PubMed.
- M. Montazer and V. Allahyarzadeh, Electroless Plating of Silver Nanoparticles/Nanolayer on Polyester Fabric Using AgNO3/NaOH and Ammonia, Ind. Eng. Chem. Res., 2013, 52, 8436–8444 CrossRef CAS.
- X. H. Xia, J. Zeng, Q. Zhang, H. M. Christine and Y. N. Xia, Recent Developments in Shape-Controlled Synthesis of Silver Nanocrystals, J. Phys. Chem. C, 2012, 116, 21647–21656 CAS.
- Y. C. Tang, T. Wu, B. Hu, Q. Yang, L. Liu, B. Yu, Y. Ding and S. Y. Ye, Synthesis of Thermo- and pH-Responsive Ag Nanoparticle-Embedded Hybrid Microgels and their Catalytic Activity in Methylene Blue Reduction, Mater. Chem. Phys., 2015, 149, 460–466 CrossRef.
| This journal is © The Royal Society of Chemistry 2016 |