Facile synthesis of copper doped carbon dots and their application as a “turn-off” fluorescent probe in the detection of Fe3+ ions†
Abstract
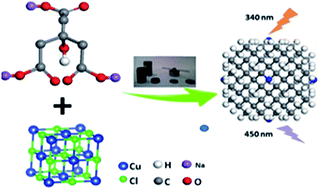
Heteroatom doped carbon dots, due to their excellent fluorescent properties and related applications, have attracted great attention from different fields. Herein, we reported for the first time a facile and economic approach to synthesize copper doped carbon dots (Cu-CDs) via a one-step hydrothermal approach. The chemical and fluorescent properties of these Cu-CDs as well as the mechanism involved for the enhancement of the fluorescent were investigated through various microscopic and spectroscopic analyses, such as transmission electron microscopy (TEM), fluorescence spectra, X-ray photoelectron spectroscopy (XPS) and Fourier transform infrared spectroscopy (FTIR). It is observed that the Cu-CDs with the average size of 3.76 nm are spherical and monodisperse. Additionally, the as-prepared Cu-CDs exhibited a strong emission at 440 nm when excited at 350 nm (λex) and the photoluminescence quantum yields (PLQY) reached 9.81% after the optimization of synthesis conditions. The excellent fluorescent properties of Cu-CDs make them great candidates for the selective and fast detection of Fe3+ in range of 0.001–200 μM, while the limit of detection (LOD) is 1 nM, which can be further applied to detect Fe3+ in human blood serum and other biomedical applications.

 Please wait while we load your content...
Please wait while we load your content...