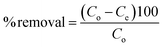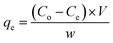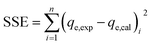Temperature dependent, shape variant synthesis of photoluminescent and biocompatible carbon nanostructures from almond husk for applications in dye removal†
Kumud Malika Tripathi‡
a,
Ankit Tyagi‡a,
Mohammad Ashfaqa and
Raju Kumar Gupta*ab
aDepartment of Chemical Engineering, Indian Institute of Technology Kanpur, Kanpur-208016, UP, India. E-mail: guptark@iitk.ac.in; Fax: +91-5122590104; Tel: +91-5122596972
bCenter for Nanosciences and Center for Environmental Science and Engineering, Indian Institute of Technology Kanpur, Kanpur-208016, UP, India
First published on 7th March 2016
Abstract
This work reports the synthesis of water soluble and photoluminescent carbon nanostructures (wsFCNS) from almond husk, a bio-waste. Effect of carbonization temperature on morphology of the synthesized carbon nanostructures is illustrated. Carbonization was carried out at three different temperatures ranging from 750 °C to 950 °C. Carbonization at higher temperature resulted in carbon nanodots having spherical morphology, while lower temperature resulted in rod shaped carbon nanostructures. Further, oxidative treatment of the as-synthesized carbon nanostructures imparts water solubility as well as photoluminescent properties over the visible to near infrared (NIR) regions of the electro-magnetic spectrum. The synthesized wsFCNS are non-toxic in nature and on direct interaction with erythrocytes, show less than 2% hemolysis. The synthesized wsFCNS were further explored for the adsorptive removal of p-nitrophenol (PNP), a model dye pollutant. The removal of PNP with wsFCNS follows pseudo first order adsorption kinetics. The proposed synthesis method could be easily scaled up for gram scale synthesis of various carbon nanostructures.
1. Introduction
Recent advances in the field of carbon based nanostructures with diverse morphologies have generated great interest in advanced scientific research owing to their excellent and unique properties.1–9 Carbon nanostructures such as fullerenes,10 multiwalled,11 and single walled carbon nanotubes,12,13 carbon nano-onions,14 carbon nanospheres,15 carbon nanorods,16 graphene,17 carbon dots,18,19 and the recently discovered graphene quantum dots (GQD)20 are reported in the literature. Among all the nano-carbons, photoluminescent carbon dots (CD) and GQD are the newer members of this family, due to their non-toxicity along with high quantum yield values in comparison to conventional materials such as semiconductor quantum dots (QD) and organic dyes.21–23 Despite having intense, tunable fluorescence and high quantum yield values, toxicity issues restrict the potential applications of QD in biological sciences. Another important, challenging issue which restricts the application of QD in biological sciences is their hydrophobicity. Higher aqueous solubility, excellent biocompatibility and high photo-stability, have made CD and GQD promising candidates for various advanced potential applications, especially in the fields of optical bio-imaging,18,24–26 cell labeling20 and drug delivery.27Photoluminescent properties, vitally associated with excitation dependent tunable photoluminescence (PL) emissions of carbon nanostructures have made them efficient probes to use as “nanolights”.28 High fluorescent brightness of carbon dots in the cellular environment along with their non-cytotoxicity have made them a promising candidate, for in vivo imaging applications.29 However, most of the carbon based nanostructures reported earlier exhibited photoluminescent properties mostly restricted over the blue/green region of the visible spectrum. Despite a few published reports,30,31 there is still need to explore facile and green synthetic methodologies to synthesize NIR emitting carbon nanostructures. Recently, NIR emitting fluorescent probes have received increasing attention, since in this region biological cells and tissues are nearly transparent and can be easily visualized and distinguished from unhealthy cells.32–35
The size and shape of carbon nanostructures have a strong influence on their electrical, optical and sensing properties.36 So, morphology-controlled fabrications of carbon nanostructures as well as a broadening of their practical applications are in great demand. Up to now, chemical modification and template based synthesis have been extensively used to fabricate the desired morphology of carbon nanostructures.37–42 High cost, expensive instruments and tedious synthetic procedures limits the wider applicability of the template based technique, which requires modification of the entire synthetic methodology to synthesize a particular morphology at low cost. These days, expensive and extensive synthetic approaches, are slowly being replaced by soot based synthetic techniques such as combustion, conventional pyrolysis and flame synthesis.14,23,43 Soot based synthetic techniques are superlative owing to their low cost and simplistic approaches. Researchers are also optimizing greener routes for the fabrication of carbon nanostructures from natural bio-precursors.22,44 There is an abundance of carbonaceous sources “bio-waste” available to us. Such as langsat empty fruit bunch,45 orange peel,46 rise husk,47 egg,48 grass,49 pomelo peel,50 watermelon peel,51 sweet pepper,32 rice,52 wood wool,43 silk cocoon,53 industrial waste,54 lignin,55 winter melon,56 fullerene waste soot57 and so on.
Herein, we have chosen almond husk as a “free carbon precursor” (normally discarded as a bio-waste), for the synthesis of various carbon nanostructures.58 The use of almonds as an energy source and nutrition leads to an increase in bio-waste and issues related to its appropriate removal. Proper handling of these massive discarded almond husks for their second life usage is still scarce and generally involves traditional techniques, such as direct burning or sanitary landfill for biodegradation. Although traditional techniques are convenient, they lead to air pollution. The processing of almond husks for second life usage into fruitful applications via a convenient, economic and efficient approach has significant advantages. We have chosen almond husks as a carbon precursor to improve the cost-effectiveness and also to reduce the environmental burden up to some extent. This natural bio-waste can be utilized as a “green, low cost and efficient precursor” for the gram scale synthesis of shape variant carbon nanostructures. Although, there are a few studies available on the carbonization of bio-waste in an inert atmosphere,14,43,47,52,53 temperature dependent size and shape variant synthesis of carbon nanostructures utilizing a single precursor is still not known.
The synthesis of shape variant photoluminescent carbon nanostructures via changing the carbonization temperature while using the same source of bio-mass is described herein. We have evaluated the effect of carbonization temperature on the structural and morphological behaviour of synthesized wsFCNS having NIR emitting properties. Utilizing almond husk as a naturally occurring “free carbon precursor” to yield shape variant carbon nanostructures could provide an efficient strategy to fabricate nano-carbons on the gram scale. Synthesized wsFCNS are non-toxic in nature since they exhibited less than 2% hemolysis on direct interaction with erythrocytes and hence offer potential for biological applications. The wsFCNS were further utilized for adsorptive removal of PNP, a model dye pollutant and showed excellent dye removal performance. PNP removal of 91% was observed in 1.5 h and the kinetic study of adsorption of PNP exhibited pseudo first order kinetics.
2. Experimental
2.1 Materials
Almond husks were obtained from diverse geographical regions of India and manually chopped into small pieces before carbonization. Triton X-100 and ethylenediamine tetra acetate (EDTA) were purchased from Merck, India. Blood samples were taken from a healthy volunteer. p-Nitrophenol was purchased from Sigma Aldrich, India. All the chemicals were high purity grades and used as obtained without any further purification.2.2 Methods
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio. The diluted blood samples were centrifuged at 2400 rpm for 10 min to separate erythrocytes from plasma. The middle layer (supernatant) was washed thrice with PBS via centrifugation at 2400 rpm for 15 min. Different concentrations (10–100 μg mL−1) of water soluble carbon nano rods (wsCNR), water soluble carbon nano particles (wsCNP) and water soluble carbon nano dots (wsCND) were added into suspensions of washed erythrocytes then incubated in a shaking water bath at 37 °C for 1 h. The samples were collected after incubation, via centrifuging at 3000 rpm for 10 min. Percent hemolysis (%H) was determined spectrophotometrically by measuring absorbance at 540 nm using the following equation:60,61
1 ratio. The diluted blood samples were centrifuged at 2400 rpm for 10 min to separate erythrocytes from plasma. The middle layer (supernatant) was washed thrice with PBS via centrifugation at 2400 rpm for 15 min. Different concentrations (10–100 μg mL−1) of water soluble carbon nano rods (wsCNR), water soluble carbon nano particles (wsCNP) and water soluble carbon nano dots (wsCND) were added into suspensions of washed erythrocytes then incubated in a shaking water bath at 37 °C for 1 h. The samples were collected after incubation, via centrifuging at 3000 rpm for 10 min. Percent hemolysis (%H) was determined spectrophotometrically by measuring absorbance at 540 nm using the following equation:60,61here, PBS reagent was used as negative control (0% lysis of erythrocytes) and Triton X-100 (0.2%) was used as positive control (100% lysis of erythrocytes).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 5 min and the supernatant solution was analysed with a UV-Visible spectrometer to determine the concentration of PNP.
000 rpm for 5 min and the supernatant solution was analysed with a UV-Visible spectrometer to determine the concentration of PNP.The percentage removal of PNP was calculated by using the following relation:
The equilibrium adsorption uptake, qe (mg of adsorbate per g of adsorbent) was calculated as:
3. Results and discussion
wsFCNS were effectively synthesized via a simple pyrolytic route using “almond husk” as a carbon rich precursor material. The size and shape of the synthesized carbon nanostructures were found to be dependent on the carbonization temperature. At a lower temperature of carbonization (750 °C), the almond husk transformed to carbon nanorods, 850 °C produces nanoparticles; while a higher carbonization temperature (950 °C) resulted in carbon nanodots. Oxidation of these wiCNS obtained at 3 different carbonization temperatures with conc. HNO3 produces a number of hydrophilic carboxylic acid groups on their surface and hence introduces aqueous solubility and PL properties as well. A schematic representation for the synthetic process of the three different types of carbon nanostructures is illustrated in Scheme 1. Controlled pyrolysis of almond husk resulted in a carbon cluster, this cluster assembled to give a water insoluble carbonaceous material (wiCNS), oxidation with conc. HNO3 introduces water solubility and PL (wsFCNS). Changes in the carbonization temperature of the almond husks resulted in different morphologies of the synthesized wsFCNS. The difference in morphology at different carbonization temperatures is due to the thermal cutting of larger particles into smaller ones with an increase in the carbonization temperature.64 At higher pyrolytic temperatures the carbon content is greater with a progressive reduction in the particle size.65 At higher temperatures above 800 °C the majority of the amorphous carbon or volatile impurities start to decay and cause a weight loss with a fall-off of rod-like structures. At 850 °C, the surface of the obtained wiCNP has more surface defects. With further increase in the carbonization temperature to 950 °C, a larger size CNP breaks down into smaller CND and causes the disappearance of the larger particles. However, the actual impact of the carbonization temperature on the morphological variations of the carbon nanostructures is still a matter of debate and is difficult to explain precisely. | ||
| Scheme 1 Schematic representation for the synthesis of wsFCNS from almond husk at three different carbonization temperatures; (750 °C (wsCNR), 850 °C (wsCNP) and 950 °C (wsCND)). | ||
3.1 Microscopic studies
Visualisation of surface morphology and shape were performed with FESEM. Fig. 1 demonstrated the FESEM images of wsFCNS synthesized at three different (750 °C, 850 °C and 950 °C) carbonization temperatures. FESEM images depicted in Fig. 1 clearly shows the morphological variations in carbon nanostructures having a homogenous distribution of the same morphology for all three wsFCNS. Carbon nanostructures synthesized at a 750 °C carbonization temperature exhibited a rod-like morphology (Fig. 1(a)), at 850 °C nanoparticles (Fig. 1(b)) while at 950 °C carbon nanodots were observed (Fig. 2(c)). wsCNDs have a spherical morphology without accompanying any other morphological impurities of nanostructures. These results confirmed the effect of carbonization temperature on the morphology of the synthesized carbon nanostructures. | ||
| Fig. 1 FESEM images of (a) wsCNR; (b) wsCNP; (c) wsCND; TEM images of (d) wsCNR; (e) wsCNP; (f) wsCND and; corresponding size distribution histogram of (g) wsCNR; (h) wsCNP; (i) wsCND. | ||
 | ||
| Fig. 2 (a) UV-Visible absorption spectrum of aqueous solution of wsCNP; (b) FTIR spectrum of wsCNP at room temperature; (c) Raman spectra and; (d) TGA analysis curves of wsCNP and wiCNP. | ||
The actual microstructural and morphological identities of wsFCNS have been characterized by TEM microscopic studies. TEM images of the aqueous solutions of wsCNR, wsCNP and wsCND are shown in Fig. 1(d)–(f), which reveal the presence of uniform rods and spherical nanostructures, respectively. The particle size distributions of wsFCNS calculated from the corresponding TEM images by counting 100 random particles are shown in Fig. 1(g)–(i). The particle sizes as calculated from the TEM images of wsCNR, wsCNP and wsCND, are in the range of 100–350 nm, 10–60 and 5–20 nm, respectively. The wsCNR prepared at 750 °C have the highest particle size, most of the rods having diameter in the range of 150–250 nm, wsCND prepared at 950 °C have the lowest particle size with a 5–15 nm in diameter, while wsCNP synthesized at 850 °C have a particle size 20–50 nm in diameter. TEM image of wsCND (Fig. 1(f)) confirms their spherical nature, uniform dispersion and narrow size distribution. These results clearly confirm that carbonization temperature has a strong influence on the morphology of synthesized carbon nanostructures.
3.2 UV-Vis/FTIR/Raman spectroscopy studies
Absorption spectra of each of the aqueous solution of wsFCNS consists of a broad band in the UV region in between 250 to 350 nm with a continuous decrease up to 800 nm. Fig. 2(a), S2(a) and S3(a)† show UV-Visible absorption spectra for various carbon nanostructures. These peaks are ascribed to n–π* and π–π* transitions of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and C
O and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, respectively.59 However, the accurate assignment of the peaks is still a matter of debate66 and hence the different peaks observed for different wsFCNS are hard to explain.
C, respectively.59 However, the accurate assignment of the peaks is still a matter of debate66 and hence the different peaks observed for different wsFCNS are hard to explain.
As reported earlier, acid oxidation imparts hydrophilicity to the carbon nanostructures due to the incorporation of surface carboxylic acid groups.14,18,24,25 The high density incorporation of carboxylic acid groups on wsFCNS were analyzed and confirmed by other spectroscopic measurements such as FTIR, zeta potential and Raman spectroscopy.
FTIR spectra confirms the surface functionalization of wsFCNS with carboxylate groups by showing the presence of characteristic –COOH absorption bands as shown in Fig. 2(b), S2(b) and S3(b).† A characteristic sharp peak ∼1720 cm−1 in all the spectra are assigned for –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching of the carboxylic group and a broad band at ∼3450 cm−1 are due to the –O–H stretching of –COOH. The peaks ∼858 cm−1 in wsCNR, ∼945 cm−1 in wsCNP and ∼860 cm−1 in wsCND are assigned to the out of plane bending of –C–H.61 A peak ∼2350 cm−1 in all the spectra arises due to the interruption of atmospheric CO2.62 The peak ∼1600–1650 cm−1 is characteristic for C
O stretching of the carboxylic group and a broad band at ∼3450 cm−1 are due to the –O–H stretching of –COOH. The peaks ∼858 cm−1 in wsCNR, ∼945 cm−1 in wsCNP and ∼860 cm−1 in wsCND are assigned to the out of plane bending of –C–H.61 A peak ∼2350 cm−1 in all the spectra arises due to the interruption of atmospheric CO2.62 The peak ∼1600–1650 cm−1 is characteristic for C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C vibrational modes perceptible in all the spectra. The presence of surface carboxyl groups on wsFCNS can be further confirmed by the negative value of the zeta potential of −30.2, −32.6 and −35.4 mV for wsCNR, wsCNP and wsCND, respectively, in their aqueous solutions. Clearly, carboxylic groups can be ascribed due to the oxidation of carbon nanostructures and termed as “surfacial defects”.
C vibrational modes perceptible in all the spectra. The presence of surface carboxyl groups on wsFCNS can be further confirmed by the negative value of the zeta potential of −30.2, −32.6 and −35.4 mV for wsCNR, wsCNP and wsCND, respectively, in their aqueous solutions. Clearly, carboxylic groups can be ascribed due to the oxidation of carbon nanostructures and termed as “surfacial defects”.
Raman spectroscopy is a powerful tool for the characterization of edge defects, disorder and in plane crystalline size of graphitic carbon.67 Raman spectra of carbon nanostructures displayed two distinct bands in the range of ∼1335–1350 cm−1 (D Band) and ∼1580–1598 cm−1 (G Band) (Table S1†) generic for carbon based nanomaterials.68,69 The intensity of G band (sp2 hybridized carbons) to D band (sp3 hybridized carbons) ratio noticeably decreases after derivatization. Such a decrease in ratio is a diagnostic of the incorporation of structural defects in terms of sp3 hybridized carbon atoms in sp2 hybridized carbon domains after oxidation and confirms the covalent functionalization of wsFCNS with carboxylic acid groups.70,71 The D band become sharper in wsFCNS (red line) in comparison to wiCNS (black line) with a reversal in relative intensity as demonstrated in Fig. 2(c), S2(c) and S3(c).†
The integrated intensity (IG/ID) ratio of G to D band is well known for the quantitative characterization of carbon nanostructures through the computational Lorentzian curve-fitting algorithm and provides an effective tool to measure crystalline order and the in-plane graphite crystal size of nano carbons.67 Increase in the D to G intensity ratio is an indication of a reduction of crystal size and orientation.72,73 Crystallite size (La) of wsFCNS synthesized at different carbonization temperatures before and after oxidative treatment was determined by the general formula proposed by Cançado et al. (eqn (1)) where λl is the laser excitation wavelength.74 It is interesting to note that the carbonization temperature has a significant effect on the crystallinity of the synthesized carbon nanostructures. The crystallite size of wsCNDs synthesized at higher carbonization temperatures is smaller than wsCNR synthesized at lower carbonization temperature. Also, the crystallite size of carbon nanostructures decreased after functionalization as presented in Table S1.†
| +La = (2.4 × 10−10)λl4(ID/IG)−1 | (1) |
3.3 TGA studies
Thermogravimetric analyses confirmed that the as-synthesized carbon nanostructures are thermally more stable than wsFCNS. Analyses of weight loss with temperature are illustrated in Fig. 2(d), S2(d) and S3(d).† The wsCNR synthesized from almond husk at 750 °C carbonization temperature, start losing weight with a steady decrease until 1000 °C and lost 42.9% of its weight while wiCNR, lost 12.9% of its weight (Fig. S2(d)†). wsCNP obtained at 850 °C, lost 37.1% weight while the corresponding wiCNP lost 19.7% of its total weight (Fig. 2(d)). Similar weight loss was also observed for wsCND synthesized at 950 °C, which lost 29.7% of its weight compared to wiCNDs which lost 18.1% of its total weight (Fig. S3(d)†).Weight loss indicated the decarboxylation process of surface carboxylic acid groups or formation of CO2.66 It was observed that an increase in the carbonization temperature of almond husk (i.e. from 750 °C to 950 °C) also had a significant effect on the thermal behavior of the carbon nanostructures. Increase in the carbonization temperature resulted in thermally more stable wsCND than wsCNR obtained at lower carbonization temperature; weight loss in wsCNR is more prominent than in wsCND.
3.4 Photoluminescence (PL) emission–excitation and fluorescence imaging studies
Fundamentally as well as application wise, PL is the most significant properties of carbon nanostructures owing to their future applications in biological imaging, bio-sensing and optoelectronic devices.75 To understand the PL properties of wsCNR, wsCNP and wsCND synthesized at different carbonization temperatures from the same source, their PL emission spectra were compared as a function of excitation wavelength (λex). The wsFCNS synthesized from almond husk at 750 °C, 850 °C and 950 °C carbonization temperatures exhibit strong emission in a wide range from the visible to NIR region. The PL spectra of wsFCNS with tunable λex from 400–620 nm with 20 nm continuous increments are illustrated in Fig. 3(a)–(c). The PL emission profile of aqueous solutions of wsCNR, wsCNP, and wsCND are very similar without any significant changes in the emission centre. The PL intensity decreases gradually and the emission wavelength (λem) showed a bathochromic shift with an increase in the excitation wavelength. These type of PL profiles i.e. dependence of intensity and emission centred upon λex are characterized as a generic feature of different types of carbon nanostructures.21 At a particular excitation wavelength, λem of all the three samples (wsCNR, wsCNP, and wsCND) are almost invariant indicating that their PL properties are from a common origin owing to their common carbogenic core. Photoluminescent emissions in carbon nanostructures are intimately associated with structural (surfacial) defects and these promote the transitions like band-gap transitions corresponding to conjugated π-domains.21 These surfacial defects present on wsFCNS impart tunable-photoluminescence properties. Hence, all the three samples (wsCNR, wsCNP, and wsCND) show very similar optical properties. This could be due to the same type of surface defect and surface functionality. The maximum emission peaks centered at 546 nm (wsCNR), 548 nm (wsCNP) and 550 nm (wsCND) are achieved on excitation with 400 nm as shown in Fig. 3(a)–(c), respectively. All the wsFCNS exhibit tunable emission behavior in a wide range from the visible to NIR region ranging from ∼542 nm to ∼800 nm with a 20 nm incessant increase of λex from 400 to 620 nm. It is worth noting that the NIR emissions red shifted 765 nm in wsCNR, 775 nm in wsCNP to 790 nm in wsCND. The PL intensity of wsFCNS depends upon their particle size when compared at 400 nm excitation wavelength for the same concentration of 3 solutions. Smaller structures (wsCND) show higher PL intensity as compared to larger structures (wsCNR) (Fig. S4†). This observation is consistent with the earlier reported literature.76 wsFCNS exhibit weak emission intensity in the NIR region which is considered as suitable region for biological imaging of cells and tissues.32,33 The excitation wavelengths in the NIR region are very low and this results in weak emission intensities of wsFCNS. For multi-colored emissions from the same carbon nanoparticle, the exact mechanism is still not clearly understood.The plausible mechanism for the PL behavior of wsFCNS can be explained on the basis of a generic photophysical mechanism with photogeneration of electron–hole pairs associated with quantum-confinement effects.77 The presence of different types of surface energy traps associated with different ‘surface states’ due to functionalization and different particle sizes are responsible for the excitation dependent emission behavior of these carbon nanostructures.14,24,78 Since the energy gap is directly related to the size of nanoparticles in light of the quantum confinement effect. Thus, at lower wavelengths smaller size particles are excited while at higher wavelengths larger size particles are excited. So, the PL intensity depends upon the total number of particles excited at a particular wavelength. Here, we observed a maximum PL intensity at 400 nm, indicating that the maximum number of particles are excited at this wavelength.
The corresponding PL excitation spectra analyzed at 640 nm emission wavelength for wsCNR, wsCNP and wsCND are illustrated in Fig. 3(d)–(f), respectively. All show a PL band ∼470 nm. The photostability tests of wsCNR, wsCNP and wsCND are illustrated in Fig. 3(g)–(i), respectively. The fluorescence intensity in all the cases does not change even after 5 h as is clear from Fig. 3(g)–(i) measured at 460 nm λex and 640 nm λem revealing that wsFCNS have a larger resistance to photo-bleaching. These results are in corroboration with the earlier reported carbon nanoparticles.14,24 Quantum yield was measured by taking quinine sulphate as standard at 460 nm excitation wavelength and found to be ∼1.3% for wsCNR; ∼1.9% for wsCNP and ∼1.6% for wsCND (slightly varying from batch to batch for all carbon nanostructures) and is comparable with earlier reported carbon nanostructures.22,23,44
The full width at half maximum (FWHM) values of wsCNR, wsCNP, and wsCND calculated through the computational Gaussian curve-fitting algorithm of λem at λex of 460 nm are only 68, 65, 51 nm, respectively which are significantly smaller than those early reported carbon nanostructures. These results are in agreement with the histogram (obtained by TEM images) as shown in Fig. 2(g)–(i) and further confirm the narrow size distribution of wsFCNS.77,79 The PL properties of wsCNR, wsCNP, and wsCND are very well associated with their intense fluorescence microscopic images as illustrated in Fig. 4(a)–(f). The samples for optical microscopic imaging were prepared by drop-casting 10 μL aqueous solutions of wsFCNS on a glass slide and then dried under a 100 W table lamp for 1 h. wsFCNS showed strong green and red emissions in the visible region at λex = 488 nm, λex = 535 nm as depicted in Fig. 4(a)–(f).
 | ||
| Fig. 4 Green and red (λex = 488 nm, λex = 535 nm, respectively) emitting fluorescence images of (a), (d) wsCNR; (b), (e) wsCNP and (c), (f) wsCND. | ||
3.5 In vitro hemolysis studies
The % hemolysis increased with an increase in the concentration of wsFCNS for all the wsFCNS due to an increased lysis of the erythrocytes. In vitro hemolysis study of erythrocytes with wsFCNS is shown in Fig. 5. At a smaller concentration (10 μg mL−1) negligible hemolysis occurs and it remained less than 2% up to a concentration of 100 μg mL−1 for all the carbon nanostructures. Such a significantly low value of the % hemolysis confirms the biocompatible (non-toxic) nature of the synthesized wsFCNS and these can be further explored for various bio-applications.3.6. Removal of p-nitrophenol (PNP) studies
BET surface area of wsCNR, wsCNP, and wsCND was found to be 20.6, 265.6, and 1.3 m2 g−1, respectively. Total pore volume of wsCNR, wsCNP, and wsCND was found to be 0.024 cm3 g−1, 0.162 cm3 g−1, and 0.005 cm3 g−1, respectively. At higher temperature above 900 °C, surface areas of carbon nanostructures are known to be decreased as the extent of graphitization increases which is in accordance to the previous reports.64,80 It is clear from the adsorption–desorption curve and pore size distribution curve (Fig. S5, Table S2†) that there is very less number of pores present in wsCND which lead to the decrement in the surface area. wsCNP were chosen for PNP removal due to having a higher surface area. Fig. 6(a) shows the percentage removal of PNP with time for wsCNP. As the time increased, the % PNP removal increased very fast up to 1.5 h and reached a value of ∼91% and after that it attained equilibrium showing ∼92% removal even after 12 h. The absorption spectra of PNP before and after treatment with wsCNP are shown in Fig. 6(b) and corresponding digital images are shown in the inset.The adsorption of PNP molecules from solution to the solid phase can be considered as a reversible process. In this process an equilibrium exists between the solution and solid phase. Non-dissociating molecular adsorption of PNP on the adsorbents can be assumed. Also, no PNP was present initially over the surface of wsCNP, (adsorbent). The uptake of PNP molecules by wsFCNS sample can be fitted according to the pseudo-first-order kinetic model:62,63,81,82
| qt = qe[1 − exp(−kft)] |
PNP adsorption can also be fitted according to the pseudo-second-order kinetic model:62,63,81,82
The sum of square of error (SSE) analysis was used to find the best kinetic model to fit the experimental data. The SSE is given by the following equation:82
Fig. 6(c) and S6† shows that the experimental values of equilibrium uptake qe,exp are closer to qe,cal values in the pseudo first order kinetic model as compared to the pseudo second order kinetic model. The rate constant for the pseudo first order kinetic model was 0.0385 ± 0.0013 min−1.
Initially, a large number of vacant sites was available for the adsorption of PNP molecules over the wsCNP surface. Because of this, fast removal of PNP takes place in the initial 1.5 h. After adsorption, the number of vacant sites decreases and it becomes difficult for further adsorption of PNP molecules due to the repulsive forces between the PNP molecule already adsorbed on the surface of wsCNP and the PNP molecules in the bulk. Because of this, the overall adsorption slows down and equilibrium is achieved.81 Hydrogen bonding plays an important role in the adsorption of PNP molecules over the wsCNP surface. PNP has a lone pair of electrons on the –OH group and also due to –NO2 group, it can form an anion on the oxygen atom of the –OH group which interacts with the wsCNP to form a strong hydrogen bond. It may also be expected that both the –OH and –NO2 groups present in the PNP may interact individually or simultaneously in the adsorption process. PNP removal with pseudo first order kinetics is well supported by the literature.83
4. Conclusion
In summary, we report for the first time a simple, green and economical route to synthesize shape variant photoluminescent carbon nanostructures by varying the carbonization temperature. Carbon nanostructures such as nanorods, nanoparticles and nano dots were synthesized using the same carbon precursor i.e. almond husk, a bio-waste. Furthermore, interestingly these wsFCNS exhibited tunable PL emissions profiles, also covering the NIR region up to some extent. Tunable PL properties are more or less similar for all the different morphologies, indicating temperature-dependent formation of carbon nanostructures with different morphologies from the same parent compound. These wsFCNS are biocompatible in nature and are nontoxic to erythrocytes. The wsFCNS were also used as adsorbents for the removal of PNP and removal up to 91% having pseudo first order adsorption kinetics was obtained. Aqueous solubility along with high photo-stability, of these shape variant carbon nanostructures, could make these materials useful as next generation green nano materials. The proposed synthesis method could be easily scaled up for gram scale synthesis of various carbon nanostructures.Acknowledgements
KMT thanks IITK for providing infrastructure. RKG acknowledges financial assistance from Department of Science and Technology (DST), India INSPIRE Faculty Award (Project No. IFA-13 ENG-57) and Indian Institute of Chemical Engineers (IIChE) (Project No. AKD/R&D/2013/15152). DST support is acknowledged to the Center for Nanosciences.References
- R. H. Baughman, A. A. Zakhidov and W. A. de Heer, Science, 2002, 297, 787–792 CrossRef CAS PubMed.
- Y.-P. Sun, K. Fu, Y. Lin and W. Huang, Acc. Chem. Res., 2002, 35, 1096–1104 CrossRef CAS PubMed.
- C. Aprile, R. Martín, M. Alvaro, J. C. Scaiano and H. Garcia, ChemPhysChem, 2009, 10, 1305–1310 CrossRef CAS PubMed.
- C. N. R. Rao, R. Seshadri, A. Govindaraj and R. Sen, Mater. Sci. Eng., R, 1995, 15, 209–262 CrossRef.
- E. Thune, T. Cabioc’h, M. Jaouen and F. Bodart, Phys. Rev. B, 2003, 68, 115434 CrossRef.
- M. Yamada, T. Akasaka and S. Nagase, Chem. Rev., 2013, 113, 7209–7264 CrossRef CAS PubMed.
- S. Qu, X. Liu, X. Guo, M. Chu, L. Zhang and D. Shen, Adv. Funct. Mater., 2014, 24, 2689–2695 CrossRef CAS.
- A. Begum, K. M. Tripathi and S. Sarkar, Chem.–Eur. J., 2014, 20, 16657–16661 CrossRef CAS PubMed.
- A. Tyagi, K. M. Tripathi and R. K. Gupta, J. Mater. Chem. A, 2015, 3, 22507–22541 CAS.
- H. W. Kroto, J. R. Heath, S. C. O’Brien, R. F. Curl and R. E. Smalley, Nature, 1985, 318, 162–163 CrossRef CAS.
- G. Pastorin, W. Wu, S. Wieckowski, J.-P. Briand, K. Kostarelos, M. Prato and A. Bianco, Chem. Commun., 2006, 1182–1184 RSC.
- E. S. Jeng, A. E. Moll, A. C. Roy, J. B. Gastala and M. S. Strano, Nano Lett., 2006, 6, 371–375 CrossRef CAS PubMed.
- S. K. Sonkar, K. M. Tripathi and S. Sarkar, J. Nanosci. Nanotechnol., 2014, 14, 2532–2538 CrossRef CAS PubMed.
- P. Dubey, K. M. Tripathi and S. K. Sonkar, RSC Adv., 2014, 4, 5838–5844 RSC.
- A. Nieto-Marquez, R. Romero, A. Romero and J. L. Valverde, J. Mater. Chem., 2011, 21, 1664–1672 RSC.
- H. Yu, Q. Zhang, J. B. Joo, N. Li, G. D. Moon, S. Tao, L. Wang and Y. Yin, J. Mater. Chem. A, 2013, 1, 12198–12205 CAS.
- S. K. Cushing, M. Li, F. Huang and N. Wu, ACS Nano, 2014, 8, 1002–1013 CrossRef CAS PubMed.
- D. G. Babar, S. K. Sonkar, K. M. Tripathi and S. Sarkar, J. Nanosci. Nanotechnol., 2014, 14, 2334–2342 CrossRef CAS PubMed.
- G. E. LeCroy, S. K. Sonkar, F. Yang, L. M. Veca, P. Wang, I. Kenneth, N. Tackett, J.-J. Yu, E. Vasile, H. Qian, Y. Liu, P. G. Luo and Y.-P. Sun, ACS Nano, 2014, 8, 4522 CrossRef CAS PubMed.
- J.-e. Park, E. D. Grayfer, Y. Jung, K. Kim, K.-K. Wang, Y.-R. Kim, D. Yoon, H. Cheong, H.-E. Chung, S.-J. Choi, J.-H. Choy and S.-J. Kim, J. Mater. Chem. B, 2013, 1, 1229–1234 RSC.
- P. G. Luo, F. Yang, S.-T. Yang, S. K. Sonkar, L. Yang, J. J. Broglie, Y. Liu and Y.-P. Sun, RSC Adv., 2014, 4, 10791–10807 RSC.
- P. Dubey, K. M. Tripathi, R. Mishra, A. Bhati, A. Singh and S. K. Sonkar, RSC Adv., 2015, 5, 87528–87534 RSC.
- K. M. Tripathi, A. K. Sonker, S. K. Sonkar and S. Sarkar, RSC Adv., 2014, 4, 30100–30107 RSC.
- M. Ghosh, S. K. Sonkar, M. Saxena and S. Sarkar, Small, 2011, 7, 3170–3177 CrossRef CAS PubMed.
- S. K. Sonkar, M. Ghosh, M. Roy, A. Begum and S. Sarkar, Mater. Express, 2012, 2, 105–114 CrossRef CAS.
- S. K. Bhunia, A. Saha, A. R. Maity, S. C. Ray and N. R. Jana, Sci. Rep., 2013, 3, 1473 Search PubMed.
- X. T. Zheng, H. L. He and C. M. Li, RSC Adv., 2013, 3, 24853–24857 RSC.
- S. N. Baker and G. A. Baker, Angew. Chem., Int. Ed., 2010, 49, 6726–6744 CrossRef CAS PubMed.
- P. G. Luo, S. Sahu, S.-T. Yang, S. K. Sonkar, J. Wang, H. Wang, G. E. LeCroy, L. Cao and Y.-P. Sun, J. Mater. Chem. B, 2013, 1, 2116–2127 RSC.
- H. Y. Ko, Y. W. Chang, G. Paramasivam, M. S. Jeong, S. Cho and S. Kim, Chem. Commun., 2013, 49, 10290–10292 RSC.
- B. Yin, J. Deng, X. Peng, Q. Long, J. Zhao, Q. Lu, Q. Chen, H. Li, H. Tang, Y. Zhang and S. Yao, Analyst, 2013, 138, 6551–6557 RSC.
- A. Salinas-Castillo, M. Ariza-Avidad, C. Pritz, M. Camprubi-Robles, B. Fernandez, M. J. Ruedas-Rama, A. Megia-Fernandez, A. Lapresta-Fernandez, F. Santoyo-Gonzalez, A. Schrott-Fischer and L. F. Capitan-Vallvey, Chem. Commun., 2013, 49, 1103–1105 RSC.
- S. Zhu, J. Zhang, S. Tang, C. Qiao, L. Wang, H. Wang, X. Liu, B. Li, Y. Li, W. Yu, X. Wang, H. Sun and B. Yang, Adv. Funct. Mater., 2012, 22, 4732–4740 CrossRef CAS.
- K. Welsher, Z. Liu, D. Daranciang and H. Dai, Nano Lett., 2008, 8, 586–590 CrossRef CAS PubMed.
- L. Wu, M. Luderer, X. Yang, C. Swain, H. Zhang, K. Nelson, A. J. Stacy, B. Shen, G. M. Lanza and D. Pan, Theranostics, 2013, 3, 677–686 CrossRef CAS PubMed.
- A. Albanese, P. S. Tang and W. C. W. Chan, Annu. Rev. Biomed. Eng., 2012, 14, 1–16 CrossRef CAS PubMed.
- D. Feng, Y. Lv, Z. Wu, Y. Dou, L. Han, Z. Sun, Y. Xia, G. Zheng and D. Zhao, J. Am. Chem. Soc., 2011, 133, 15148–15156 CrossRef CAS PubMed.
- H. Orikasa, T. Akahane, M. Okada, Y. Tong, J.-i. Ozaki and T. Kyotani, J. Mater. Chem., 2009, 19, 4615–4621 RSC.
- J. Liu, S. Z. Qiao, H. Liu, J. Chen, A. Orpe, D. Zhao and G. Q. Lu, Angew. Chem., Int. Ed., 2011, 50, 5947–5951 CrossRef CAS PubMed.
- D. Gu, H. Bongard, Y. Deng, D. Feng, Z. Wu, Y. Fang, J. Mao, B. Tu, F. Schüth and D. Zhao, Adv. Mater., 2010, 22, 833–837 CrossRef CAS PubMed.
- M. Li and J. Xue, J. Colloid Interface Sci., 2012, 377, 169–175 CrossRef CAS PubMed.
- Y. Yang, D. Wu, S. Han, P. Hu and R. Liu, Chem. Commun., 2013, 49, 4920–4922 RSC.
- S. K. Sonkar, M. Roy, D. G. Babar and S. Sarkar, Nanoscale, 2012, 4, 7670–7675 RSC.
- K. M. Tripathi, A. K. Sonker, A. Bhati, J. Bhuyan, A. Singh, A. Singh, S. Sarkar and S. K. Sonkar, New J. Chem., 2016, 40, 1571–1579 RSC.
- K. Y. Foo and B. H. Hameed, Bioresour. Technol., 2012, 116, 522–525 CrossRef CAS PubMed.
- S. Sahu, B. Behera, T. K. Maiti and S. Mohapatra, Chem. Commun., 2012, 48, 8835–8837 RSC.
- H. Muramatsu, Y. A. Kim, K.-S. Yang, R. Cruz-Silva, I. Toda, T. Yamada, M. Terrones, M. Endo, T. Hayashi and H. Saitoh, Small, 2014, 10, 2766–2770 CrossRef CAS PubMed.
- J. Wang, C.-F. Wang and S. Chen, Angew. Chem., Int. Ed., 2012, 51, 9297–9301 CrossRef CAS PubMed.
- M. J. Krysmann, A. Kelarakis and E. P. Giannelis, Green Chem., 2012, 14, 3141–3145 RSC.
- W. Lu, X. Qin, S. Liu, G. Chang, Y. Zhang, Y. Luo, A. M. Asiri, A. O. Al-Youbi and X. Sun, Anal. Chem., 2012, 84, 5351–5357 CrossRef CAS PubMed.
- J. Zhou, Z. Sheng, H. Han, M. Zou and C. Li, Mater. Lett., 2012, 66, 222–224 CrossRef CAS.
- S. K. Sonkar, M. Saxena, M. Saha and S. Sarkar, J. Nanosci. Nanotechnol., 2010, 10, 4064–4067 CrossRef CAS PubMed.
- M. Roy, T. Kusurkar, S. Maurya, S. Meena, S. Singh, N. Sethy, K. Bhargava, R. Sharma, D. Goswami, S. Sarkar and M. Das, 3 Biotech, 2014, 4, 67–75 CrossRef.
- M. Ashokkumar, N. T. Narayanan, B. K. Gupta, A. L. M. Reddy, A. P. Singh, S. K. Dhawan, B. Chandrasekaran, D. Rawat, S. Talapatra, P. M. Ajayan and P. Thanikaivelan, ACS Sustainable Chem. Eng., 2013, 1, 619–626 CrossRef CAS.
- S. Chatterjee, E. B. Jones, A. C. Clingenpeel, A. M. McKenna, O. Rios, N. W. McNutt, D. J. Keffer and A. Johs, ACS Sustainable Chem. Eng., 2014, 2, 2002–2010 CrossRef CAS.
- Y.-Q. Li, Y. A. Samad, K. Polychronopoulou, S. M. Alhassan and K. Liao, ACS Sustainable Chem. Eng., 2014, 2, 1492–1497 CrossRef CAS.
- C. Hu, C. Yu, M. Li, X. Fan, J. Yang, P. Zhang, S. Wang, Z. Zhao and J. Qiu, ACS Sustainable Chem. Eng., 2014, 2, 14–18 CrossRef CAS.
- H. Hasar and Y. Cuci, Environ. Technol., 2000, 21, 1337–1342 CrossRef CAS.
- P. Dubey, S. K. Sonkar, S. Majumder, K. M. Tripathi and S. Sarkar, RSC Adv., 2013, 3, 7306–7312 RSC.
- D. Fischer, Y. Li, B. Ahlemeyer, J. Krieglstein and T. Kissel, Biomaterials, 2003, 24, 1121–1131 CrossRef CAS PubMed.
- M. Ashfaq, S. Khan and N. Verma, Biochem. Eng. J., 2014, 90, 79–89 CrossRef CAS.
- J. P. Kushwaha, V. C. Srivastava and I. D. Mall, Bioresour. Technol., 2010, 101, 3474–3483 CrossRef CAS PubMed.
- D. Rameshraja, V. C. Srivastava, J. P. Kushwaha and I. D. Mall, Chem. Eng. J., 2012, 181–182, 343–351 CrossRef CAS.
- N. K. Chaudhari, M. Y. Song and J.-S. Yu, Sci. Rep., 2014, 4, 5221 CAS.
- D. Lopez, I. Y. Abe and I. Pereyra, Diamond Relat. Mater., 2015, 52, 59–65 CrossRef CAS.
- M. P. Sk, A. Jaiswal, A. Paul, S. S. Ghosh and A. Chattopadhyay, Sci. Rep., 2012, 2, 383 Search PubMed.
- M. A. Pimenta, G. Dresselhaus, M. S. Dresselhaus, L. G. Cancado, A. Jorio and R. Saito, Phys. Chem. Chem. Phys., 2007, 9, 1276–1290 RSC.
- K. M. Tripathi, A. Begum, S. K. Sonkar and S. Sarkar, New J. Chem., 2013, 37, 2708–2715 RSC.
- C. T. Cioffi, A. Palkar, F. Melin, A. Kumbhar, L. Echegoyen, M. Melle-Franco, F. Zerbetto, G. M. A. Rahman, C. Ehli, V. Sgobba, D. M. Guldi and M. Prato, Chem.–Eur. J., 2009, 15, 4419–4427 CrossRef CAS PubMed.
- H. Li, X. He, Z. Kang, H. Huang, Y. Liu, J. Liu, S. Lian, C. H. A. Tsang, X. Yang and S.-T. Lee, Angew. Chem., Int. Ed., 2010, 49, 4430–4434 CrossRef CAS PubMed.
- P. Dubey, D. Muthukumaran, S. Dash, R. Mukhopadhayay and S. Sarkar, Pramana J. Phys., 2005, 65, 681–697 CrossRef CAS.
- E. D. Obraztsova, M. Fujii, S. Hayashi, V. L. Kuznetsov, Y. V. Butenko and A. L. Chuvilin, Carbon, 1998, 36, 821–826 CrossRef CAS.
- L. Zhou, C. Gao, D. Zhu, W. Xu, F. F. Chen, A. Palkar, L. Echegoyen and E. S.-W. Kong, Chem.–Eur. J., 2009, 15, 1389–1396 CrossRef CAS PubMed.
- L. G. Cançado, K. Takai, T. Enoki, M. Endo, Y. A. Kim, H. Mizusaki, A. Jorio, L. N. Coelho, R. Magalhães-Paniago and M. A. Pimenta, Appl. Phys. Lett., 2006, 88, 163106 CrossRef.
- H. Li, Z. Kang, Y. Liu and S.-T. Lee, J. Mater. Chem., 2012, 22, 24230–24253 RSC.
- S. C. Ray, A. Saha, N. R. Jana and R. Sarkar, J. Phys. Chem. C, 2009, 113, 18546–18551 CAS.
- J. Deng, Q. Lu, N. Mi, H. Li, M. Liu, M. Xu, L. Tan, Q. Xie, Y. Zhang and S. Yao, Chem.–Eur. J., 2014, 20, 4993–4999 CrossRef CAS PubMed.
- B. De and N. Karak, RSC Adv., 2013, 3, 8286–8290 RSC.
- Z.-A. Qiao, Y. Wang, Y. Gao, H. Li, T. Dai, Y. Liu and Q. Huo, Chem. Commun., 2010, 46, 8812–8814 RSC.
- Y. Xia, Z. Yang and R. Mokaya, Chem. Mater., 2006, 18, 140–148 CrossRef CAS.
- A. Srivastav and V. C. Srivastava, J. Hazard. Mater., 2009, 170, 1133–1140 CrossRef CAS PubMed.
- S. Kumar, V. C. Srivastava and R. P. Badoni, Fuel, 2011, 90, 3209–3216 CrossRef CAS.
- B. K. Singh and N. S. Rawat, J. Chem. Technol. Biotechnol., 1994, 61, 57–65 CrossRef CAS.
Footnotes |
| † Electronic supplementary information (ESI) available: Characterization techniques, solubility images of wsFCNS, UV-Vis, FTIR, Raman and TGA spectra of wsCNP and wsCND, comparison of PL intensity, pseudo second order model, crystalline size calculation, and calibration curve. See DOI: 10.1039/c5ra27432j |
| ‡ Contributed equally. |
| This journal is © The Royal Society of Chemistry 2016 |