Dye aggregation in layer-by-layer dyeing of cotton fabrics
Farzana Khan,
Panpan Liu,
Fujun Xu,
Ying Ma* and
Yiping Qiu*
Key Lab of Fabrics Science and Technology of Ministry of Education, College of Textiles, Donghua University, Shanghai 201620, P. R. China. E-mail: yingma@dhu.edu.cn; Tel: +86 21 67792763
First published on 12th February 2016
Abstract
This work presents the dye aggregation behaviors in layer-by-layer (LbL) dyed cotton fabrics. Fluorescein isothiocyanate labeled poly(allylamine hydrochloride) (PAH-FITC) was LbL-assembled on cotton with poly(acrylic acid) (PAA) or alone. The obtained (PAA/PAH-FITC)*n and (PAH-FITC)*n films demonstrated that the LbL-assembly technique is suitable for homogenously dyeing the complex fabrics. Importantly, different aggregation behaviors of dye FITC were exhibited in the two matrixes of (PAA/PAH-FITC)*n and (PAH-FITC)*n, showing the significant role of LbL-assembly in textile dyeing. Moreover, for the deposition of PAH-FITC, the LbL assembly of PAH-FITC is superior to the continuous immersion of cotton fabric in PAH-FITC solution in terms of the deposition amount, presenting a unique advantage of LbL-assembly in textile dyeing.
Introduction
With the improvement of technology and human-centered service, high quality textiles have received more and more interest due to their beautiful visual effects and desirable specific functions. Among various fabric materials, cotton, as a widely used natural fabric material, has occupied over 50% of the market share globally. The cotton fabrics consisting of cellulose fibers possess abundant hydroxyl groups (–OH), which could easily bond to other functional groups.1–3 So far, considerable efforts have been devoted to the functionalization of cotton fabrics, such as the hydrophobicity,4 anti-microbial activity,5 UV protection,6 flame retardancy,7 and even wearable computing functions.8 Textile dyeing, which refers to colored organic compounds that could be absorbed onto surfaces of textile substrates and then diffused into these substrates, is one of the most frequently used functionalization processes to enhance visual effects in textile industry.9,10Layer-by-layer (LbL) assembly, since it was optimized by G. Decher in 1990s, has attracted considerable attention due to its characteristics of easy processing and versatility in the preparation of functional materials.11–13 The versatility of LbL-assembly are embodied by: (i) various building blocks, including polyelectrolytes,14 macromolecules,15 nanoparticles,16 metal oxide precursors17 and so on; (ii) various driving forces, such as electrostatic interaction,18 hydrogen bonding,19 charge transfer interaction,20 metal ion coordination,21 covalent bonding,22 molecular recognition,23 and hydrophobic interaction;24 (iii) various substrates with different materials and shapes.25 On the extremely rough and soft fabrics, LbL-assembly has been well applied without major changes in weight, texture, breathability of fabrics.26 As published by Sun and co-workers, the LbL-assembled coating have been used in the flame retardancy and self-healing superhydrophobility, providing a practical method to enhance the washing durability of the flame-retardant fabrics.27 Recently, we utilized the LbL-assembly technique to endow fabrics with high-efficiency EMI shielding and fast in-plan thermal dissipation.28
Although the LbL-assembly technique has shown promising in the textile industry, little attention has been paid to the application in textile dyeing. Herein, we LbL-assembled the fluorescein isothiocyanate labeled poly(allylamine hydrochloride) (PAH-FITC) with poly(acrylic acid) (PAA) to dye the cotton fabrics and to study the aggregation behaviors of dyes in LbL-assembled polymer matrixes. For comparison, PAH-FITC was alone deposited on cotton by the LbL process as well. The obtained (PAA/PAH-FITC)*n and (PAH-FITC)*n films on cotton were characterized by SEM, UV-vis, fluorescence and IR spectroscopy. Interestingly, in the two LbL-assembled architectures, the dye of FITC exhibits different aggregation behaviors, which is of significance in textile dyeing. Moreover, for the deposition of PAH-FITC, the LbL assembly of PAH-FITC is superior to a continuous immersion of cotton fabric in PAH-FITC solution in the deposition amount, presenting the unique advantage of LbL-assembly on the textile dyeing.
Experimental section
Materials
100% cotton plain weave fabric (72 ends × 60 picks) has a density of 80 g m−2. 3-Chloro-2-hydroxypropyltrimethylammonium chloride (CHP3MAC) with a concentration of 65% (w/v) in water was purchased from Tokyo Chemical Industry Co. Ltd. Fluorescein isothiocyanate (FITC), poly(allylamine hydrochloride) (PAH, Mw ∼ 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) and poly(acrylic acid) (PAA, Mw ∼ 1800) were from Sigma-Aldrich. Anionic direct dye (Sirius Turquoise) was purchased from DyStar Co. Ltd. Dimethyl sulfoxide (DMSO) was procured from Shanghai Lingfeng Chemical Reagent Co. Ltd. Sodium bicarbonate, sodium carbonate was purchased from Shanghai Titanchem Co. Ltd. Dialysis tube with a 7000 molecular weight cut off was from Shanghai Yuan Ye Shengwu. Deionized water was used in all experiments. And all chemicals were used as received without further purification.
000) and poly(acrylic acid) (PAA, Mw ∼ 1800) were from Sigma-Aldrich. Anionic direct dye (Sirius Turquoise) was purchased from DyStar Co. Ltd. Dimethyl sulfoxide (DMSO) was procured from Shanghai Lingfeng Chemical Reagent Co. Ltd. Sodium bicarbonate, sodium carbonate was purchased from Shanghai Titanchem Co. Ltd. Dialysis tube with a 7000 molecular weight cut off was from Shanghai Yuan Ye Shengwu. Deionized water was used in all experiments. And all chemicals were used as received without further purification.
Polyelectrolyte functionalization: fluorescein isothiocyanate labeled poly(allylamine hydrochloride) (PAH-FITC)
FITC labeled PAH (PAH-FITC) was prepared by a protein labeling technique with slight modification.29 Firstly, 25 mL PAH (4 mg mL−1) was dissolved in 500 mL sodium carbonate buffer with a pH of 9.0 at which FITC would present a dianionic form and yellowish-green dye. And then, 10 mg of FITC was added into 10 mL of DMSO solvent, then ultra-sonicating for 10 min. Afterwards, these two solutions were mixed together and gently stirred for 6 h. The prepared PAH-FITC solution (1% labeling degree) was purified by Millipore water with a dialysis membrane in darkness for 48 h to be assembled.Pre-treatment of cotton fabrics
Cotton fabric was washed by ethanol and DI water to remove the dirt and dust, using as a substrate of polyelectrolyte films. Cotton fabric was first treated to obtain enough charges on its surface: 50 g L−1 CHP3MAC and 18 g L−1 sodium hydroxide (NaOH) were mixed to produce 2,3-epoxypropyltrimethylammonium chloride (EP3MAC) by a ring-opening polymerization, which could connect with cotton fabrics to provide many positive charges.30 As shown in Scheme 1, EP3MAC reacted with the hydroxyl groups of cellulose fibers to produce enough positive charges on cotton for the films to be deposited. After the immersion in EP3MAC solution for 30 min, the fabrics were uniformly coated, then rinsed with Mill-Q water and dried at ambient conditions.LbL dyeing of cotton fabrics
Fluorescein isothiocyanate labeled poly(allylamine hydrochloride) (PAH-FITC) was LbL assembled with poly(acrylic acid) (PAA) on cotton fabrics, as depicted in Scheme 2. Firstly, the cationized cotton fabric after pre-treating was immersed in a PAA solution (1 mg mL−1, pH = 3.5) for 15 min and then rinsed 3 times in a distilled water bath for 1 min. The negatively charged polyelectrolyte of PAA was adsorbed onto the positively charged cotton through the electrostatic interaction. Sequentially, the fabric was then immersed in the PAH-FITC solution (1 mg mL−1, pH = 9.0) for 15 min for depositing cationic PAH-FITC on cotton. The deposition of PAA and PAH-FITC could be repeated until the desired film is obtained, denoted as (PAA/PAH-FITC)*n, where n is the number of deposition cycles. Furthermore, the (PAH-FITC)*n films were also deposited on cotton through alternately immersing in the PAH-FITC solution (1 mg mL−1, pH = 9.0) for 15 min and the water bath for 1 min, 3 times. All the deposition and rinsing processes were performed in the incubator at 37 °C and 100 rpm for a uniform coverage. In the end, the obtained samples were dried at 60 °C in an oven. Additionally, the anionic direct dye of Sirius Turquoise (ST) was used for coloring the cotton fabrics. Firstly, the solution of PAH-ST was prepared by mixing the dye of ST (0.2 mg mL−1) with the PAH solution (1 mg mL−1). Then, the cotton were dyed with the (PAA/PAH-ST)*n and (PAH-ST)*n by using the above-mentioned LbL process of PAH-ST and PAA.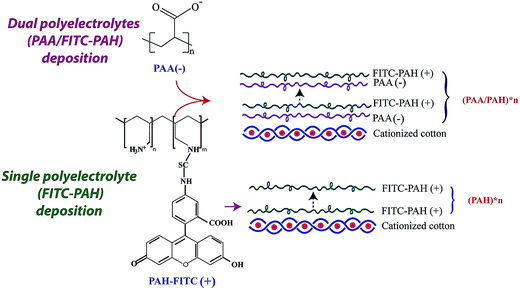 | ||
| Scheme 2 Schematic representation of the LbL dyeing of cotton by (a) (PAA/PAH-FITC)*n and (b) (PAH-FITC)*n. | ||
AATCC standard washing
AATCC test method of 61-2013, as a standard test for colorfastness, was used to characterize the stability of dyed fabrics. The 50 × 100 mm dyed fabrics were washed in a stainless steel canister with an AATCC detergent (WOB) water solution (0.15%, 150 mL) for 20 min at 31 °C.Characterization
The surface morphology of obtained samples was observed by scanning electron microscopy (SEM) (TM 3000, Hitachi, Germany) at an acceleration voltage of 5 kV. A FTIR spectrometer of Nicolet 6700 (Thermo Fisher) was used to analyze the composition of polyelectrolyte films over a range of 600–4000 cm−1. The absorption spectra of the films were determined by a UV-visible-near infra-red (UV-VIS-NIR) spectrophotometer of U-4100 (Hitachi, Japan). Fluorescence spectra were measured on a Quant Master 40 fluorescence spectrometer (Photon Technology International Inc., USA). The fluorescent images were captured using a confocal laser scanning microscope (CLSM, Carl Zeiss LSM 700, Jena, Germany) using 488 nm excitation. Optical micrographs were obtained using an optical microscopy (Nikon, Japan). The K/S values were measured by a spectrophotometer (Datacolor Spectraflash SF600).Results and discussion
Fluorescein isothiocyanate labeled poly(allylamine hydrochloride) (PAH-FITC) exhibited the dye and fluorescence of the dye FITC, as shown in Fig. 1. The absorption characteristic peak of PAH-FITC was shown at the wavelength of 497 nm, which is associated with benzene rings of FITC. Under a UV-light of 365 nm, the orange dye of PAH-FITC solution irradiated the fluorescent yellowish-green dye. It is obvious that the LbL-assembly process of PAH-FITC on fabrics could be monitored by tracing FITC. In Fig. 2, the dyed cotton fabrics with the LbL-assembled films of (PAA/PAH-FITC)*8 and (PAH-FITC)*8 were characterized by optical microscopy, scanning electron microscopy (SEM). From the photos of uncoated (Fig. 2a) and coated (Fig. 2b and c) samples, the (PAA/PAH-FITC)*8 and (PAH-FITC)*8 films were noticed by the dye change of cotton fabrics from white to yellow. The yellow dye was originally from the fluorescent FITC, which was labeled on the building block of PAH. The dye of (PAA/PAH-FITC)*8 cotton was obviously stronger than that of the (PAH-FITC)*8, implying the more deposition of PAH-FITC in the due of polyelectrolytes. The photographs of SEM (Fig. 2e and f) also demonstrates the uniform covering of (PAA/PAH-FITC)*8 and (PAH-FITC)*8 films on fibers. Compared to the (PAH-FITC)*8, the (PAA/PAH-FITC)*8 represents a smoother morphology, which is attributed to the negatively charged PAA that stretches the curly polymer chains of positively charged PAH by the electrostatic interactions between PAA and PAH. The profiles of two films on cotton were shown in Fig. 2h and i of cross-sectional SEM images. Compared with the (PAH-FITC)*8, the (PAA/PAH-FITC)*8 was observed clearly. The result is consistent with that of the optical microscopy. Additionally, the confocal laser scanning microscopy (CLSM) visually presented the fluorescence of (PAA/PAH-FITC)*8 and (PAH-FITC)*8 in Fig. 3. In both of these two images, the dye of bright green appeared along the length of the fibers, showing the almost uniform covering of LbL-assembled films on each fiber. However, unexpectedly, the thick (PAA/PAH-FITC)*8 exhibited a similar fluorescent intensity with (PAH-FITC)*8. | ||
| Fig. 1 (a) UV-vis absorption spectrum of PAH-FITC solution. (b) Photos of PAH-FITC solution under the white light and UV light of 365 nm. | ||
 | ||
| Fig. 3 Confocal laser scanning microscopic images of (a) (PAA/PAH-FITC)*8 and (b) (PAH-FITC)*8 on cotton. | ||
The UV-vis spectra of LbL-assembled (PAA/PAH-FITC)*n and (PAH-FITC)*n films were measured and shown in Fig. 4. With the deposition of PAH-FITC, the absorption signals of (PAA/PAH-FITC)*n films went up and exhibited two characteristic peaks around 503 and 461 nm in Fig. 4a. It is generally known that dye molecules, which possess the conjugated functional groups, could easily form the aggregates and even assembles.31,32 The characteristic peaks at 503 and 461 nm correspond to the monomeric and H-aggregated FITC, respectively.33 Compared to the foreseeable deposition of dual polyelectrolytes (PAA and PAH) on fabrics, the adsorption behavior of single PAH-FITC on fabric was interesting. As shown in Fig. 4b, without the assistance of PAA, the building block of PAH-FITC also could be LbL-deposited on the complex fabric material with a slow increase. On one hand, the abundant hydroxy groups of cotton fibers could interact with the amine groups of PAH and thus introduce the dyes onto fabrics. On the other hand, the growth of (PAH-FITC)*n on cotton was possibly driven by the strong capillary force from the porous fabrics. It can be seen that the LbL deposition could be performed on textiles, both for the dual polyelectrolytes and even single polyelectrolyte. More importantly, the different aggregation behaviors in the two LbL-assembled architectures were noteworthy. The (PAH-FITC)*n films mainly exhibited the characteristic peaks at 503 nm with FITC monomers, showing more H-aggregates of FITC in (PAA/PAH-FITC)*n films than in (PAH-FITC)*n films. It is well known that the driving force of the LbL-assembled (PAA/PAH-FITC)*n films is the electrostatic interaction and hydrogen bonding between the PAA and PAH-FITC.34 The chains of PAH-FITC could be dragged closer to one another by the PAA, contributing to the H-aggregates of FITC at 461 nm induced by π–π* interaction between the benzene rings of FITC. At the beginning of the assembly of (PAA/PAH-FITC)*n films, the H-aggregation of FITC was not obvious due to the restriction of the substrate with the polymeric chains. With the deposition of PAA/PAH-FITC, the characteristic peak of H-aggregated FITC increased rapidly and blue-shifted slightly from 474 to 461 nm. Therefore, we suspect that the interactions between PAA and PAH, that is, the driving force of (PAA/PAH-FITC)*n films, reduced the distances between the groups of FITC, increasing the H-aggregates of FITC. Precisely because of the H-aggregates of FITC without fluorescence, the (PAA/PAH-FITC)*8 with higher UV-vis absorption unexpectedly exhibits a similar fluorescent intensity with (PAH-FITC)*8, as shown in Fig. 3. Considering that the effects of dye aggregates on photophysical properties, it is believed that the controllable aggregation behaviors could play an important role in textile dyeing.
 | ||
| Fig. 4 (a) Evolution of UV-vis absorption spectra of (a) (PAA/PAH-FITC)*n and (b) (PAH-FITC)*n on cotton. | ||
To demonstrate the stability, the cotton fabrics coated with (PAA/PAH-FITC)*8 and (PAH-FITC)*8 films were washed in DI water with shaking for 0.5 and even 24 h. As illustrated in Fig. 5a, after washing, the absorption maxima of the (PAA/PAH-FITC)*8 slightly decreased from original 1.179 to 1.157 and 1.100 units, while that of the (PAH-FITC)*8 were from 1.014 to 0.995 and 0.942 units. Moreover, the (PAA/PAH-FITC)*8 and (PAH-FITC)*8 cotton fabrics endured the AATCC standard washing (test method: 61-2013), which is a standard test for the colorfastness.35 The unclear decrease is from 1.179 to 1.120 units. These results indicated the satisfactory stability of LbL-dyed fabrics. Additionally, for detecting the role of LbL process, we did a parallel experiment on the dyed cotton fabrics prepared by the continuous immersion in the PAH-FITC solution (1 mg mL−1, pH = 9.0). As shown in Fig. 5b, the continuous immersion for 15 and 30 min yielded the adsorption of 0.685 and 0.857 units, respectively. Elongating the immersion time to 6 h and even 12 h, the adsorption of PAH-FITC did not change much but reached a constant value. The adsorption of PAH-FITC in the continuous immersion was less than that in LbL process for the same accumulated immersion times. The reason could be that the rinsing steps removed the excessive polymers adsorbed on the complex surface and thus assisted more dyes to bond with and diffused into fibers. This appears to indicate the advantages of LbL deposition for the construction of functional fabrics.
 | ||
| Fig. 5 Maximum of UV-vis spectra of (a) (PAA/PAH-FITC)*8 and (PAH-FITC)*8 on cotton under different condition, (b) the cottons immersed in PAH-FITC solution for different times. | ||
As shown in Fig. 6, the fluorescence intensities of (PAA/PAH-FITC)*n and (PAH-FITC)*n onto cotton fabrics were characterized by fluorescence spectrometer. Under the excitation of 470 nm, the samples of (PAA/PAH-FITC)*n on cotton (n = 2, 4, 6, 8) emitted the fluorescence with the intensities of 3.32 × 10−6, 3.83 × 10−6, 3.87 × 10−6, 4.29 × 10−6 and 3.44 × 10−6 units, respectively. An obvious reduction in fluorescence termed as quenching, was observed at the (PAA/PAH-FITC)*10, which is induced by excessive fluorescent agents adsorbed on fabric. Moreover, the characteristic peaks were red-shifted with the deposition of the films, attributed to the interactions of PAA with PAH-FITC which decreased the energy of highly polarized π* orbital around fluorescent agents of FITC. In the (PAH-FITC)*n, without the PAA, the fluorescent emission peaks were fixed at 529 nm, and the quenching came early at the 8-cycle due to the higher polarization. It was noted that the increase of fluorescence at the 10-cycle was resulted from the different aggregation degrees of dyes on the complex fabrics. The cotton samples with LbL-assembled (PAA/PAH-FITC)*8 and (PAH-FITC)*8 also were also examined by FTIR spectroscopy in Fig. 7. Compared to the cationized cotton, the (PAA/PAH-FITC)*8 film exhibited several characteristic peaks of the –COOH group at 1702 cm−1, –OH bending vibration at 1031 cm−1, and the N–H bending vibration at 1634 cm−1 and –NH2 deformation mode at 1552 cm−1, demonstrating the adsorption of PAA and PAH on cotton. For the thin (PAH-FITC)*8 film, the peaks at 1057 and 1033 cm−1 declared the existence of PAH on cotton once again.
 | ||
| Fig. 6 Evolution of fluorescent emission spectrum of (a) (PAA/PAH-FITC)*n and (b) (PAH-FITC)*n on cotton. | ||
 | ||
| Fig. 7 FTIR spectra of (a) cotton, (b) (PAH-FITC)*8 on cotton, (c) (PAA/PAH-FITC)*8 on cotton and (d) PAH-FITC solution. | ||
To prove the generality of LbL process, we dyed the cotton fabrics with an anionic direct dye of Sirius Turquoise (ST), as show in Fig. 8. The dye of ST was bonded with the cationic polymer of PAH to produce the PAH-ST by the electrostatic interaction between ST and PAH. Then, the (PAA/PAH-ST)*n and (PAH-ST)*n were prepared on cotton using the LbL process. From the photographs of LbL-dyed samples, the blue color gradually deepened with the deposition of PAH-ST. The value of K/S, which is the relative color strength and color depth of the dyed fabrics, is commonly used to represent the amount of dye fixation on a given fabric. As shown in the curve of K/S values as a function of the number of deposition cycles (Fig. 8a and b), the dyes on fabrics linearly increased for the (PAA/PAH-ST)*n and (PAH-ST)*n systems. The growth rate of (PAA/PAH-ST)*n is higher than that of the (PAH-ST)*n, showing that PAA did play an assistant role in the adsorption of PAH-ST on cotton. This result is consistent with the results from the dyed fabrics with FITC. The dye ST could be identified by its characteristic peaks at 600, 328, 290 and 216 nm in UV-vis spectra of Fig. 8c. These absorption peaks are attributed to the chromophore and unsaturated groups of ST. In Fig. 8d, the similar peaks were found in the cotton fabric coated with LbL-assembled (PAA/PAH-ST)*n and (PAH-ST)*n. For the (PAA/PAH-ST)*n samples, three characteristic peaks were detected at 600, 328 and 216 nm and their adsorption maxima increased with the deposition of (PAA/PAH-ST)*n. The adsorption curves of (PAH-ST)*n samples obviously changed, compared with (PAA/PAH-ST)*n. The disappearance of the peak at 216 nm and the raise of peak at 290 nm could be attributed to the different aggregation behaviors of dyes in two LbL-assembled systems.
Conclusion
In the present research, the cotton fabrics were dyed through the LbL-assembly of PAH-FITC and PAA. As a control, PAH-FITC was alone deposited on cotton by the LbL process as well. The obtained (PAA/PAH-FITC)*n and (PAH-FITC)*n films demonstrated that the LbL-assembly technique is suitable for homogenously dyeing the complex fabrics. Interestingly, in the two LbL-assembled architectures, the dyes exhibit different aggregation behaviors, which is of significance in textile dyeing. Moreover, for the deposition of PAH-FITC, the LbL assembly of PAH-FITC is superior to the continuous immersion of cotton fabric in terms of PAH-FITC solution, presenting the unique advantage of LbL-assembly on the textile dyeing.Acknowledgements
This work was supported by National Natural Science Foundation of China (No. 21304016 and 51303025), Ph.D. Program Foundation of Ministry of Education of China (No. 20130075120004), Shanghai Natural Science Foundation (No. 12ZR1440500), the Fundamental Research Funds for the Central Universities and DHU Distinguished Young Professor Program.References
- B. Hahn, Agr. Hist., 2015, 89, 119–121 CrossRef.
- J. Alongi and G. Malucelli, RSC Adv., 2015, 5, 24239–24263 RSC.
- M. M. G. Fouda, E. S. Abdel-Halim and S. S. Al-Deyab, Carbohydr. Polym., 2013, 92, 943–954 CrossRef CAS PubMed.
- Z. Xu, Y. Zhao, H. Wang, X. Wang and T. Lin, Angew. Chem., Int. Ed., 2015, 54, 4527–4530 CrossRef CAS PubMed.
- X. Hu, M. Tian, L. Qu, S. Zhu and G. Han, Carbon, 2015, 95, 625–633 CrossRef CAS.
- N. A. Ibrahim, M. R. El-Zairy, B. M. Eid, E. M. R. El-Zairy and E. M. Emam, Carbohydr. Polym., 2015, 119, 182–193 CrossRef CAS PubMed.
- M. Zhang, D. Zang, J. Shi, Z. Gao, C. Wang and J. Li, RSC Adv., 2015, 5, 67780–67786 RSC.
- M. Stoppa and A. Chiolerio, Sensors, 2014, 14, 11957–11992 CrossRef CAS PubMed.
- J. Marshall, in The theory of dyeation of textiles, Dyers Company Publication Trust, 1975 Search PubMed.
- R. N. Wijesena, N. D. Tissera and K. M. N. de Silva, Carbohydr. Polym., 2015, 134, 182–189 CrossRef CAS PubMed.
- G. Decher, Science, 1997, 277, 1232–1237 CrossRef CAS.
- X. Zhang, H. Chen and H. Zhang, Chem. Commun., 2007, 1395–1405 RSC.
- J. Zhu, H. Zhang and N. A. Kotov, ACS Nano, 2013, 7, 4818–4829 CrossRef CAS PubMed.
- Q. Yi, D. Wen and G. B. Sukhorukov, Langmuir, 2012, 28, 10822–10829 CrossRef CAS PubMed.
- A. S. Angelatos, B. Radt and F. Caruso, J. Phys. Chem. B, 2005, 109, 3071–3076 CrossRef CAS PubMed.
- E. C. Dreaden, S. W. Morton, K. E. Shopsowitz, J.-H. Choi, Z. J. Deng, N.-J. Cho and P. T. Hammond, ACS Nano, 2014, 8, 8374–8382 CrossRef CAS PubMed.
- X. Shi, S. Han, R. J. Sanedrin, F. Zhou and M. Selke, Chem. Mater., 2002, 14, 1897–1902 CrossRef CAS.
- Y. Wang, A. S. Angelatos and F. Caruso, Chem. Mater., 2007, 20, 848–858 CrossRef.
- C. Sung, A. Vidyasagar, K. Hearn and J. L. Lutkenhaus, Langmuir, 2012, 28, 8100–8109 CrossRef CAS PubMed.
- S. Srivastava, P. Podsiadlo, K. Critchley, J. Zhu, M. Qin, B. S. Shim and N. A. Kotov, Chem. Mater., 2009, 21, 4397–4400 CrossRef CAS.
- C. H. H. Traulsen, E. Darlatt, S. Richter, J. Poppenberg, S. Hoof, W. E. S. Unger and C. A. Schalley, Langmuir, 2012, 28, 10755–10763 CrossRef CAS PubMed.
- C.-J. Huang and F.-C. Chang, Macromolecules, 2009, 42, 5155–5166 CrossRef CAS.
- J.-I. Anzai, Y. Kobayashi, N. Nakamura, M. Nishimura and T. Hoshi, Langmuir, 1998, 15, 221–226 CrossRef.
- J. F. Quinn, A. P. R. Johnston, G. K. Such, A. N. Zelikin and F. Caruso, Chem. Soc. Rev., 2007, 36, 707–718 RSC.
- D. Lee, M. F. Rubner and R. E. Cohen, Nano Lett., 2006, 6, 2305–2312 CrossRef CAS PubMed.
- S. W. Ali, S. Rajendran and M. Joshi, Polym. Polym. Compos., 2010, 18, 175–187 CAS.
- S. Chen, X. Li, Y. Li and J. Sun, ACS Nano, 2015, 9, 4070–4076 CrossRef CAS PubMed.
- L. Zou, S. Zhang, Y. Li, C. Lan, Y. Qiu and Y. Ma, Adv. Mater. Interfaces DOI:10.1002/admi.201500476.
- N. Kato and F. Caruso, J. Phys. Chem. B, 2005, 109, 19604–19612 CrossRef CAS PubMed.
- Q. Wang and P. J. Hauser, Carbohydr. Polym., 2010, 81, 491–496 CrossRef CAS.
- P. Guo, P. Chen, W. Ma and M. Liu, J. Mater. Chem., 2012, 22, 20243–20249 RSC.
- O. Lavinda, I. Mironova, S. Karimi, F. Pozzi, J. Samson, H. Ajiki, L. Massa and K. Ramig, Dyes Pigm., 2013, 96, 581–589 CrossRef CAS.
- H. K. Cho, S. Lone, D. D. Kim, J. H. Choi, S. W. Choi, J. H. Cho, J. H. Kim and I. W. Cheong, Polymer, 2009, 50, 2357–2364 CrossRef CAS.
- S. S. Shiratori and M. F. Rubner, Macromolecules, 2000, 33, 4213–4219 CrossRef CAS.
- Y. Liu, J. Li, X. Cheng, X. Ren and T. S. Huang, J. Mater. Chem. B, 2015, 3, 1446–1454 RSC.
| This journal is © The Royal Society of Chemistry 2016 |



