Synthesis of porous Cu7.2S4 sub-microspheres by an ion exchange method for high-performance supercapacitors
Abstract
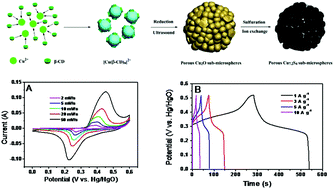
The porous Cu7.2S4 sub-microspheres were synthesized by ion exchange reaction using Cu2O as a precursor. The morphology and structure of the obtained samples were characterized by XRD, SEM, and TEM analysis in detail and the formation mechanism was studied. The pseudo-capacitive properties were evaluated by CV and galvanostatic charge–discharge tests in 6 M KOH solution. The as-prepared porous Cu7.2S4 sub-microspheres electrode has a specific capacitance of 491.5 F g−1 at 1 A g−1 and exhibits good electrochemical performance as an electrode material for supercapacitors.

 Please wait while we load your content...
Please wait while we load your content...