Double-crosslinked network design for self-healing, highly stretchable and resilient polymer hydrogels†
Yinlei Linab,
Deliu Heb,
Zhifeng Chenb,
Liying Wangb and
Guangji Li*b
aSchool of Light Industry and Food Science, South China University of Technology, Guangzhou 510640, China
bSchool of Materials Science and Engineering, South China University of Technology, Guangzhou 510640, China. E-mail: gjli@scut.edu.cn
First published on 20th January 2016
Abstract
Hydrophobic stearyl methacrylate (C18) was captured into sodium dodecyl sulfate (SDS) micelles and self-assembled nanoparticles of an amphiphilic polyurethane macromonomer PU–HEMA were prepared, and they were used as a physical crosslinker and a multifunctional covalent crosslinker, respectively. A reaction system containing acrylamide (AM), the C18 in the micelles and the self-assembled PU–HEMA nanoparticles was designed for constructing chemically and physically double-crosslinked network (CPDN) hydrogels. The prepared CPDN hydrogels have a tensile elongation at break of at least 1400% and can rapidly and fully recover their original shapes in compression experiments, exhibiting superior stretchability and resilience. In addition, the CPDN hydrogels possess a remarkable self-healing behavior and strong self-healing ability even at ambient temperature without the need for any stimulus or healing agent. The novel strategy developed in this study to construct highly stretchable, resilient and self-healing polymer hydrogels is simple yet extremely versatile, thereby opening up a new avenue for the construction of desired functional CPDN hydrogels with excellent mechanical properties.
Introduction
Polymer hydrogels are soft and wet polymeric materials maintaining a three-dimensional network structure and absorbing large amounts of water.1,2 Due to their excellent biocompatibility and other unique properties such as tissue-like elasticity, polymer hydrogels have been widely used as biomaterials in the fields of tissue engineering, gene and drug delivery systems, cell cultures, and wound healing etc.3–5 However, most of the polymer hydrogels prepared in a conventional way are often brittle and exhibit a poor mechanical performance, including low stretchability and a high stress–strain hysteresis, thereby limiting their potential applications as biomaterials.6–10 In the last few decades, much work in the field of hydrogel science has been dedicated to improving the mechanical properties of hydrogels. Great progress has been made in developing novel hydrogels with excellent mechanical properties, such as double-network (DN) hydrogels,11 nanocomposite (NC) hydrogels,12 slide-ring (SR) hydrogels,13 macromolecular microsphere composite (MMC) hydrogels,14 microgel-reinforced hydrogels,1 and so forth. In recent years, some hydrogels showing excellent resilience have been reported.8,15 This is of significance for the practical applications of hydrogels. On the other hand, like classic hydrogels, these novel hydrogels will be permanently damaged on being cut or ruptured. This implies that in spite of their excellent mechanical properties, these hydrogels still lack self-healing ability, i.e. the ability to autonomously repair occurring cracks without (much) intentional human interaction.16It has been recognized that biological materials like skin tissue, bones or tendons have a long lifetime because they can heal damage autonomously and spontaneously, and not because they repeatedly increase their resistance to failure.17,18 Thus, self-healing hydrogels, one of the most important self-healing polymeric materials, have attracted the extensive attention of many scientists over the past decade.19,20 Different approaches or strategies have been explored to obtain self-healing hydrogels. The strategies used to design self-healing hydrogels are mainly based on various forms of dynamically reversible covalent chemistry21,22 and non-covalent interactions, which include metal–ligand interactions,23,24 host–guest interactions,25–27 hydrophobic interactions,28 hydrogen bonding,29 ionic interactions,19,30 etc. Among the various non-covalent interactions, hydrophobic interactions and hydrogen bonding are the common mechanisms used in bulk self-healing materials and play a dominant role in the formation of large biological systems.31 Therefore, it is a logical strategy to apply hydrophobic interactions to construct robust hydrogels with both self-healing behavior and outstanding mechanical properties. In particular, O. Okay and co-workers28 presented a simple strategy to create strong hydrophobic interactions by incorporating hydrophobic chains into hydrophilic polymer network chains for the preparation of tough and self-healing hydrogels. The finite lifetime of hydrophobic associations between stearyl methacrylate (C18) blocks leads to the formation of hydrogels with a high degree of toughness. The hydrogel samples bearing C18 blocks broke at elongation values of 3600% and could provide a self-healing efficiency in terms of the elongation at break of about 100%. However, these hydrogels usually show low tensile strength and resilience.
Recently, J. Zheng and co-workers9 developed a new type of fully physically linked DN hydrogel, for which the double network structure consists of a hydrogen-bond crosslinked network as the first network and strong hydrophobic interactions as the second network. These fully physically linked DN hydrogels exhibit excellent mechanical strength, high toughness and notable self-healing properties. Thus, this is a feasible and useful strategy for constructing DN hydrogels with both outstanding mechanical properties and self-healing behavior and for scientifically designing the double network structures of polymeric hydrogels. The structural diversity and designability of polymers and polymeric networks make it possible to implement the above strategy.
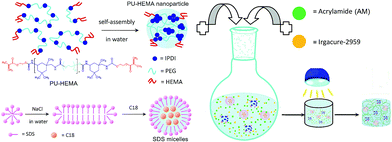
Herein, we present a simple yet extremely versatile strategy based on the construction of a chemically and physically double-crosslinked network (CPDN) for designing and fabricating self-healing, highly stretchable and resilient polymer hydrogels, as schematically illustrated in Scheme 1. An amphiphilic polyurethane macromonomer, PU–HEMA, is synthesized from 3-isocyanatomethyl-3,5,5-trimethylcyclohexyl isocyanate (IPDI), 2-hydroxyethyl methacrylate (HEMA) and poly(ethylene glycol) (PEG). The as-prepared PU–HEMA will self-assemble into nanoparticles in water and can be used as a multifunctional covalent crosslinker. C18 is dissolved in an aqueous NaCl solution containing sodium dodecyl sulfate (SDS) or SDS–NaCl, so as to form multifunctional SDS micelles that can be used as a physical crosslinker. The designed CPDN hydrogels are prepared using UV-induced free radical copolymerization of acrylamide (AM) monomers, PU–HEMA and SDS micelles using Irgacure-2959 as a UV-initiator. In this hydrogel system, both networks, the covalently crosslinked network of polyurethane chains and the physically crosslinked network resulting from strong hydrophobic interactions between the alkyl chains of the C18 units within the SDS micelles, can be formed. Our work proves that the obtained CDPN hydrogels not only exhibit properties of being highly stretchable and resilient, but also possess a unique self-healing behavior. To our knowledge, chemically and physically crosslinked DN hydrogels with self-healing, highly stretchable and resilient properties have not been reported so far.
Experimental
Materials
Acrylamide (AM, 98%), 2-hydroxyethyl methacrylate (HEMA, 97%), sodium dodecyl sulfate (SDS, 99%) and 2-hydroxy-4-(2-hydroxyethoxy)-2-methylpropiophenone (Irgacure-2959, 98%) were purchased from Aldrich and used as received. 3-Isocyanatomethyl-3,5,5-trimethylcyclohexyl isocyanate (IPDI), stearyl methacrylate (C18, 95%), polyethylene glycol (PEG-1k, Mw = 1000), and NaCl (99%) were purchased from Aladdin and used as received. All other reagents and solvents were of analytical grade and used without further purification, unless indicated otherwise.Synthesis of the polyurethane macromonomer PU–HEMA
A polyurethane macromonomer, PU–HEMA, was synthesized according to the procedure described in Scheme S1 (ESI†). PEG-1k (100.0 g, 0.10 mol) was placed in a three-necked flask and dried under vacuum at 90 °C overnight. A catalyst DBTDL (0.3 g) was then added to the flask, followed by stirring for 30 min at 60 °C. Then, IPDI (29.0 g, 0.13 mol) was added dropwise to the flask under mechanical stirring. The reaction mixture was heated to 85 °C and kept at 85 °C for 3 h to obtain an intermediate product, an isocyanate (–NCO) terminated polyurethane prepolymer (PU–NCO). When the reaction mixture had cooled down to 80 °C, HEMA (4.1 g, 0.03 mol) was added and reacted with PU–NCO at 80 °C for 4 h. The expected PU–HEMA was produced. The structures of the intermediate product PU–NCO and the final product PU–HEMA were confirmed using data from their FTIR spectra (ESI, Fig. S1†).Preparation of the hydrogels
The compositions of the designed hydrogels and the control sample PSN containing no PU–HEMA are listed in Table 1. First, the SDS micelles containing C18 and the self-assembled nanoparticles of PU–HEMA were prepared, respectively. SDS and NaCl were dissolved in water at 35 °C to form a transparent SDS–NaCl solution; then C18 was added to the SDS–NaCl solution under stirring at 35 °C, followed by further stirring for about 2 h, and so generating the desired SDS micelle solution. Aqueous solutions of the self-assembled PU–HEMA nanoparticles with different compositions were prepared at ambient temperature by adding portionwise PU–HEMA to a certain amount of deionized water followed by sonicating for 5 min until no precipitates were observed. Next, the SDS micelle solution and the solution of PU–HEMA nanoparticles were mixed together; AM was dissolved in the mixed solution with the aid of sonication to form the reactant solution; and Irgacure-2959 was then added to the reactant solution with stirring in the dark until complete dissolution was achieved. Subsequently, the prepared reactant solution containing Irgacure-2959 was transferred into a cylindrical mould and irradiated with 365 nm UV light in a UV oven at ambient temperature for 1 h to initiate the copolymerization of AM, C18 in the SDS micelles and the PU–HEMA nanoparticles, thereby obtaining the CPDN hydrogel products (see Fig. S2 in ESI†). In the copolymerization system, AM is firstly initiated and polymerized by the water-soluble initiator (Irgacure-2959). When a growing radical moves and encounters a micelle, C18 monomers within the SDS micelle are immediately polymerized and they are incorporated into the main chain of PAM.| Sample | AM [g] | PU–HEMA [g] | SDS [g] | C18 [mg] | NaCl [mg] | H2O [g] | Irgacure-2959 [mg] |
|---|---|---|---|---|---|---|---|
| PSN | 1.3 | 0 | 0.3 | 22 | 23 | 10 | 40 |
| CSN | 1.3 | 0.1 | 0 | 0 | 0 | 10 | 40 |
| CPDN1 | 1.3 | 0.05 | 0.3 | 22 | 23 | 10 | 40 |
| CPDN2 | 1.3 | 0.1 | 0.3 | 22 | 23 | 10 | 40 |
| CPDN3 | 1.3 | 0.2 | 0.3 | 22 | 23 | 10 | 40 |
| CPDN4 | 1.3 | 0.3 | 0.3 | 22 | 23 | 10 | 40 |
For comparison purposes, a physically crosslinked hydrogel sample (PSN) without PU–HEMA nanoparticles and a chemically crosslinked hydrogel sample (CSN) without SDS and C18 were also fabricated as control samples according to the above-described protocol, respectively.
Characterization of the products
1H and 13C NMR spectra were measured at ambient temperature on a Bruker Avance III 400 spectrometer (400 MHz). The samples were dissolved in deuterated chloroform (CDCl3). Fourier-transform infrared (FTIR) spectra of the dry hydrogel samples were recorded on a Bruker VERTEX70 FTIR spectrometer with an ATR accessory for solids. PU–NCO and PU–HEMA samples were drop-coated onto a KBr plate, respectively, and then their FTIR spectra were recorded.The average size of the self-assembled PU–HEMA nanoparticles was measured using dynamic light scattering (DLS) at 25 °C on a Beckman Coulter N5 submicron particle size analyser (Beckman Coulter, Inc., USA). Each sample was measured three times and the average diameter of the nanoparticles was calculated. The morphology of the self-assembled PU–HEMA nanoparticles was observed with transmission electron microscopy (TEM) on a Hitachi High-Technologies H-7650 electron microscope using an acceleration voltage of 80 kV. A specimen suspension (0.04 g mL−1) was prepared at ambient temperature. A drop of the suspension was then placed on a 200-mesh formvar/carbon-coated copper grid and allowed to dry at ambient temperature to prepare the TEM samples.
Measurement of the mechanical properties of the hydrogels
Rod-shaped hydrogel samples (5.5 mm diameter and 60.0 mm length) and cylindrical-shaped hydrogel samples (13.0 mm diameter and 15.0 mm height) were used for measurement of the tensile and compressive mechanical properties, respectively. All the tensile and compression tests were carried out under air at ambient temperature using an electrical universal material testing machine with a 50 N loading cell (Shimadzu AG-X plus). The cross-head speeds for the tensile and compression measurements were 50 mm min−1 and 10 mm min−1, respectively. Cyclic compression tests were performed by performing subsequent trials following the first loading–unloading cycle.The self-healing process and deformation behavior of the prepared hydrogel were recorded using the images obtained with an ordinary camera and the optical microscopy images obtained with a Keyence VHX-2000 optical microscope.
Results and discussion
Structural characterization of the synthesized products
The chemical structure of the synthesized PU–HEMA was characterized using 1H and 13C NMR analyses. Fig. 1 and 2 display the 1H NMR spectrum and the 13C NMR spectrum of PU–HEMA, respectively. As shown in Fig. 1, the peaks around 3.5–3.6 ppm and 0.8–1.0 ppm are assigned to the methylene protons from the PEG-1k units and to the methyl protons on the cyclohexyl rings from the IPDI units, respectively.32–34 Also, a peak at 1.9 ppm attributed to the methyl protons from the segment –C(CH3)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2 in the HEMA units and peaks at 5.5 and 6.1 ppm attributed to the methylene protons from the same segment were observed.35 In the 13C NMR spectrum of PU–HEMA shown in Fig. 2, peaks at 136.0, 125.6 and 18.0 ppm can be seen, which are assigned to the carbons of segment –C(CH3)
CH2 in the HEMA units and peaks at 5.5 and 6.1 ppm attributed to the methylene protons from the same segment were observed.35 In the 13C NMR spectrum of PU–HEMA shown in Fig. 2, peaks at 136.0, 125.6 and 18.0 ppm can be seen, which are assigned to the carbons of segment –C(CH3)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH2 from the HEMA units, respectively.36 The peaks appearing at 156.8 and 155.6 ppm are attributed to the carbons of –NH–COO– in the polyurethane chains.34,37 In addition, a group of peaks between 54.8 and 72.5 ppm, and another group of peaks between 23.2 and 46.9 ppm, which are assigned to the carbons of the segment –CH2CH2–O– from the PEG-1k units38 and to the carbons on the cyclohexyl rings from the IPDI units,37 respectively, were also observed. These analyses indicate that PU–HEMA is synthesized via reaction of PU–NCO and HEMA. The completeness of the reaction between PU–NCO and HEMA can be confirmed through characterization of the functional groups using FTIR spectroscopy (ESI, Fig. S1†).
CH2 from the HEMA units, respectively.36 The peaks appearing at 156.8 and 155.6 ppm are attributed to the carbons of –NH–COO– in the polyurethane chains.34,37 In addition, a group of peaks between 54.8 and 72.5 ppm, and another group of peaks between 23.2 and 46.9 ppm, which are assigned to the carbons of the segment –CH2CH2–O– from the PEG-1k units38 and to the carbons on the cyclohexyl rings from the IPDI units,37 respectively, were also observed. These analyses indicate that PU–HEMA is synthesized via reaction of PU–NCO and HEMA. The completeness of the reaction between PU–NCO and HEMA can be confirmed through characterization of the functional groups using FTIR spectroscopy (ESI, Fig. S1†).
The prepared hydrogels were analyzed using FTIR to characterize their macromolecular structures. Fig. 3 shows the FTIR spectra of the hydrogel sample CPDN2 and the corresponding control sample PSN. From the FTIR spectrum of the PSN sample, characteristic peaks at 2922 cm−1 and 2855 cm−1 due to the stretching of the methylene groups of the C18 units can be clearly observed.28a In the FTIR spectrum of CPDN2 shown in Fig. 3, the above-mentioned peaks at 2922 cm−1 and 2855 cm−1 can be also observed; in addition, characteristic peaks assigned to the –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching and –N–H deformation vibration of the urethane groups from the PU–HEMA chains appear at 1713 cm−1 and 1556 cm−1, respectively.39–41 But both these peaks do not appear in the FTIR spectrum of PSN, a control sample containing no PU–HEMA. This suggests formation of the expected CPDN hydrogels via the UV-induced copolymerization of AM, C18 in the micelles and the self-assembled PU–HEMA nanoparticles.
O stretching and –N–H deformation vibration of the urethane groups from the PU–HEMA chains appear at 1713 cm−1 and 1556 cm−1, respectively.39–41 But both these peaks do not appear in the FTIR spectrum of PSN, a control sample containing no PU–HEMA. This suggests formation of the expected CPDN hydrogels via the UV-induced copolymerization of AM, C18 in the micelles and the self-assembled PU–HEMA nanoparticles.
The microstructure of the self-assembled PU–HEMA nanoparticles in water was characterized using TEM and DLS, respectively. The TEM image in Fig. 4(a) indicates that the self-assembled PU–HEMA nanoparticles were nearly spherical and that their diameter was about 20 nm. According to the size distribution curves shown in Fig. 4(b), the average diameter of the nanoparticles was also about 20 nm at 25 °C, which is consistent with the results observed via TEM.
 | ||
| Fig. 4 (a) TEM image of the self-assembled nanoparticles of PU–HEMA. (b) The size distribution curves of the self-assembled nanoparticles of PU–HEMA. | ||
Mechanical properties of the hydrogels
The as-prepared CPDN hydrogels possess extraordinary mechanical toughness and can withstand different forms of large deformation without any damage or breakage. As shown in Fig. 5, the rod-like sample of the CPDN hydrogel could be stretched to dozens of times its original length and knotted, it can even be stretched after being knotted without any damage; while the cylindrical CPDN hydrogel sample could be compressed significantly and did not break at all even at an extremely high strain. More importantly, the compressed CPDN hydrogel could recover automatically and rapidly to its original shape upon removing the compression stress, exhibiting excellent shape-recovery ability (Movies S1 and S2, ESI†).To further quantitatively evaluate their mechanical properties, especially the ultrahigh toughness, tension tests were conducted on the CPDN hydrogels and the control samples PSN and CSN hydrogels possessing a single crosslinked network structure. The obtained tensile stress–strain curves are shown in Fig. 6. It can be seen from Fig. 6 that the PSN hydrogel exhibits good extensibility, but poor strength (14 kPa) and low resilience. Compared with this, the CPDN hydrogels containing a small amount of PU–HEMA (0.05–0.3 g) can have excellent tensile properties. As the PU–HEMA content in the CPDN hydrogels increases from 0.05 g to 0.3 g, their tensile strength at the same elongation increases, and their elongation at break decreases from 3100% to 1400%. In particular, the CPDN2 hydrogel exhibits greater stretchability than a control sample of the CSN hydrogel with the same PU–HEMA content. These results indicate that the designed double-crosslinked network structure in the hydrogels can significantly improve their stretchability.
Also, cyclic compression tests were performed on the cylindrical CPDN hydrogels to investigate their resilience. Fig. 7 illustrates representative stress–strain curves during cyclic loading–unloading compression tests of the samples. As shown in Fig. 7, for all of the CPDN hydrogels, the compressive stress gradually increases with increasing strain until it exceeds 70% and then it sharply increases; for a given strain, the compression stress that the CPDN hydrogels can withstand increases with increasing PU–HEMA content. No fracturing occurs for the CPDN hydrogels even under a high strain of 80%, exhibiting excellent compression-resistant properties. Furthermore, it can be observed from Fig. 7 that in cyclic compression tests (see Fig. S3 in ESI†) of the CPDN hydrogels, the stress–strain curves recorded during the unloading process are close to those during the loading process, thus producing small stress–strain hysteresis loops; and that the second stress–strain hysteresis loop for each measured CPDN hydrogel sample almost overlaps with its first hysteresis loop. These experimental results prove that the prepared CPDN hydrogels can recover their original shape and state immediately upon removal of the compression stress and show a fully reversible behavior under repeated cyclic compression. This means that the CPDN hydrogels possess obvious elastic deformation features.
The mechanical properties of the CPDN hydrogels depend not only on their compositions and macromolecular structures, but also on their morphological structures. A typical SEM image reflecting the morphology of a freeze-dried CPDN2 hydrogel sample is shown in Fig. 8. It can be clearly seen from the SEM image that the CPDN hydrogel has a porous architecture with pore diameters at the micrometer level. Inside the micropores there are a great many filamentous structures, which may contribute to the further improvement in the mechanical performance of the hydrogels.
Self-healing behavior of the CPDN hydrogels
The as-prepared CPDN hydrogels show not only highly stretchable and resilient properties, but also self-healing behavior at ambient temperature. As shown in Fig. 9(a), two originally rod-shaped hydrogel CPDN2 samples colored with and without a red dye were cut into two parts respectively; then, these four ‘hydrogel parts’ were alternately arranged so as to make the ‘hydrogel parts’ of different colors adjacent and the freshly fractured surfaces of the adjacent parts were brought into contact, thus merging into a single rod-shaped hydrogel sample. After standing for 30 min at ambient temperature, the sample cut marks at the interfaces between two adjacent hydrogel parts disappeared almost completely, and the sample could be bent without breaking, exhibiting its notable self-healing ability. In addition to the rod-shaped hydrogel CPDN2, a corresponding hydrogel film can also exhibit self-healing behavior. Fig. 9(b) illustrates optical microscopy images that reflect the self-healing process of an incision generated by deliberately cutting the CPDN2 film. These observations indicate that the incision on the CPDN2 hydrogel film can autonomously self-heal within 30 minutes at ambient temperature without the need for any stimulus or healing agent. On the other hand, the self-healing efficiency is also an important parameter for characterizing self-healing behavior. When recovery of the elongation at break was used to evaluate the self-healing degree, the elongation at break of the self-healed CPDN hydrogel samples CPDN1, CPDN2, CPDN3 and CPDN4 could recover to about 93%, 86%, 77% and 52% of their original elongation at break after 30 min of healing time, respectively (see Fig. S4 in ESI†). The results show that the self-healing properties of the samples decline with increasing PU–HEMA content. This can be explained by a reduced mobility of the PAM chains at a high density of covalent bonding. The self-healing ability of the CPDN hydrogels can be attributed to their unique macromolecular architecture. In the macromolecular system forming the CPDN hydrogels, there exists double-crosslinked networks, the covalently or chemically crosslinked network and the non-covalently or physically crosslinked network produced by the copolymerization of AM with both the amphiphilic macromonomer PU–HEMA and the polymer chains of C18, respectively. The former can impart excellent mechanical properties; and the latter can impart reversible crosslinking characteristics due to the association of the blocks of C18 in an aqueous environment via hydrophobic interactions. It is the dynamic nature of the junction zones between the polymer chains constituting a reversible physically crosslinked network that can confer self-healing properties. Once cracks or fractures occur in the CPDN hydrogels, self-healing will be triggered via the hydrophobic interactions between the hydrophobic chains. In addition to the reversible hydrophobic association effect, the dynamic hydrogen-bonding interactions between the urethane groups in the macromolecular system forming the CPDN hydrogels can also contribute to their outstanding self-healing properties and even to their excellent mechanical properties.16 | ||
| Fig. 9 Images demonstrating the self-healing of the CPDN 2 hydrogel. (a) Photographs showing the macroscopic self-healing behavior; (b) optical microscopy images of the self-healing process. | ||
Conclusions
In summary, a reaction system containing SDS micelles as a physical crosslinker, an amphiphilic polyurethane macromonomer PU–HEMA as a multifunctional covalent crosslinker and monomer AM was designed for constructing a chemically and physically double-crosslinked network hydrogel, a CPDN hydrogel; and then a group of novel CPDN hydrogels with self-healing, highly stretchable and resilient properties were successfully prepared through the copolymerization of the AM, PU–HEMA and C18 contained in the reaction system. The obtained CPDN hydrogels possess both a chemically crosslinked macromolecular network and a physically crosslinked network based on hydrophobic interactions. The synergistic effects of a chemically and physically double-crosslinked network not only greatly enhance the mechanical properties of the CPDN hydrogels by effectively dissipating energy, but also give them superior stretchable, resilient and self-healing properties via the reversible reconstruction mechanism of the physically crosslinked network. The tensile strength of the CPDN hydrogels containing a small amount of PU–HEMA could be significantly improved. Most importantly, the CPDN hydrogels exhibit rapidly and fully recoverable behavior during compression experiments. In contrast to conventional chemically crosslinked DN hydrogels, the prepared CPDN hydrogels can autonomously repair occurring incisions or cracks at ambient temperature without the need for any stimulus or healing agent. This research has developed a simple yet extremely versatile strategy for designing and preparing highly stretchable, resilient and self-healing polymer hydrogels via the construction of a chemically and physically double-crosslinked network. This strategy opens up a new avenue for the construction of desired functional CPDN hydrogels with excellent mechanical properties, thus promoting an in-depth study and understanding of the fundamental structure–property relationships of CPDN hydrogels.Acknowledgements
Financial support for this work from the Natural Science Foundation of Guangdong Province, China (grant No. 2014A030313246) is gratefully acknowledged. We would also like to express our gratitude to Dr Larry Fowler for his friendly help with revision of the English.Notes and references
- J. Hu, K. Hiwatashi, T. Kurokawa, S. M. Liang, Z. L. Wu and J. P. Gong, Macromolecules, 2011, 44, 7775–7781 CrossRef CAS.
- A. Nakayama, A. Kakugo, J. P. Gong, Y. Osada, M. Takai, T. Erata and S. Kawano, Adv. Funct. Mater., 2004, 14, 1124–1128 CrossRef CAS.
- J. Kopeček and J. Yang, Polym. Int., 2007, 56, 1078–1098 CrossRef.
- L. Yu and J. Ding, Chem. Soc. Rev., 2008, 37, 1473–1481 RSC.
- T. Vermonden, R. Censi and W. E. Hennink, Chem. Rev., 2012, 112, 2853–2888 CrossRef CAS PubMed.
- (a) J. Cui, M. A. Lackey, A. E. Madkour, E. M. Saffer, D. M. Griffin, S. R. Bhatia, A. J. Crosby and G. N. Tew, Biomacromolecules, 2012, 13, 584–588 CrossRef CAS PubMed; (b) E. M. Saffer, M. A. Lackey, D. M. Griffin, S. Kishore, G. N. Tew and S. R. Bhatia, Soft Matter, 2014, 10, 1905–1916 RSC.
- P. Calvert, Adv. Mater., 2009, 21, 743–756 CrossRef CAS.
- C. He, Z. Zheng, D. Zhao, J. Liu, J. Ouyang and H. Wang, Soft Matter, 2013, 9, 2837–2844 RSC.
- Q. Chen, L. Zhu, H. Chen, H. Yan, L. Huang, J. Yang and J. Zheng, Adv. Funct. Mater., 2015, 25, 1598–1607 CrossRef CAS.
- Y. Sun, G. Gao, G. Du, Y. Cheng and J. Fu, ACS Macro Lett., 2014, 3, 496–500 CrossRef CAS.
- J. P. Gong, Y. Katsuyama, T. Kurokawa and Y. Osada, Adv. Mater., 2003, 15, 1155–1158 CrossRef CAS.
- K. Haraguchi and T. Takehisa, Adv. Mater., 2002, 14, 1120–1124 CrossRef CAS.
- Y. Okumura and K. Ito, Adv. Mater., 2001, 13, 485–487 CrossRef CAS.
- T. Huang, H. Xu, K. Jiao, L. Zhu, H. R. Brown and H. Wang, Adv. Mater., 2007, 19, 1622–1626 CrossRef CAS.
- (a) Y. Cui, M. Tan, A. Zhu and M. Guo, RSC Adv., 2014, 4, 56791–56797 RSC; (b) T. Zhao, M. Tan, Y. Cui, C. Deng, H. Huang and M. Guo, Polym. Chem., 2014, 5, 4965–4973 RSC.
- Y. Lin and G. Li, J. Mater. Chem. B, 2014, 2, 6878–6885 RSC.
- S. Billiet, X. K. D. Hillewaere, R. F. A. Teixeira and F. E. Du Prez, Macromol. Rapid Commun., 2013, 34, 290–309 CrossRef CAS.
- V. Amendola and M. Meneghetti, Nanoscale, 2009, 1, 74–88 RSC.
- Q. Wang, J. L. Mynar, M. Yoshida, E. Lee, M. Lee, K. Okuro, K. Kinbara and T. Aida, Nature, 2010, 463, 339–343 CrossRef CAS PubMed.
- Z. Wei, J. H. Yang, J. Zhou, F. Xu, M. Zrínyi, P. H. Dussault, Y. Osada and Y. M. Chen, Chem. Soc. Rev., 2014, 43, 8114–8131 RSC.
- G. Deng, C. Tang, F. Li, H. Jiang and Y. Chen, Macromolecules, 2010, 43, 1191–1194 CrossRef CAS.
- Z. Wei, J. H. Yang, X. J. Du, F. Xu, M. Zrinyi, Y. Osada, F. Li and Y. M. Chen, Macromol. Rapid Commun., 2013, 34, 1464–1470 CrossRef CAS PubMed.
- T. Sato, M. Ebara, S. Tanaka, T. Asoh, A. Kikuchi and T. Aoyagi, Phys. Chem. Chem. Phys., 2013, 15, 10628–10835 RSC.
- J. Yuan, X. Fang, L. Zhang, G. Hong, Y. Lin, Q. Zheng, Y. Xu, Y. Ruan, W. Weng, H. Xia and G. Chen, J. Mater. Chem., 2012, 22, 11515–11522 RSC.
- K. Miyamae, M. Nakahata, Y. Takashima and A. Harada, Angew. Chem., Int. Ed., 2015, 54, 8984–8987 CrossRef CAS PubMed.
- P. Wei, X. Yan and F. Huang, Chem. Soc. Rev., 2015, 44, 815–832 RSC.
- M. Tan, Y. Cui, A. Zhu, H. Han, M. Guo and M. Jiang, Polym. Chem., 2015, 6, 7543–7549 RSC.
- (a) D. C. Tuncaboylu, M. Sari, W. Oppermann and O. Okay, Macromolecules, 2011, 44, 4997–5005 CrossRef CAS; (b) D. C. Tuncaboylu, M. Sahin, A. Argun, W. Oppermann and O. Okay, Macromolecules, 2012, 45, 1991–2000 CrossRef CAS.
- (a) J. Cui and A. del Campo, Chem. Commun., 2012, 48, 9302–9304 RSC; (b) M. M. C. Bastings, S. Koudstaal, R. E. Kieltyka, Y. Nakano, A. C. H. Pape, D. A. M. Feyen, F. J. van Slochteren, P. A. Doevendans, J. P. G. Sluijter, E. W. Meijer, S. A. J. Chamuleau and P. Y. W. Dankers, Adv. Healthcare Mater., 2014, 3, 70–78 CrossRef CAS PubMed; (c) A. Phadke, C. Zhang, B. Arman, C. C. Hsu, R. A. Mashelkar, A. K. Lele, M. J. Tauber, G. Arya and S. Varghese, Proc. Natl. Acad. Sci. U. S. A., 2012, 109, 4383–4388 CrossRef CAS PubMed.
- T. L. Sun, T. Kurokawa, S. Kuroda, A. B. Ihsan, T. Akasaki, K. Sato, M. A. Haque, T. Nakajima and J. P. Gong, Nat. Mater., 2013, 12, 932–937 CrossRef CAS PubMed.
- A. Sánchez-Iglesias, M. Grzelczak, T. Altantzis, B. Goris, J. Pérez-Juste, S. Bals, G. V. Tendeloo, S. H. Donaldson Jr, B. F. Chmelka, J. N. Israelachvili and L. M. Liz-Marzán, ACS Nano, 2012, 6, 11059–11065 CrossRef PubMed.
- J. Zhang, N. Wang, W. Liu, X. Zhao and W. Lu, Soft Matter, 2013, 9, 6331–6337 RSC.
- M. Tan, T. Zhao, H. Huang and M. Guo, Polym. Chem., 2013, 4, 5570–5576 RSC.
- B. Mendoza-Novelo, J. L. Mata-Mata, A. Vega-González, J. V. Cauich-Rodíguez and Á. Marcos-Fernández, J. Mater. Chem. B, 2014, 2, 2874–2882 RSC.
- R. Ferrari, Y. Yu, M. Morbidelli, R. A. Hutchinson and D. Moscatelli, Macromolecules, 2011, 44, 9205–9212 CrossRef CAS.
- B. Kalra, A. Kumar, R. A. Gross, M. Baiardo and M. Scandola, Macromolecules, 2004, 37, 1243–1250 CrossRef CAS.
- L. Torini, J. F. Argillier and N. Zydowicz, Macromolecules, 2005, 38, 3225–3236 CrossRef CAS.
- (a) M. Castano, K. S. Seo, E. H. Kim, M. L. Becker and J. E. Puskas, Macromol. Rapid Commun., 2013, 34, 1375–1380 CrossRef CAS PubMed; (b) X. Zhang, F. Chen, Z. Zhong and R. Zhuo, Macromol. Rapid Commun., 2010, 31, 2155–2159 CrossRef CAS PubMed.
- L. Yao, C. Wu, Z. Yang, W. Qiu, P. Cui and T. Xu, J. Appl. Polym. Sci., 2012, 124, E216–E224 CrossRef CAS.
- Z. Wang, L. Yu, M. Ding, H. Tan, J. Li and Q. Fu, Polym. Chem., 2011, 2, 601–607 RSC.
- H. Sardon, J. P. K. Tan, J. M. W. Chan, D. Mantione, D. Mecerreyes, J. L. Hedrick and Y. Y. Yang, Macromol. Rapid Commun., 2015, 36, 1761–1767 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental and synthesis details. See DOI: 10.1039/c5ra26770f |
| This journal is © The Royal Society of Chemistry 2016 |