Magnetic graphene-based cyanopropyltriethoxysilane as an adsorbent for simultaneous determination of polar and non-polar organophosphorus pesticides in cow’s milk†
Hamid Rashidi Nodeha,
Wan Aini Wan Ibrahim *ab,
Mohd Marsin Sanagiac and
Hassan Y. Aboul-Eneind
*ab,
Mohd Marsin Sanagiac and
Hassan Y. Aboul-Eneind
aDepartment of Chemistry, Faculty of Science, Universiti Teknologi Malaysia, 81310 UTM Johor Bahru, Johor, Malaysia. E-mail: wanaini@kimia.fs.utm.my; waini@utm.my; Fax: +60-7-5566162; Tel: +60-7-5534311
bSeparation Science and Technology Group, Frontier Materials Research Alliance, Universiti Teknologi Malaysia, 81310 UTM Johor Bahru, Johor, Malaysia
cIbnu Sina Institute for Scientific and Industrial Research, Universiti Teknologi Malaysia, 81310 UTM Johor Bahru, Johor, Malaysia
dNational Research Centre, Department of Pharmaceutical and Medicinal Chemistry, Dokki, 12311 Cairo, Egypt
First published on 8th February 2016
Abstract
A new adsorbent based on magnetic nanoparticles (Fe3O4), graphene (G) and cyanopropyltriethoxysilane (CNPrTEOS) was fabricated and applied in the magnetic solid phase extraction (MSPE) of organophosphorus pesticides. The synthesized adsorbent was characterized using Fourier transform infrared spectroscopy, scanning electron microscopy, energy-dispersive X-ray spectroscopy and transmission electron microscopy. The Fe3O4@G–CNPrTEOS MSPE was used for the simultaneous preconcentration of four polar and non-polar organophosphorus pesticides namely phosphamidon, dimethoate, diazinon and chlorpyrifos in fresh cow’s milk. Under optimum conditions, Fe3O4@G–CNPrTEOS MSPE shows good linearity in the concentration range of 0.1–200 ng mL−1, low limits of detection (0.01–0.6 ng mL−1) and high enrichment factors (2400). The precision of the MSPE method was investigated using intraday (% RSD 1.7–9.4%, n = 3) and interday (% RSD 10.7–17.6%, n = 15) studies. The synthesized adsorbent can be reused at least 10 times without significant loss of extraction efficiency. The Fe3O4@G–CNPrTEOS MSPE method offers good recovery (82–94%) for spiked milk samples as compared to commercial CNPr-SPE (42–59%) and C18-SPE (58–68%). The Fe3O4@G–CNPrTEOS MSPE method involves only dilution of the cow’s milk analysed without any treatment prior to analysis, and fewer interferences from protein fractions were observed.
1. Introduction
Organophosphorus pesticides (OPPs) are widely used in agriculture to control insects. They can be transferred to cow’s milk as a result of veterinary treatment against ectoparasite insects, inhalation or ingestion via grass/corn silage feed or the water supply.1–3 OPP residues have been reported in several raw and commercial pasteurized milk samples in Italy, Mexico and Spain.1,3–5 The presence of organochlorine pesticides has also been reported in cow’s milk.6 The presence of OPP residues in milk is a significant risk for human health since milk is widely drank by adults, children and infants.3 The maximum residual level (MRL) for OPPs in milk is between 0.01 mg kg−1 to 0.05 mg kg−1 as set by the European Union (EU) Commission 396/2005 (pesticides database, product milk).7 Due to the low MRL, a sensitive and accurate analytical method is imperative. Sample preparation is the critical stage in an analytical process (i.e., extraction, isolation and preconcentration) prior to instrument analysis.8 Due to the existence of complex matrices in biological samples, selective and cost effective sample preparation techniques are essential.9–11Graphene provides great potential benefits in analytical sample preparation. Graphene is likely to be a suitable sorbent for the removal/adsorption of organic compounds due to its high stability, large surface area, high adsorption capacity, rich sp2 bonding and large delocalized π–π electrons.12–14 An exceptional advantage of graphene is that both sides of the graphene planes are available for adsorption compared to other carbon-based nanomaterials. The hydrophobic properties of graphene also lead to high absorption affinity of non-polar compounds. Graphene has been shown to adsorb pesticides from water, juice, fruit, vegetables and oil.15–23 Graphene was successfully used as a solid phase extraction (SPE) sorbent for phthalate and polycyclic aromatic hydrocarbons extraction from soy bean milk and dairy milk with high recovery and less interference.24,25 In order to increase the graphene selectivity and sensitivity, it was modified using magnetic nanoparticles (MNPs), amine, composite nanoadsorbent, silica, titanium, alumina, polymers, membrane, carbon and carbon nanotubes (CNTs).26–34 Even though graphene-based SPE methods provide great adsorption potential, the technique is tedious, time-consuming and there is a loss of graphene weight.
In 1999, SPE based on MNPs was developed for sample preparation which is called magnetic solid phase extraction (MSPE).35 MSPE is fast, requires small amounts of solvent and is easy to separate the adsorbent from the sample by using an external magnet.31–35 MSPE has been used successfully to preconcentrate analytes from biological samples.33,34 For example, Fe3O4-functionalized C18 was used as an adsorbent for the extraction of aromatic amines from urine,36 and Fe3O4/CNTs have been used to determine estrogen in milk.28 Fe3O4–molecularly imprinted polymer (MIP) has also been used for the adsorption of melamine from milk,32 and Fe3O4@SiO2–MIPs for estrogen preconcentration from human plasma.37 Puerarin was extracted from rat plasma using Fe3O4@SiO2–C18.38
In this study, the combination of MNPs for ease of separation, and graphene’s large surface area for enhanced adsorption, were exploited to produce Fe3O4–graphene (Fe3O4–G) and then further modified with polar cyanopropyltriethoxysilane (CNPrTEOS) to enhance the capability for simultaneous preconcentration of polar and non-polar OPPs. First, Fe3O4@G was synthesized using a one-pot method, followed by reduction using lithium aluminum hydride and sodium borohydride. Then, CNPrTEOS was added onto the Fe3O4@G using a simple sol–gel process to produce Fe3O4@G–CNPrTEOS. The newly synthesized adsorbent was applied for the first time as an MSPE adsorbent for the simultaneous preconcentration of polar and non-polar OPPs from fresh cow’s milk samples.
The logarithm of the octanol–water partition coefficient, log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KO/W, identifies the polarity indices of organic compounds; log
KO/W, identifies the polarity indices of organic compounds; log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KO/W value for polar compounds is <2.0, mid-polar is 2.0–3.0 and non-polar is >3.0.39 The log
KO/W value for polar compounds is <2.0, mid-polar is 2.0–3.0 and non-polar is >3.0.39 The log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KO/W values of the OPPs studied are shown in Table 1.39 Both polar and non-polar OPPs were successfully extracted simultaneously using the newly synthesized Fe3O4@G–CNPrTEOS due to the presence of both hydrophilic (–C
KO/W values of the OPPs studied are shown in Table 1.39 Both polar and non-polar OPPs were successfully extracted simultaneously using the newly synthesized Fe3O4@G–CNPrTEOS due to the presence of both hydrophilic (–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N) and hydrophobic (graphene) properties. The proposed Fe3O4@G–CNPrTEOS MSPE method shows fast extraction, high recovery and high enrichment factors for the selected OPPs. Previous studies have reported that the fat and protein matrix in milk samples should be treated with acid in order to remove its effects.40,41 However, in our study, we found that the as prepared Fe3O4@G–CNPrTEOS can be used directly to analyse fresh cow’s milk using simple dilution with deionized water without further sample preparation.
N) and hydrophobic (graphene) properties. The proposed Fe3O4@G–CNPrTEOS MSPE method shows fast extraction, high recovery and high enrichment factors for the selected OPPs. Previous studies have reported that the fat and protein matrix in milk samples should be treated with acid in order to remove its effects.40,41 However, in our study, we found that the as prepared Fe3O4@G–CNPrTEOS can be used directly to analyse fresh cow’s milk using simple dilution with deionized water without further sample preparation.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KO/W values and MRLs
KO/W values and MRLs
| OPPs name | Structure | log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KO/W39 KO/W39 |
MRLs in milk (mg kg−1) |
|---|---|---|---|
| Phosphamidon | 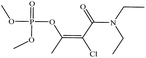 |
0.3 | 0.05 |
| Dimethoate | 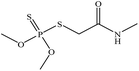 |
0.7 | 0.05 |
| Diazinon |  |
3.8 | 0.02 |
| Chlorpyrifos | 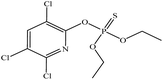 |
4.7 | 0.01 |
| Hexaconazole (internal standard) | 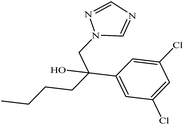 |
3.6 | 0.02 |
2. Experimental
2.1 Materials
AR-grade acetone, HPLC grade methanol, ethanol (97%), sulfuric acid (98%) and nitric acid (65%) were obtained from QReC (Selangor, Malaysia). Acetonitrile, hydrogen peroxide, potassium permanganate and ammonia (32%) were from Merck (Schuchardt, Germany). Graphite powder and CNPrTEOS were obtained from Sigma Aldrich (St. Louis, MO, USA). Chlorpyrifos, hexaconazole, lithium aluminum hydride (LiAlH4) and sodium borohydride (NaBH4) were from Riedel-de Haen (Sleeze, Germany). Pestanal grade diazinon, dimethoate and phosphamidon, iron(III) chloride hexahydrate (FeCl3·6H2O) and ammonium iron(II) sulfate hexahydrate ((NH4)2Fe(SO4)2·6H2O) were from Fluka (Hanover, Germany). Appropriate amounts of the selected OPPs (Table 1) were weighed and dissolved in 10 mL HPLC-grade methanol to produce a stock solution of 1000 mg L−1. This was kept in a refrigerator until use. Commercial CNPr-SPE and C18-SPE cartridges (100 mg) were obtained from Supelco (PA, USA).Fresh cow’s milk samples were obtained directly from the transfer truck tank from Johor Bahru, Malaysia, and diluted with deionized water prior to use.
2.2 Gas chromatography conditions
The four selected OPPs were determined using gas chromatography with micro-electron capture detector (GC-μECD). The column used was HP-5MS (30 m × 0.32 mm i.d. and 0.25 μm film thickness). Before operating the GC-μECD for OPPs analysis, oven temperature, temperature rate, inlet temperature, inlet gas flow rate, detector temperature and make up gas (nitrogen) volume were optimized. The optimum conditions obtained were as follows: nitrogen gas as back inlet carrier gas at a flow rate of 1 mL min−1 and also back detector make-up gas (5 mL min−1). The temperature was set at 280 °C and 300 °C for back inlet and back detector, respectively. The initial oven temperature was set at 70 °C (0 min) and it was then increased at a rate of 50 °C min−1 to 180 °C (held for 1 min) and ramped at 30 °C min−1 to 280 °C (held for 1 min).2.3 Instruments
Deionized water was obtained from a Milli-Q system from Millipore (Bedford, MA, USA). The four OPPs were analyzed using an Agilent 7600A GC-μECD from Agilent Technologies Inc. (Santa Clara, CA, USA). A 1600 series Perkin-Elmer Spectrophotometer (MA, USA) was used for FTIR spectra recording in transmission mode in the range of 400–4000 cm−1. The scanning electron microscope equipped with energy-dispersive X-ray spectroscopy (SEM/EDX) model JSM6390 from JEOL (Tokyo, Japan) was used for morphology and composition studies of the synthesized adsorbent. The transmission electron microscope (TEM) images were obtained using a JEM-2100 electron microscope from JEOL Ltd. (Tokyo, Japan) operated at 200 kV.2.4 Synthesis of magnetic graphene-based cyanopropyltriethoxysilane
Natural graphite (1 g) was dispersed in 50 mL concentrated HNO3/H2SO4 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 v/v) mixture followed by slow addition of 3 g of KMnO4 with stirring for 24 h to give a brown product. Then the mixture was poured into 200 g of ice followed by addition of 5 mL of H2O2 and a yellow product (graphene oxide, GO) was formed. To obtain graphene from GO, LiAlH4 (0.5 g) and NaBH4 (0.5 g) were applied as reducing agents followed by heating at 90 °C and stirring for 5 h. A dark color product (graphene) was obtained and was diluted with deionized water (400 mL), centrifuged and oven dried at 100 °C for 24 h. Previously hydrazine was used as a reducing agent, although it exhibited high efficiency it is highly toxic and hazardous to the environment. The graphene obtained was dispersed in 30 mL deionized water and sonicated for 30 min, then it was added to FeCl3·6H2O (0.05 g) and (NH4)2Fe(SO4)2·6H2O (0.1 g). The mixture was degassed by bubbling nitrogen for 20 min and then 5 mL of ammonia solution (25%) was added slowly into it. The solution was stirred for 2 h. The dark product (Fe3O4@G) was washed with 500 mL deionized water and collected with the assistance of an external magnet.
3 v/v) mixture followed by slow addition of 3 g of KMnO4 with stirring for 24 h to give a brown product. Then the mixture was poured into 200 g of ice followed by addition of 5 mL of H2O2 and a yellow product (graphene oxide, GO) was formed. To obtain graphene from GO, LiAlH4 (0.5 g) and NaBH4 (0.5 g) were applied as reducing agents followed by heating at 90 °C and stirring for 5 h. A dark color product (graphene) was obtained and was diluted with deionized water (400 mL), centrifuged and oven dried at 100 °C for 24 h. Previously hydrazine was used as a reducing agent, although it exhibited high efficiency it is highly toxic and hazardous to the environment. The graphene obtained was dispersed in 30 mL deionized water and sonicated for 30 min, then it was added to FeCl3·6H2O (0.05 g) and (NH4)2Fe(SO4)2·6H2O (0.1 g). The mixture was degassed by bubbling nitrogen for 20 min and then 5 mL of ammonia solution (25%) was added slowly into it. The solution was stirred for 2 h. The dark product (Fe3O4@G) was washed with 500 mL deionized water and collected with the assistance of an external magnet.
The sol–gel process was then used to functionalize the Fe3O4@G with cyano groups to produce Fe3O4@G–CNPrTEOS. About 0.5 g of the Fe3O4@G was dispersed in 50 mL of water![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) methanol mixture (1
methanol mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 v/v) and sonicated for 30 min. Then, 1.5 mL of ammonia solution (32%) was added followed by the addition of 0.5 mL of CNPrTEOS with continuous stirring for 30 min. The mixture was left at room temperature until it formed a gel. The product was washed sequentially with methanol (20 mL), acetone (50 mL) and water (100 mL) before final drying at 100 °C for 24 h. The schematic of Fe3O4@G–CNPrTEOS synthesis is shown in Fig. 1.
5 v/v) and sonicated for 30 min. Then, 1.5 mL of ammonia solution (32%) was added followed by the addition of 0.5 mL of CNPrTEOS with continuous stirring for 30 min. The mixture was left at room temperature until it formed a gel. The product was washed sequentially with methanol (20 mL), acetone (50 mL) and water (100 mL) before final drying at 100 °C for 24 h. The schematic of Fe3O4@G–CNPrTEOS synthesis is shown in Fig. 1.
2.5 GC-μECD instrument calibration
The instrument calibration was performed using the internal standard (hexaconazole 100 ng mL−1) method using five point calibration for each OPP standard. The concentration range for standard chlorpyrifos and diazinon were from 10 to 500 ng mL−1 and for standard phosphamidon and dimethoate from 200 to 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ng mL−1 (see ESI 1†). Injections were performed in triplicate (n = 3) for each calibration point. Calibration curves were obtained by plotting (AS/AIS) versus (CS/CIS), where AS is the peak area of standard, AIS is the peak area of internal standard (IS), CS is the standard OPPs concentration, and CIS is the concentration of the IS. The instrument limit of detection (LOD) and limit of quantification (LOQ) were obtained from (3 × SD)/m and (10 × SD)/m, respectively,42 where SD is the standard deviation of the lowest standard concentration of OPPs.43
000 ng mL−1 (see ESI 1†). Injections were performed in triplicate (n = 3) for each calibration point. Calibration curves were obtained by plotting (AS/AIS) versus (CS/CIS), where AS is the peak area of standard, AIS is the peak area of internal standard (IS), CS is the standard OPPs concentration, and CIS is the concentration of the IS. The instrument limit of detection (LOD) and limit of quantification (LOQ) were obtained from (3 × SD)/m and (10 × SD)/m, respectively,42 where SD is the standard deviation of the lowest standard concentration of OPPs.43
2.6 Pesticides standard and cow’s milk sample preparation
Stock solutions of OPPs were prepared in methanol (1000 mg L−1) and diluted with deionized water for analysis of cow’s milk samples. For the optimization of effective parameters, standard OPP solutions (0.1 ng mL−1 of each standard chlorpyrifos and diazinon; 10 ng mL−1 of each phosphamidon and dimethoate) were spiked into a cow milk sample. The spiked level is 5–100× lower than the MRL set by the EU. However, the milk sample (10 mL) was diluted to 100 mL with deionized water to decrease the matrix effects from fat and protein (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 v/v, milk
10 v/v, milk![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water). The spiked OPPs were then extracted from the diluted cow’s milk samples by using the MSPE method based on the newly synthesized Fe3O4@G–CNPrTEOS sorbent.
water). The spiked OPPs were then extracted from the diluted cow’s milk samples by using the MSPE method based on the newly synthesized Fe3O4@G–CNPrTEOS sorbent.
2.7 Optimization of effective parameters on extraction performance
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 v/v) and 100 mg of adsorbent. Efficiency of the parameters was evaluated using relative peak area ratio (peak area of OPPs/peak area of IS).
10 v/v) and 100 mg of adsorbent. Efficiency of the parameters was evaluated using relative peak area ratio (peak area of OPPs/peak area of IS).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 v/v), respectively, and acetonitrile (2 mL for CNPr-SPE and 3 mL for C18-SPE) as the eluting solvent. Efficiency of the parameters was also evaluated using relative peak area ratio (peak area of OPPs/peak area of IS).
10 v/v), respectively, and acetonitrile (2 mL for CNPr-SPE and 3 mL for C18-SPE) as the eluting solvent. Efficiency of the parameters was also evaluated using relative peak area ratio (peak area of OPPs/peak area of IS).2.8 Magnetic solid phase extraction procedure
The synthesized adsorbent (100 mg) was dispersed in 120 mL of prepared cow’s milk. The milk solution was vortexed for 2 min and the adsorbent was collected using an external magnet. The sample solution was decanted and the adsorbent was washed with 5 mL deionized water and finally transferred to a 15 mL centrifuge tube. Thereafter, the extracted OPPs were desorbed from the adsorbent using 1 mL acetonitrile with the aid of 1 min agitation time. The solvent was dried under a gentle stream of nitrogen flow and reconstituted with a solution of 50 μL of 100 ng mL−1 hexaconazole (internal standard prepared in methanol). 1 μL of the reconstituted solution was injected into GC-μECD for analysis. Fig. 2 shows the schematic of MSPE for cow’s milk preparation.Conventional SPE was performed for comparison as follows; SPE cartridges were conditioned by passing 5 mL methanol, followed by 10 mL deionized water. Then, 4 mL and 5 mL of diluted cow’s milk (each spiked with 10 ng mL−1 of standard OPPs) were each passed through the CNPr-SPE and C18-SPE cartridge, respectively. After washing with water and 30 min air drying, the adsorbed OPPs were eluted from the CNPr-SPE and C18-SPE column using 2 and 3 mL acetonitrile, respectively. The eluate was then dried using a gentle stream of nitrogen gas (∼30 min) and the dried eluate was reconstituted with 50 μL of 100 ng mL−1 hexaconazole (internal standard) prepared in methanol before 1 μL was injected into the GC-μECD for analysis.
2.9 Method validation
The linearity was studied by replicating three analyses (n = 3) of the concentration range (0.1–10 ng mL−1 of each chlorpyrifos and diazinon; 2–200 ng mL−1 of each phosphamidon and dimethoate). The LOD, LOQ, and % recovery were calculated using eqn (1)–(3), respectively.42 The precision (% RSD) was performed using intra-day (repeatability) and inter-day (reproducibility) method. Three extractions (n = 3) were performed in one day and continued for five consecutive days (n = 15). The reusability of the as synthesized Fe3O4@G–CNPrTEOS was assessed for 10 consecutive adsorption–desorption cycles (extraction) using 100 mg of the adsorbent (10 ng mL−1 of each OPPs). The used adsorbent was washed sequentially using 2 mL methanol, 3 mL acetonitrile and finally 5 mL deionized water after each extraction.
 | (1) |
 | (2) |
 | (3) |
2.10 Milk sample analysis
The field applicability of the developed MSPE, C18-SPE and CNPr-SPE methods were studied for cow’s milk analysis. Unspiked cow’s milk samples (blank) were used for the three methods. The sample used for MSPE was spiked with 1 ng mL−1 of diazinon and chlorpyrifos and 20 ng mL−1 of phosphamidon and dimethoate. The spiked level was 5 ng mL−1 of diazinon and chlorpyrifos, and 100 ng mL−1 of phosphamidon and dimethoate for both the C18 SPE and CNPr-SPE. Optimized MSPE conditions were applied for these three extraction methods. Precision of the extraction process for all three methods was tested by repeating 3 times (% RSD 1.7–9.4%, n = 3) and 15 times (% RSD 10.7–17.6%, n = 15) and % R was calculated from the average data.3. Results and discussion
3.1 Characterization
FTIR spectra of GO, graphene and Fe3O4@G–CNPrTEOS are shown in Fig. 3. For GO (Fig. 3A), a broad band at 3400 cm−1 indicates O–H stretching vibration related to water. Due to its variety of functional groups, GO shows different bands; a peak at 1716 cm−1 indicates C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching for carboxylic groups, and carboxylic stretching vibration relates to C–OH at 1120 cm−1. Stretching vibrations for alkoxy (C–O) were observed at 1036 cm−1 and epoxy signals were observed at 1200 cm−1 and 820 cm−1. A peak at 1310 cm−1 indicates aromatic C
O stretching for carboxylic groups, and carboxylic stretching vibration relates to C–OH at 1120 cm−1. Stretching vibrations for alkoxy (C–O) were observed at 1036 cm−1 and epoxy signals were observed at 1200 cm−1 and 820 cm−1. A peak at 1310 cm−1 indicates aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C. Graphene shows fewer peaks in comparison to GO (Fig. 3B); the lump of the functional groups was removed from the GO in the reaction, followed by reduction using LiAlH4 and NaBH4. The as synthesized Fe3O4@G shows a peak at 1310 cm−1 related to aromatic C
C. Graphene shows fewer peaks in comparison to GO (Fig. 3B); the lump of the functional groups was removed from the GO in the reaction, followed by reduction using LiAlH4 and NaBH4. The as synthesized Fe3O4@G shows a peak at 1310 cm−1 related to aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C on graphene sheets and a peak at 584 cm−1 reveals Fe–O in Fe3O4. The FTIR spectra for synthesized Fe3O4@G–CNPrTEOS (Fig. 3C) shows two extra peaks at 2242 cm−1 and 1070 cm−1, identified as –C
C on graphene sheets and a peak at 584 cm−1 reveals Fe–O in Fe3O4. The FTIR spectra for synthesized Fe3O4@G–CNPrTEOS (Fig. 3C) shows two extra peaks at 2242 cm−1 and 1070 cm−1, identified as –C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N and Si–O, respectively. The peak at 2800 cm−1 is from the methyl groups of CNPrTEOS.
N and Si–O, respectively. The peak at 2800 cm−1 is from the methyl groups of CNPrTEOS.
The SEM morphology shows that CNPrTEOS nanoparticles were successfully modified on the graphene sheets (Fig. 4A). The TEM micrograph (Fig. 4B) shows the small Fe3O4 particles size and thin graphene sheets produced. Fig. 4C shows the EDX analysis of the synthesized adsorbent and the expected elements including C, O, N, Si and Fe were observed in the spectra.
 | ||
| Fig. 4 (A) SEM image, (B) TEM image and (C) EDX spectra of the as synthesized magnetic Fe3O4@G–CNPrTEOS adsorbent. | ||
3.2 GC-μECD instrument calibration
Linearity for standard diazinon and chlorpyrifos was obtained in the concentration range 10–500 ng mL−1 and for standard phosphamidon and dimethoate 200–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ng mL−1 (see ESI 1†). The instrument LOD obtained was 1.7, 2.8, 26.7 and 52.8 ng mL−1 for chlorpyrifos, diazinon, phosphamidon and dimethoate, respectively. The instrument LOQ obtained was 5.6, 9.3, 88.9 and 175.9 ng mL−1 for chlorpyrifos, diazinon, phosphamidon and dimethoate, respectively. The dynamic range was carried out by limit of linearity obtained in the range of 5.6 to 10
000 ng mL−1 (see ESI 1†). The instrument LOD obtained was 1.7, 2.8, 26.7 and 52.8 ng mL−1 for chlorpyrifos, diazinon, phosphamidon and dimethoate, respectively. The instrument LOQ obtained was 5.6, 9.3, 88.9 and 175.9 ng mL−1 for chlorpyrifos, diazinon, phosphamidon and dimethoate, respectively. The dynamic range was carried out by limit of linearity obtained in the range of 5.6 to 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ng mL−1 (for all the OPPs).
000 ng mL−1 (for all the OPPs).
3.3 Magnetic solid phase extraction optimization
3.4 Method validation
Under the optimal experimental conditions, linearity, LOD, LOQ, precision (intra-day and inter-day), % recovery, and EF of the three proposed methods were studied. Optimum conditions for MSPE are as follows; 120 mL sample volume, 2 min extraction time, 1 mL acetonitrile as desorption solvent, 1 min agitation time during the desorption process and 100 mg adsorbent dosage. The results achieved were compared with those using C18-SPE and CNPr-SPE cartridges. The extraction methods were assessed using matrix-matched calibration (milk samples were spiked with different concentrations of OPPs). To obtain the method linearity, the standard solutions in milk samples (0.1–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ng mL−1) were analyzed by MSPE, C18-SPE and CNPr-SPE followed by GC-ECD (Table 2). The proposed three methods provided good coefficient of determination (R2 ≥ 0.996) for all OPPs. The LOD values were calculated on the basis of (3 × SD)/m (n = 3) and the values obtained in the range from 0.01–0.58 ng mL−1 using the Fe3O4@G–CNPrTEOS MSPE. The LOQ value was calculated on the basis of (10 × SD)/m (n = 3). The MSPE method exhibited lower LOD (20–65×) and higher EF (25–30×) as compared to the C18-SPE and CNPr-SPE cartridges. This indicates that both cartridges are probably easily influenced by the cow’s milk matrix and interfering species occupy the adsorption sites. Otherwise, the as synthesized adsorbent was less affected possibly due to the higher electrostatic interaction (δ+ and δ−) and large π–π interaction (phenyl ring) between the analytes and adsorbent. The LOD obtained for the Fe3O4@G–CNPrTEOS MSPE was well below (58–1000× lower) the MRLs (10–50 ng kg−1) set by the EU.
000 ng mL−1) were analyzed by MSPE, C18-SPE and CNPr-SPE followed by GC-ECD (Table 2). The proposed three methods provided good coefficient of determination (R2 ≥ 0.996) for all OPPs. The LOD values were calculated on the basis of (3 × SD)/m (n = 3) and the values obtained in the range from 0.01–0.58 ng mL−1 using the Fe3O4@G–CNPrTEOS MSPE. The LOQ value was calculated on the basis of (10 × SD)/m (n = 3). The MSPE method exhibited lower LOD (20–65×) and higher EF (25–30×) as compared to the C18-SPE and CNPr-SPE cartridges. This indicates that both cartridges are probably easily influenced by the cow’s milk matrix and interfering species occupy the adsorption sites. Otherwise, the as synthesized adsorbent was less affected possibly due to the higher electrostatic interaction (δ+ and δ−) and large π–π interaction (phenyl ring) between the analytes and adsorbent. The LOD obtained for the Fe3O4@G–CNPrTEOS MSPE was well below (58–1000× lower) the MRLs (10–50 ng kg−1) set by the EU.
| Analyte | MSPE Fe3O4@G–CNPrTEOS | C18-SPE | CNPr-SPE | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Linearity (ng mL−1) | LOD (ng mL−1) | LOQ (ng mL−1) | EFa | Linearity (ng mL−1) | LOD (ng mL−1) | LOQ (ng mL−1) | EFa | Linearity (ng mL−1) | LOD (ng mL−1) | LOQ (ng mL−1) | EFa | |
a Enrichment factor calculated using equation;  , Vaq is sample volume (diluted milk) and Vorg is reconstituted organic solvent volume (50 μL). , Vaq is sample volume (diluted milk) and Vorg is reconstituted organic solvent volume (50 μL). |
||||||||||||
| Phosphamidon | 2–200 | 0.56 | 1.8 | 2400 | 100–1000 | 22.3 | 74.2 | 80 | 100–1000 | 11.1 | 37.1 | 100 |
| Dimethoate | 2–200 | 0.58 | 1.9 | 2400 | 100–1000 | 18.8 | 62.3 | 80 | 100–1000 | 19.6 | 65.5 | 100 |
| Diazinon | 0.1–10 | 0.02 | 0.08 | 2400 | 5–50 | 1.2 | 3.9 | 80 | 5–50 | 1.3 | 4.1 | 100 |
| Chlorpyrifos | 0.1–10 | 0.01 | 0.05 | 2400 | 5–50 | 0.52 | 2.4 | 80 | 5–50 | 0.53 | 1.7 | 100 |
The repeatability of the proposed MSPE Fe3O4@G–CNPrTEOS was investigated using intra-day (n = 3) and inter-day precision (n = 15) studies. The repeatability obtained was as follows; intra-day % RSD 1.7–9.4%, n = 3 and inter-day % RSD 10.7–17.6%, n = 15. Although inter-day precision shows slightly high % RSD (17.6%) yet single factor ANOVA analysis shows reliable results (p > 0.05) that demonstrate that there is no significant difference between inter-day precisions.
3.5 Milk sample analysis
The MSPE method based on the newly synthesized Fe3O4@G–CNPrTEOS was applied for the determination of OPPs in fresh milk (120 mL). The OPP concentrations were calculated for MSPE, C18-SPE and CNPr-SPE methods using calibration curves, and the results are listed in Table 3. In our study we did not detect any of the studied OPPs in the milk analyzed, thus we spiked the samples to assess the suitability (% R) of the developed method. However, OPP residues have been found in Italian raw milk (0.005–0.018 mg kg−1),1,3 Mexican commercial pasteurized milk (0.005–0.02 mg kg−1)4 and Spanish raw milk (0.005–0.2 mg kg−1).5 The Fe3O4@G–CNPrTEOS MSPE method gave a much better recovery of the OPPs (82–94%) compared to C18-SPE (43–59%) and CNPr-SPE (58–68%). Fig. 7 shows the chromatogram of the (A) unspiked and (B, C and D) spiked milk samples.| Analytes | Extraction methods | ||||||
|---|---|---|---|---|---|---|---|
| Unspiked (for three methods) | Fe3O4@G–CNPrTEOS MSPE | C18-SPE | CNPr-SPE | ||||
| Spiked (ng mL−1) | % R (% RSD, n = 3)b | Spiked (ng mL−1) | % R (% RSD, n = 3) | Spiked (ng mL−1) | % R (% RSD, n = 3) | ||
| a nd = not detected (OPPs studied was not detected in the fresh milk samples using all three methods). Blank was used for the three methods.b (n = 3) extraction recovery were analyzed for three times. | |||||||
| Phosphamidon | nd | 20 | 84 (4.1) | 100 | 59 (6.3) | 100 | 60 (9.3) |
| Dimethoate | nd | 20 | 82 (5.6) | 100 | 55 (4.4) | 100 | 57 (6.2) |
| Diazinon | nd | 1 | 94 (9.1) | 5 | 49 (3.6) | 5 | 68 (7.1) |
| Chlorpyrifos | nd | 1 | 89 (7.2) | 5 | 43 (3.8) | 5 | 64 (9.9) |
3.6 Proposed adsorption mechanism
Fig. 8 shows that the newly synthesized Fe3O4@G–CNPrTEOS adsorbent has five possible interaction points to adsorb OPPs. Hydrophobic interactions occur between the graphene sheets and the non-polar chlorpyrifos (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KO/W = 4.6) and diazinon (log
KO/W = 4.6) and diazinon (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KO/W = 3.5). Hydrophilic interactions occur between the hydrophilic tail of (CNPrTEOS) and polar phosphamidon (log
KO/W = 3.5). Hydrophilic interactions occur between the hydrophilic tail of (CNPrTEOS) and polar phosphamidon (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KO/W = 0.3) and dimethoate (log
KO/W = 0.3) and dimethoate (log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) KO/W = 0.7). Strong π–π interactions are found between the large delocalized electrons on graphene and the phenyl ring from diazinon and chlorpyrifos. These made Fe3O4@G–CNPrTEOS an effective adsorbent towards the non-polar OPPs. Also, hydrophilic interactions (dipole–dipole interactions (δ+ and δ−) and H-bonding) occur between OH bonded to Si from the CNPrTEOS chain and polar OPPs, causing good adsorption affinity towards the polar phosphamidon and dimethoate. Therefore, the MSPE Fe3O4@G–CNPrTEOS adsorbent is capable of simultaneous extraction of polar and non-polar OPPs from a complex fresh milk matrix with minimum matrix effects.
KO/W = 0.7). Strong π–π interactions are found between the large delocalized electrons on graphene and the phenyl ring from diazinon and chlorpyrifos. These made Fe3O4@G–CNPrTEOS an effective adsorbent towards the non-polar OPPs. Also, hydrophilic interactions (dipole–dipole interactions (δ+ and δ−) and H-bonding) occur between OH bonded to Si from the CNPrTEOS chain and polar OPPs, causing good adsorption affinity towards the polar phosphamidon and dimethoate. Therefore, the MSPE Fe3O4@G–CNPrTEOS adsorbent is capable of simultaneous extraction of polar and non-polar OPPs from a complex fresh milk matrix with minimum matrix effects.
3.7 Comparison with other techniques
Table 445–54 shows the LOD and recovery of the developed Fe3O4@G–CNPrTEOS MSPE method compared to other methods used for pesticide determination in milk. Pesticides isolated from milk samples using silica–C18 as a conventional SPE sorbent with LC-MS gave an LOD 20× lower49 than the Fe3O4@G–CNPrTEOS. However, SPE is a tedious and time consuming method for milk sample preparation due to the need for protein and fat removal using acid.The MSPE method and liquid–liquid extraction (LLE) followed by dispersive primary secondary amine clean-up (DPSA), and SPE with GC-MS gave an LOD (3.3–296 pg mL−1) that is 1.5–30× lower than the current Fe3O4@G–CNPrTEOS MSPE method.47 However, the LLE procedure is lengthier than the proposed Fe3O4@G–CNPrTEOS and the proposed MSPE method is greener.
Polydimethylsiloxane-SPME (PDMS-SPME) using gas chromatography-nitrogen phosphorus detector (GC-NPD) also gave a slightly lower LOD (2× lower) (0.05 ng mL−1).53 All the other methods listed in Table 4 produced LODs which are higher than the current LOD using Fe3O4@G–CNPrTEOS MSPE. For example triazines analysis in milk sample using diphasic dialysis extraction provided an LOD that is 100× higher50 compared to the current MSPE method.
Dispersive liquid–liquid microextraction based on the solidification of floating organic droplets (DLLME-SFO),45 solid matrix dispersion extraction (SMDE) method51 and Fe3O4@G–CNPrTEOS MSPE method provided good and comparable recovery for OPPs determination from fresh milk. The current Fe3O4@G–CNPrTEOS MSPE and DLLME-SFO method45 gave the same LOD but the SMDE method51 gave a much higher LOD (30–2600×).
The proposed Fe3O4@G–CNPrTEOS MSPE method also shows a 10–250× lower LOD for OPPs from milk samples compared to polydimethylsiloxane-divinylbenzene (PDMS-DVB) SPME using GC-MS (2.1 ng mL−1),46 LLE using GC-NPD (1–25 ng g−1)48 and C18-SPE using GC-NPD (1.0 ng mL−1).54
MSPE using Fe3O4@SiO2@C18 with GC-MS52 gave comparable LOD to the current method. This is due to π–π interactions between graphene and the OPPs and also electrostatic interactions. DLLME-SFO with GC-FPD45 gave the same LOD (0.1 ng mL−1) to the current Fe3O4@G–CNPrTEOS MSPE method. In contrast, C18-SPE provides only hydrophobic interactions and PDMS-DVB SPME provides less π–π interaction as compared to graphene-based sorbents. The proposed Fe3O4@G–CNPrTEOS MSPE method can be directly used in fresh milk samples without the need for tedious steps (removal of the fat and protein) and only a small amount of adsorbent (100 mg) is required to achieve the low LOD (ensuring compliance with MRL set by the EU) and good recovery of the OPPs.
| Adsorbent | Analyte | Sample | Method | LOD (ng mL−1) | % R | Detector | Ref. |
|---|---|---|---|---|---|---|---|
| a Dispersive liquid–liquid microextraction based on solidification of the floating organic droplet.b Gas chromatography-flame photometric detection.c Headspace solid phase microextraction.d Dispersive primary secondary amine.e Gel permeation chromatography.f Dispersive solid phase extraction; microextraction.g Gas chromatography-nitrogen phosphorus detection.h Solid matrix dispersion extraction.i OCPs: organochlorine pesticides; PCBs: polychlorinated biphenyls. | |||||||
| Fe3O4@G–CNPrTEOS | OPPs | Milk | MSPE | 0.1 | 82–104 | GC-μECD | This study |
| — | OPPs | Milk | DLLME-SFOa | 0.1 | 80–106 | GC-FPDb | 45 |
| PDMS-DVB | OPPs | Milk | HS-SPMEc | 2.1 | — | GC-MS | 46 |
| — | OPPs | Milk | LLE-DPSAd cleanup-SPE | 0.0033–0.296 | 34–102 | GC-MS | 47 |
| — | OPPs | Milk | LLE-GPCe clean up | 1–25 ng g−1 | 50.3–139.4 | GC-FPDb | 48 |
| Silica–C18 | Pesticides | Milk | DSPEf | 0.005 | 77.5 | LC-MS | 49 |
| — | Triazines | Milk | Dialysis | 10.0 | 89–116 | GC-NPDg | 50 |
| Flux-calcined macroporous diatomaceous material | OPPs | Milk | SMDEh | 3–260 ng g−1 | 76–109 | GC-FPDb | 51 |
| Fe3O4@SiO2@C18 | Endocrine disruptors (OCPs and PCBs)i | Milk | MSPE | 0.1–0.3 | 79–116 | GC-MS | 52 |
| PDMS | OPPs | Milk | SPME | 0.053 | — | GC-NPDg | 53 |
| C18 | OPPs | Milk | SPE | 1 | 44–117 | GC-NPDg | 54 |
4. Conclusion
The current study aims to report the efficient extraction of OPPs in fresh cow’s milk based on MSPE using newly synthesized Fe3O4@G–CNPrTEOS as an adsorbent with minimal sample preparation. The proposed MSPE was used for simultaneous direct determination and preconcentration of polar and non-polar OPPs from fresh cow’s milk. The results obtained indicate that the Fe3O4@G–CNPrTEOS MSPE method exhibits 50× lower LOD and higher enrichment (25–30×) than both commercial CNPr-SPE and C18-SPE cartridges. Recovery of the spiked OPPs (0.1 ng mL−1 of each standard chlorpyrifos and diazinon; 10 ng mL−1 of each phosphamidon and dimethoate) from fresh cow’s milk is excellent for the Fe3O4@G–CNPrTEOS MSPE (>82%) compared to the recovery using CNPr-SPE (>42%) and C18-SPE (>58%). The Fe3O4@G–CNPrTEOS MSPE exhibits great potential as an alternative sorbent for simultaneous polar and non-polar OPPs extraction from complex matrices such as milk. The main advantages of the proposed MSPE method are that only a simple dilution using deionized water is needed for the analysis of OPPs in milk, emulsions do not occur and so the method is simple.Acknowledgements
The authors would like to thank the Ministry of Education Malaysia for financial support through the Research Grants Number Q.J130000.262.10J43 and R.J130000.7826.3F452. Universiti Teknologi Malaysia is also acknowledged for the International Doctoral Fellowship (IDF) award received by H. R. Nodeh.References
- G. Pagliuca, A. Serraino, T. Gazzotti, E. Zironi and A. Borsari, J. Dairy Res., 2006, 73, 340–344 CrossRef CAS PubMed.
- T. Kiljanek, A. Niewiadowska and S. Semeniuk, Bull. Vet. Inst. Pulawy, 2013, 57, 185–189 CAS.
- T. Gazzotti, P. Sticca, E. Zironi, B. Lugoboni, A. Serraino and G. Pagliuca, Bull. Environ. Contam. Toxicol., 2009, 82, 251–254 CrossRef CAS PubMed.
- I. O. J. H. Salas, M. M. González, M. Noa, N. A. Pérez, G. Díaz, R. Gutiérrez and H. Zazueta, J. Agric. Food Chem., 2003, 51, 4468–4471 CrossRef PubMed.
- M. J. Melgar, M. Santaeufemia and M. A. Garcia, J. Environ. Sci. Health, Part B, 2010, 45, 595–600 CrossRef CAS PubMed.
- E. Kampire, B. T. Kiremire, S. A. Nyanzi and M. Kishimba, Chemosphere, 2011, 84, 923–927 CrossRef CAS PubMed.
- Regulation (EC) No 396/2005 of the European parliament and of the council of 23 February 2005 on maximum residue levels of pesticides in or on food and feed of plant and animal origin, Off. J. Eur. Union, 2005, L70, 1–16.
- A. A. Elbashir, M. M. A. Omar, W. A. Wan Ibrahim, O. J. Schmitz and H. Y. Aboul-Enein, Crit. Rev. Anal. Chem., 2014, 44, 107–141 CrossRef CAS PubMed.
- H. Bagheri and S. Banihashemi, Anal. Chim. Acta, 2015, 886, 56–65 CrossRef CAS PubMed.
- W. A. Wan Ibrahim, K. V. Veloo and M. M. Sanagi, J. Chromatogr. A, 2012, 1229, 55–62 CrossRef CAS PubMed.
- M. M. A. Omar, W. A. Wan Ibrahim and A. A. Elbashir, Food Chem., 2014, 158, 302–309 CrossRef CAS PubMed.
- J. Wu, Y. Qian, C. Zhang, T. Zheng, L. Chen and Y. Lu, Food Analytical Methods, 2013, 6, 1448–1457 CrossRef.
- Z. Li, R. Ma, S. Bai, C. Wang and Z. Wang, Talanta, 2014, 119, 498–504 CrossRef CAS PubMed.
- Q. Liu, M. Cheng, Y. Long, M. Yu, T. Wang and G. Jiang, J. Chromatogr. A, 2014, 1325, 1–7 CrossRef CAS PubMed.
- X. Ma, J. Wang, Q. Wu, C. Wang and Z. Wang, Food Chem., 2014, 157, 119–124 CrossRef CAS PubMed.
- X. Ma, J. Wang, M. Sun, W. Wang, Q. Wu and C. Wang, Anal. Methods, 2013, 5, 2809–2815 RSC.
- D. P. Suhas, T. M. Aminabhavi, H. M. Jeongd and A. V. Raghu, RSC Adv., 2015, 5, 100984–100995 RSC.
- Z. Li, M. Hou, S. Bai, C. Wang and Z. Wang, Anal. Sci., 2013, 29, 325–331 CrossRef CAS PubMed.
- H. R. Nodeh, W. A. Wan Ibrahim, M. A. Kamboh and M. M. Sanagi, RSC Adv., 2015, 5, 76424–76434 RSC.
- A. Mehdinia, H. Khani and S. Mozaffari, Microchim. Acta, 2013, 181, 89–95 CrossRef.
- W. Guan, C. Li, X. Liu, S. Zhou and Y. Ma, Food Addit. Contam., Part A: Chem., Anal., Control, Exposure Risk Assess., 2014, 31, 250–261 CrossRef CAS PubMed.
- H. Bagheri, A. Roostaie and M. Allahdadlalouni, Microchim. Acta, 2015, 182, 617–624 CrossRef CAS.
- W. A. Wan Ibrahim, H. R. Nodeh and M. M. Sanagi, Crit. Rev. Anal. Chem., 2015 DOI:10.1080/10408347.2015.1034354.
- W. Wang, R. Ma, Q. Wu, C. Wang and Z. Wang, Talanta, 2013, 109, 133–140 CrossRef CAS PubMed.
- K. J. Huang, Y. J. Liu, J. Li, T. Gan and Y. M. Liu, Anal. Methods, 2014, 6, 194–201 RSC.
- J. Ding, Q. Gao, X. S. Li, W. Huang, Z. G. Shi and Y. Q. Feng, J. Sep. Sci., 2011, 34, 2498–2504 CrossRef CAS PubMed.
- I. Ali, Z. A. Al-Othman and A. Al-Warthan, Desalin. Water Treat., 2016, 57, 10409–10421 CrossRef CAS.
- M. Sun, X. Ma, J. Wang, W. Wang, Q. Wu and C. Wang, J. Sep. Sci., 2013, 36, 1478–1485 CrossRef CAS PubMed.
- X. Zhang, Y. Liu, C. Sun, H. Ji, W. Zhao, S. Suna and C. Zhao, RSC Adv., 2015, 5, 100651–100662 RSC.
- D. He, X. Zhang, B. Gao, L. Wang, Q. Zhao and H. Chen, Food Control, 2014, 36, 36–41 CrossRef CAS.
- A. Kaouk, T. P. Ruoko, Y. Gönüllü, K. Kaunisto, A. Mettenbörger, E. Gurevich, H. Lemmetyinen, A. Ostendorfc and S. Mathur, RSC Adv., 2015, 5, 101401–101407 RSC.
- W. A. Wan Ibrahim, H. R. Nodeh, H. Y. Aboul-Enein and M. M. Sanagi, Crit. Rev. Anal. Chem., 2015, 45, 270–287 CrossRef CAS PubMed.
- F. Meng, W. Wei, J. Chen, X. Chen, X. Xu, M. Jiang, Y. Wang, J. Lua and Z. Zhou, RSC Adv., 2015, 5, 101121–101126 RSC.
- W. Zhang, X. Shi, Y. Zhang, W. Gu, B. Li and Y. Xian, J. Mater. Chem. A, 2013, 1, 1745–1753 CAS.
- M. Šafako and I. Šafak, J. Magn. Magn. Mater., 1999, 194, 108–112 CrossRef.
- C. Jiang, Y. Sun, X. Yu, Y. Gao, L. Zhang and Y. Wang, J. Chromatogr. B, 2013, 947–948, 49–56 Search PubMed.
- S. Wang, R. Wang, X. Wu, Y. Wang, C. Xue and J. Wu, J. Chromatogr. B, 2012, 905, 105–112 CrossRef CAS PubMed.
- Q. Wang, L. Huang, P. Yu, J. Wang and S. Shen, J. Chromatogr. B, 2013, 912, 33–37 CrossRef CAS PubMed.
- M. J. Wells, Principles of Extraction and the Extraction of Semivolatile Organics from Liquids, Sample Preparation Techniques in Analytical Chemistry, 2003, vol. 162, p. 37 Search PubMed.
- A. V. H. Herrera, J. Hernández-Borges and M. A. Rodríguez-Delgado, J. Chromatogr. A, 2009, 1216, 7281–7287 CrossRef PubMed.
- L. He, Y. Su, Y. Zheng, X. Huang, L. Wu and Y. Liu, J. Chromatogr. A, 2009, 1216, 6196–6203 CrossRef CAS PubMed.
- M. Ahmadvand, H. Sereshti and H. Parastar, J. Chromatogr. A, 2015, 1413, 117–126 CrossRef CAS PubMed.
- S. C. Moldoveanu and M. Kiser, J. Chromatogr. A, 2007, 1141, 90–97 CrossRef CAS PubMed.
- F. J. Schenck and J. Casanova, J. Environ. Sci. Health, Part B, 1999, 34, 349–362 CrossRef CAS PubMed.
- X. X. Miao, D. B. Liu, Y. R. Wang, U. Y. Yang, X. Y. Yang and H. U. Gong, J. Chromatogr. Sci., 2015, 53, 1813–1820 Search PubMed.
- F. M. Rodrigues, P. R. Mesquita, L. S. Oliveira, F. S. Oliveira, A. M. Filho, P. A. P. Pereira and J. B. Andradea, Microchem. J., 2011, 98, 56–61 CrossRef CAS.
- X. Chen, P. Panuwet, R. Hunter, A. M. Riederer, G. C. Bernoudy, D. B. Barr and P. B. Ryan, J. Chromatogr. B, 2014, 970, 121–130 CrossRef CAS PubMed.
- L. Yang, H. Li, F. Zeng, Y. Liu, R. Li, H. Chen, Y. Zhao, H. Miao and Y. Wu, J. Agric. Food Chem., 2012, 60, 1906–1913 CrossRef CAS PubMed.
- T. Dagnac, M. Garcia-Chao, P. Pulleiro, C. Garcia-Jares and M. Lompart, J. Chromatogr. A, 2009, 1216, 3702–3709 CrossRef CAS PubMed.
- M. A García, M. Santaeufemia and M. J. Melgar, Food Chem. Toxicol., 2012, 50, 503–510 CrossRef PubMed.
- A. Di Muccio, P. Pelosi, I. Camoni, D. A. Barbini, R. Dommarco and T. Generali, J. Chromatogr. A, 1996, 754, 497–506 CrossRef CAS PubMed.
- M. S. Synaridou, V. A. Sakkas, C. D. Stalikas and T. A. Albanis, J. Chromatogr. A, 2014, 1348, 71–79 CrossRef CAS PubMed.
- L. Cardeal Zde and C. M. Dias Paes, J. Environ. Sci. Health, Part B, 2006, 41, 369–375 CrossRef.
- G. Pagliuca, T. E. Zironi and P. Sticca, J. Chromatogr. A, 2005, 1071, 67–70 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra26742k |
| This journal is © The Royal Society of Chemistry 2016 |







