A colorimetric and ratiometric fluorescent probe for the selective detection of cyanide anions in aqueous media and living cells
Abstract
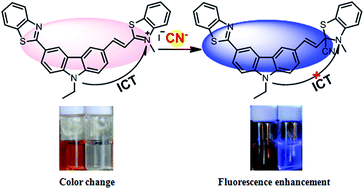
A new carbazole derivative 2-[(1E)-2-[6-(benzothiazolyl)-9-ethyl-9H-(carbazol-3-yl)]-ethenyl]-3-methyl-benzothiazolium iodide (BCB) was synthesized and its anion sensing properties were studied. BCB acts as an efficient colorimetric and ratiometric fluorescent probe used for the detection of cyanide ions in an aqueous solution even in the presence of other anions such as F−, AcO−, H2PO4−, SO42−, SO32−, S2−, SCN−, Cl−, Br−, I−, N3−, ClO4−, NO2−, HCO3− and CO32−. The cyanide addition to the benzothiazolium group induces an obstruction of the intramolecular charge transfer (ICT) and produces a dramatic hypsochromic shift in the absorption (152 nm) and emission (165 nm) profiles of BCB. The change in color of the solution was prominent and could be easily observed by the naked eye. The BCB probe forms a 1 : 1 adduct with CN− with a detection limit of 0.09 μM. The probe was successfully applied for the detection of CN− in natural water samples as well as in live HeLa cells.

 Please wait while we load your content...
Please wait while we load your content...