New fluorene-based chiral copolymers with unusually high optical activity in pristine and annealed thin films†
Min Ju Cho‡
a,
Jong-Soo Ahn‡a,
Young-Un Kima,
Hyun Ah Uma,
Paras N. Prasadb,
Geon Joon Lee*c and
Dong Hoon Choi*a
aDepartment of Chemistry, Research Institute for Natural Sciences, Korea University, 5 Anam-dong, Sungbuk-gu, Seoul 136-701, Korea. E-mail: dhchoi8803@korea.ac.kr
bInstitute for Lasers, Photonics and Biophotonics, Department of Chemistry, State University of New York at Buffalo, Buffalo, NY14260, USA
cDepartment of Electrical and Biological Physics, Kwangwoon University, 20 Kwangwoonro, Nowon-gu, Seoul 139-701, Korea. E-mail: gjlee@kw.ac.kr
First published on 24th February 2016
Abstract
Polyfluorene (PF)-based chiral alternating copolymers, i.e., PFPh and PFTh, were successfully synthesized to obtain high circular dichroism (CD) in pristine thin films. By tethering chiral side-chains to the fluorene moiety, the resultant PFPh and PFTh films intriguingly exhibited high CD even in pristine films, which was attributed to the relatively low steric hindrance between the repeating units and intermolecular interactions between the polymer chains. Compared to PFTh bearing a five-membered heteroaromatic thiophene monomer, PFPh containing a six-membered aromatic benzene monomer, showed significantly enhanced CD in an annealed film. Of particular importance, results of optical microscopy and atomic force microscopy can be used to assess the CD of a chiral polymer thin film after thermal annealing.
Introduction
Although chiroptical properties are often observed in biomaterials and organic small molecules, optically active π-conjugated polymers have attracted significant attention because of their high chiroptical properties in thin films.1–16 The highly transparent and homogeneous thin films of these polymers are easily formed using solution-processing techniques such as dip-coating, spin-coating, and drop-casting. Moreover, the circular dichroism (CD) of these thin films could be significantly enhanced through typical thermal annealing, which is better than other methods such as self-assembly or gelation in the solution state.17,18 These conjugated polymers exhibit strong absorption and CD signals in the visible-wavelength range, which is in contrast to many naturally occurring chiroptical polymers such as proteins, DNA, and cellulose.To achieve conjugated polymers with high chirality, many kinds of conjugated polymers have been designed, synthesized, and characterized through the introduction of optically active side-chain moieties. For example, a general approach involves the incorporation of phenylene-alt-phenylene or phenylene-alt-acetylene units into polymer chains such as poly(p-phenylene),2 poly(p-phenylacetylene),3,4 and polythiophene derivatives.5–8 Although the molecular structures of those polymers were modified using various chiral alkyl side-chains and repeating groups to ensure the chiral properties, they still suffered from low CD in the film state. Exceptionally, fluorene-based polymers with chiral alkyl side-chains are one of the more successful chiroptical materials. Fluorene-based homopolymers or copolymers have been reported to exhibit better chiroptical properties in thin films than other polymers.9–16
However, compared to fluorene-based conjugated homopolymers containing various chiral alkyl side-groups, copolymers with π-bridges such as five- or six-membered aromatic rings in the repeating unit have been less studied. Additionally, it has been suggested that the chirality of fluorene-based polymers in film states significantly depends on the annealing temperature and time. Therefore, it is worth determining if the chirality of polymers both in pristine and annealed films can be controlled through the incorporation of aromatic units in the backbone of a fluorene-based copolymer.
In this work, we synthesized a series of fluorene-based chiroptical copolymers with various π-extended bridges, such as thiophene, benzene, and benzo[c][1,2,5]thiadiazole (BT), which are expected to reduce the steric hindrance between the chiral alkyl-substituted fluorenes in the polymer backbone, as shown in Fig. 1. All copolymers (i.e., PFPh, PFTh, and PFBT) showed significantly different UV-Vis absorption and CD spectra in the visible region between 300 and 550 nm. It is very intriguing that the relatively high CD of the PFPh and PFTh copolymers was observed even in pristine films. In particular, the CD of the PFPh polymer bearing six-membered benzene rings was significantly enhanced in annealed film. It is very different from that of PFTh containing five-membered thiophene rings. Additionally, the chirality observed in a PFTh![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HAuCl4 blend film was recovered after exposure to hydrazine vapor; this reveals the possibility of new applications for vapor phase-sensing in the solid state.
HAuCl4 blend film was recovered after exposure to hydrazine vapor; this reveals the possibility of new applications for vapor phase-sensing in the solid state.
Results and discussion
Synthesis and characterization
The general synthetic procedures for the homopolymer and copolymers are outlined in Scheme 1. PF and PFBT were synthesized following the reported procedure.11 The PFPh copolymer was prepared via Suzuki coupling polymerization of 2,7-dibromo-9,9-bis((S)-3,7-dimethyloctyl)fluorene with 1,4-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzene. The PFTh copolymers were synthesized via Stille coupling of 2,7-dibromo-9,9-bis((S)-3,7-dimethyloctyl)fluorene with 2,5-bis-(trimethylstannyl)thiophene. To remove the low mass compounds and byproducts, the crude polymers were purified via successive Soxhlet extraction using methanol, acetone, hexanes, and chloroform. All polymers showed good solubility in methylene chloride, tetrahydrofuran (THF), chloroform, and halogenated aromatic solvents. Gel permeation chromatography (GPC) was employed to measure the molecular weights and polydispersity indices (PDIs) of the synthesized polymers. The number average molecular weight (Mn) values of PF, PFPh, PFTh, and PFBT were 7.7, 9.2, 7.7, and 8.7 kDa, respectively. Other measured and calculated parameters are tabulated in Table 1. The endothermic transition temperature (Ttrans) values obtained from differential scanning calorimetry were 56, 64, 57, and 73 °C for PF, PFPh, PFTh, and PFBT, respectively (Fig. S1†). The Ttrans values of PF and PFBT were slightly higher than those in literature because of their larger molecular weight. Additionally, the Ttrans values of the copolymers were higher than that of PF as a homopolymer. It is probable that the copolymers have relatively small molar ratios of alkyl groups to the aromatic backbone per repeating unit.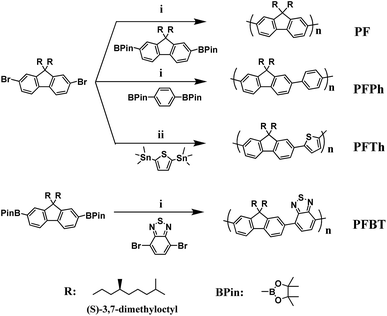 | ||
| Scheme 1 Synthetic procedure for PF-based chiroptical polymers: (i) Pd(PPh3)4, 2 M K2CO3, Aliquat 336, toluene, 100 °C, 12 h and (ii) Pd(PPh3)4, toluene, 100 °C, 5 h. | ||
| Polymer | Mn (kDa) | PDI | Pristine film | Annealed film (120 °C) | ||||
|---|---|---|---|---|---|---|---|---|
| Abs (nm) | CD (nm) | FOMa (deg) | Abs (nm) | CD (nm) | FOMa (deg) | |||
| a FOM is figure of merit (FOM = CD (deg)/absorbance).b (n) and (p) are the negative and positive bands, respectively. | ||||||||
| PF | 7.7 | 1.98 | 379 | — | — | 379 | 403 (n) | −8.30 (409 nm) |
| PFPh | 9.2 | 1.74 | 375 | 380 (n) | −0.42 (383 nm) | 375 | 388 (n) | −4.80 (393 nm) |
| PFTh | 7.7 | 2.69 | 432 | 434 (p) | +0.44 (434 nm) | 453 | 436 (p) | +0.67 (436 nm) |
| 478 (n) | −0.58 (478 nm) | 429 | 480 (n) | −0.90 (480 nm) | ||||
| PFBT | 8.7 | 1.85 | 459 | — | — | 459 | 477 (n) | −11.9 (485 nm) |
| 321 | 321 | 346 (n) | −5.90 (351 nm) | |||||
Optical properties and theoretical calculations
To investigate the optical and chiroptical properties of PF-based polymers, thin films were prepared by spin-coating a solution of the polymer in chloroform/monochlorobenzene (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v). A co-solvent system comprising both low and high boiling-point solvents was employed to generate smooth homogeneous films.11 Optical properties of polymer films made with chloroform and monochlorobenzene solutions were measured and the results were illustrated in Fig. S2–S5.† The UV-Vis absorption spectra of the PF-based polymer solutions and thin films are illustrated in Fig. 2, and the measured parameters are summarized in Table 1.
1 v/v). A co-solvent system comprising both low and high boiling-point solvents was employed to generate smooth homogeneous films.11 Optical properties of polymer films made with chloroform and monochlorobenzene solutions were measured and the results were illustrated in Fig. S2–S5.† The UV-Vis absorption spectra of the PF-based polymer solutions and thin films are illustrated in Fig. 2, and the measured parameters are summarized in Table 1.
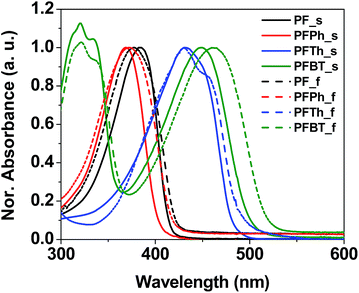 | ||
| Fig. 2 UV-Vis absorption spectra of PF-based chiroptical polymers in solution (solid line) and as thin-films (dashed line). | ||
Compared to those of the PF homopolymer, the absorption bands of copolymers PFTh and PFBT showed large bathochromic spectral shifts; this indicates that thiophene, as a strongly electron-donating unit, and BT, as an electron-withdrawing unit, effectively modulated the HOMO/LUMO levels and reduced the energy band-gap.
These results indicate that the optical properties of PF-based copolymers can be easily tuned by introducing aromatic units with different electron densities. The photoluminescence spectra of four chiral polymers are displayed in Fig. S6.†
In past studies, it was usually found that chiral alkyl chains containing polyfluorene derivatives displayed high CD in annealed thin films but weak or no CD in the pristine films.19 Additionally, to generate fairly high CD, some PF-based chiral polymers had to be thermally annealed at a high temperature for a long time.
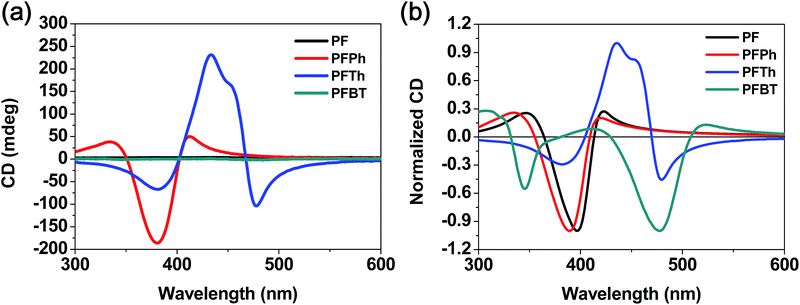
Fig. 3 shows the CD spectra of PF-based chiroptical polymers in pristine and annealed film states. PF and PFBT exhibited the same CD, which is consistent with previously reported results.11 In contrast, PFPh and PFTh displayed high CD in both pristine and annealed films. The CD spectrum of PFPh shows a strong negative band at 380 nm while that of PFTh exhibits medium-intensity negative and high-intensity positive CD bands at 390 and 467 nm, respectively. Compared to the PF homopolymer, the PFPh copolymer bearing benzene showed similar absorption and CD spectra while the PFTh copolymer, which contains thiophene, exhibited different CD; this indicates that the CD signal changes sign approximately at the center wavelength of the main π–π* transition, which is called the Cotton effect.
In order to ensure that the CD spectra really reflect chirality of the films, linear dichroism (LD) spectra of polymer films were measured. It was found that four polymers did not show significant linear dichroism effects in pristine and annealed films (Fig. S7†).
The high chiral activity of PFPh and PFTh in pristine films could not be explained through the typical characteristics of planarity and the dihedral angles between the repeating units in the polymer backbone. As shown in Fig. 4a, the optimized geometries of the simplified molecular structures had out-of-plane geometries with high twist angles of around 25.43–38.67°; these results do not correlate well with the optical activity.
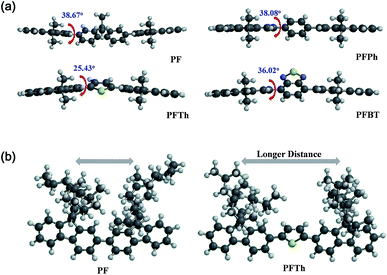 | ||
| Fig. 4 (a) Optimized molecular geometries of the PF-based chiroptical polymers with dihedral angles, and (b) the effect of the alkyl chain distance of PF and PFTh (DFT method, B3LYP/6-31G*). | ||
Therefore, these unusual phenomena must be explained in an alternate way. The potential energies of the simplified structures as a function of the torsion angles were obtained via theoretical calculations and are shown in Fig. S8 and S9.† There are significant differences in the torsional potentials of the four simplified structures: F-Ph in PFPh has the lowest torsional potential; in contrast, F-BT in PFBT has the highest torsional potential. This indicates that the formation of a helix structure is more favorable for PFPh in pristine films. The high torsional potential of PFBT could be due to charge transfer from F to the BT unit. Additionally, the PF homopolymer was composed of closely connected fluorenes bearing two bulky chiral alkyl chains; these interrupt the uniform orientation of the polymer main chain through higher steric hindrance between the chiral alkyl groups than that in PFPh and PFTh, as shown in Fig. 4b. PFBT, which contains bulky and electron-deficient BT units, had higher steric hindrance between F and the BT units, which induced strong intermolecular interactions between polymer chains. Therefore, the PF and PFBT films must be treated thermally to generate helix structures. In contrast, the molecular structures of PFPh and PFTh have less steric hindrance and fewer intermolecular interactions. Therefore, the polymer chains could re-organize to generate optical activity during spin-coating and drying.
Temperature dependence of the optical activity of the polymer films
As mentioned above, the CD of PF-based chiral copolymers significantly depend on certain factors such as annealing temperature and time, geometry of the molecular structure, and molecular weight. Among the many parameters, we mainly focused on the annealing temperature at a fixed annealing time (15 min) to observe the resultant variation of CD in this work. The four polymers showed significantly enhanced CD and reduced absorbance after annealing at different temperatures, as shown in Fig. 5 and 6. Interestingly, PF, PFPh, and PFBT exhibited larger CD than PFTh, which bears five-membered heteroaromatic thiophene units. Moreover, PFTh showed no change in CD when annealed above 90 °C even though high CD was observed in the pristine film. This was indirectly observed from the intensity of the absorption spectra at different annealing temperatures, as shown in Fig. 5 and 6.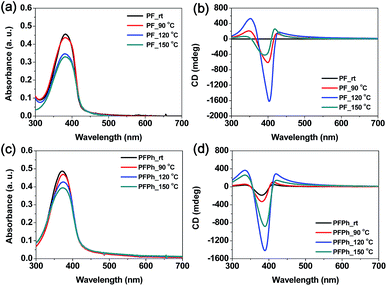 | ||
| Fig. 5 (a and c) UV-Vis absorption and (b and d) CD spectra of (a and b) PF and (c and d) PFPh in thin films. The film thicknesses of PF and PFPh were 45 and 50 nm, respectively. | ||
 | ||
| Fig. 6 (a and c) UV-Vis absorption and (b and d) CD spectra of (a and b) PFTh and (c and d) PFBT in thin films. The film thicknesses of PFTh and PFBT were 80 and 60 nm, respectively. | ||
To investigate the morphological variations, the surfaces of the polymer films before and after annealing were observed using polarized optical microscopy (OM), and their images are displayed in Fig. 7. Among the polymers, the annealed film of PFPh showed significantly different OM images: the annealed film was coarser than the pristine film and those of the other polymers. These results indicate that the CD of chiral polymers could be easily predicted by only measuring the OM image. However, the OM images did not provide sufficiently resolved images to investigate the changes of morphology in the PF, PFTh, and PFBT films. Using atomic force microscopy (AFM), highly resolved images of the surface topography of the PF-based polymer films before and after annealing were observed. All polymer films after thermal annealing showed coarser surfaces and larger domains than those of pristine films as shown Fig. 8. In particular, the thermally annealed PF, PFPh, and PFBT films exhibited markedly large island domains; this indicates that three polymers underwent significant macroscopic chain re-organization to generate the enhanced chiroptical properties.
 | ||
| Fig. 7 Cross-polarized optical microscopy images of (a and b) PF, (c and d) PFPh, (e and f) PFTh, and (g and h) PFBT in pristine films (left) and films annealed at 120 °C for 15 min (right). | ||
 | ||
| Fig. 8 AFM height images (10 μm × 10 μm) of the chiral polymer films before (left) and after thermal annealing (right): (a and e) PF; (b and f) PFPh; (c and g) PFTh; and (d and h) PFBT. | ||
Film-thickness dependence of CD in PFPh copolymers
The CD of PF-based chiral polymers is known to depend on the film thickness. Among the four polymers, these effects have already been well-studied for PF and PFBT.11 To investigate the film-thickness dependence of CD in PFPh, various samples of PFPh with different film thicknesses were prepared by varying the spin-coating speed. As shown in Fig. 9a, the CD of PFPh increased significantly with increasing film thickness. Intriguingly, we could not measure the full CD of the sample when the film thickness was greater than 56 nm because of the limited measurement range of instrument. These non-linear increases of CD made it difficult to determine the relationship between the linear increase of absorption in the pristine film and linear decrease of absorption in the annealed films, as shown in Fig. 9b. These unusual behaviors have also been mentioned in literature.11Oxidation and reduction of the HAuCl4-doped PFTh film
To investigate the effect of oxidation and reduction on the optical properties of PFTh, PFTh was doped by HAuCl4 as an oxidizing agent. Cho's group demonstrated that a HAuCl4-doped poly(3-hexylthiophene) (P3HT) thin film could be formed via a solution process.20 Accordingly, PFTh and HAuCl4 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 molar HAuCl4/polymer ratio) were dissolved in THF, and the resulting solution was spin-coated onto a glass substrate. The oxidized PFTh
3 molar HAuCl4/polymer ratio) were dissolved in THF, and the resulting solution was spin-coated onto a glass substrate. The oxidized PFTh![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HAuCl4 blend film was reduced by exposure to hydrazine vapor. The reaction mechanism for the oxidation and reduction of PFTh by HAuCl4 and hydrazine vapor is shown in Fig. 10a. After oxidation, thiophene moiety was positively polarized. Therefore, by adding sufficient hydrazine vapor, the lone pair electrons in hydrazine were donated to the relatively electron deficient thiophene moiety. As shown in Fig. 10b and c, the absorption spectrum of the HAuCl4-doped PFTh film is significantly different from that of the PFTh film, and the corresponding CD signals also disappeared. However, the absorption and CD spectra of the HAuCl4-doped PFTh film were interestingly recovered when the film was exposed to hydrazine vapor as a reduction agent.
HAuCl4 blend film was reduced by exposure to hydrazine vapor. The reaction mechanism for the oxidation and reduction of PFTh by HAuCl4 and hydrazine vapor is shown in Fig. 10a. After oxidation, thiophene moiety was positively polarized. Therefore, by adding sufficient hydrazine vapor, the lone pair electrons in hydrazine were donated to the relatively electron deficient thiophene moiety. As shown in Fig. 10b and c, the absorption spectrum of the HAuCl4-doped PFTh film is significantly different from that of the PFTh film, and the corresponding CD signals also disappeared. However, the absorption and CD spectra of the HAuCl4-doped PFTh film were interestingly recovered when the film was exposed to hydrazine vapor as a reduction agent.
Conclusions
We synthesized new PF-based chiral copolymers to investigate their optical and chiroptical behavior in pristine and thermally annealed films. The absorption and CD spectra of the copolymers were easily tuned via the introduction of five- and six-membered aromatics such as benzene, thiophene, and BT. Among the copolymers, PFPh and PFTh showed high CD in pristine-film states, which might be because of the small torsional potential energy between the repeating units and low intermolecular interactions between polymer chains. In particular, it should be emphasized that optical microscopy and atomic-force microscopy enable prediction of the CD of chiral polymer thin films after annealing. Additionally, the chirality observed in a PFTh![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HAuCl4 blend film was recovered via exposure to hydrazine vapor, which might support the sustainable chiral structure under oxidation and reduction at ambient conditions.
HAuCl4 blend film was recovered via exposure to hydrazine vapor, which might support the sustainable chiral structure under oxidation and reduction at ambient conditions.
Experimental
Materials
All commercially available materials were purchased from Aldrich, Tokyo Chemical Industry (TCI), and ACROS Co. 2,7-Dibromo-9,9-bis((S)-3,7-dimethyloctyl)fluorene, 2,7-bispinacoyl-9,9-bis((S)-3,7-dimethyloctyl)fluorene boronic ester, PF, and PFBT were prepared according to literature.11Synthesis of PFPh
To a solution of 2,7-dibromo-9,9-bis((S)-3,7-dimethyloctyl)fluorene (0.25 g, 0.4 mmol) and 1,4-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzene (0.132 g, 0.4 mmol) in toluene (15 mL) was added 2 M K2CO3 (5 mL), Pd(PPh3)4 (22.8 mg, 20.0 μmol), and a trace amount of Aliquat 336 under an argon atmosphere. The mixture was allowed to stir at 100 °C for 12 h. The cooled mixture was added to vigorously stirred methanol (200 mL), and the resulting precipitate was filtered. The crude product was purified by successive Soxhlet extraction with acetone, hexane, and chloroform. The chloroform fraction was evaporated completely and then precipitated from methanol to afford the product as a light yellow solid (0.15 g, 74%). (Mn = 9.2 kDa, PDI = 1.74). Anal. calcd for (C37H50S2)n: C, 84.35; H, 9.57; S, 6.09. Found: C, 84.13; H, 9.81; S, 6.32.Synthesis of PFTh
To a solution of 2,7-dibromo-9,9-bis((S)-3,7-dimethyloctyl)fluorene (0.31 g, 0.5 mmol) and 2,5-bis(trimethylstannyl)thiophene (0.164 g, 0.4 mmol) in toluene (15 mL) was added Pd(PPh3)4 (22.8 mg, 20.0 μmol) under an argon atmosphere. The mixture was stirred at 100 °C for 5 h. After cooling the mixture, it was added to vigorously stirred methanol (200 mL), and the resulting polymer precipitate was filtered. The crude product was purified by successive Soxhlet extraction with acetone, hexane, and chloroform. The chloroform fraction was evaporated completely and then precipitated from methanol to afford the product as a yellow solid (0.15 g, 74%). (Mn = 7.7 kDa, PDI = 2.69). Anal. calcd for (C39H52)n: C, 89.94; H, 10.06. Found: C, 89.83; H, 10.13.Instrumentation
1H NMR spectra were recorded on a Varian Mercury NMR 300 MHz spectrometer using deuterated chloroform purchased from Cambridge Isotope Laboratories, Inc. Elemental analyses for determining the C, H, and N composition were performed using an EA1112 (Thermo Electron Corp.) elemental analyzer. The molecular weights of the polymers were determined by gel permeation chromatography (Waters GPC, Waters 515 pump, Waters 410 RI, 2× PLgel Mixed-B) using polystyrene as the standard and chloroform as an eluent (T = 35 °C). Atomic force microscopy (Advanced Scanning Probe Microscope, XE-100, PSIA) operating in tapping mode with a silicon cantilever was used to characterize the surface morphologies of the polymer films. Absorption spectra of the samples in film and solution states were obtained using a UV-Vis spectrometer (HP 8453). Photoluminescence spectra were recorded using a Hitachi F-7000 fluorescence spectrophotometer. The CD spectra were measured using a spectropolarimeter (Jasco, J-815, Japan). Polarized optical microscopy was carried out using a SAMWON (SW-011863).Film sample preparation
To investigate the optical properties of the films, such as absorption and chiroptical behavior and film morphology, the film samples of the PF-based chiral polymers were deposited on glass substrates using the spin-coating method. Transparent thin films were fabricated by spin-coating with a co-solvent system, i.e., chloroform/chlorobenzene (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v%). The samples were annealed at 90, 120, or 150 °C for 15 min under ambient conditions. PFTh and HAuCl4 (1
1 v/v%). The samples were annealed at 90, 120, or 150 °C for 15 min under ambient conditions. PFTh and HAuCl4 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 molar HAuCl4/polymer ratio) were dissolved in THF, and the resulting solution was spin-coated onto a glass substrate.
3 molar HAuCl4/polymer ratio) were dissolved in THF, and the resulting solution was spin-coated onto a glass substrate.
Acknowledgements
This research was supported by the National Research Foundation of Korea (NRF2012R1A2A1A01008797) and from Key Research Institute Program (NRF201200020209). This work was supported by a grant from the National Research Foundation of Korea (NRF) funded by the Korean government (MSIP) (NRF-2010-0027963) and from Basic Science Research Program through the National Research Foundation of Korea funded by the Ministry of Education, Science and Technology, Korea [2015R1D1A1A01057501]. M. J. Cho particularly acknowledges the support from a Korea University grant (2014–2015).Notes and references
- S. Zahn and T. M. Swager, Angew. Chem., Int. Ed., 2002, 41, 4226–4230 CrossRef.
- R. Fiesel and U. Scherf, Acta Polym., 1998, 49, 445–449 CrossRef CAS.
- R. Fiesel and U. Scherf, Macromol. Rapid Commun., 1998, 19, 427–431 CrossRef CAS.
- R. Fiesel, C. E. Halkyard, M. E. Rampey, L. Kloppenburg, S. L. Studer-Martinez, U. Scherf and U. H. F. Bunz, Macromol. Rapid Commun., 1999, 20, 107–111 CrossRef CAS.
- B. M. W. Langeveld-Voss, R. A. J. Janssen, M. P. T. Christiaans, S. C. J. Meskers, H. P. J. M. Dekkers and E. W. Meijer, J. Am. Chem. Soc., 1996, 118, 4908–4909 CrossRef CAS.
- G. Lakhwani, G. Koeckelberghs, S. C. J. Meskers and R. A. J. Janssen, Chem. Phys. Lett., 2007, 437, 193–197 CrossRef CAS.
- C. R. G. Grenier, S. J. George, T. J. Joncheray, E. W. Meijer and J. R. Reynolds, J. Am. Chem. Soc., 2007, 129, 10694–10699 CrossRef CAS PubMed.
- M. M. Bournan and E. W Meijer, Adv. Mater., 1995, 7, 385–387 CrossRef.
- M. Oda, H.-G. Nothofer, U. Scherf, V. Šunjić, D. Richter, W. Regenstein and D. Neher, Macromolecules, 2002, 35, 6792–6798 CrossRef CAS.
- M. Oda, H.-G. Nothofer, G. Lieser, U. Scherf, S. C. J. Meskers and D. Neher, Adv. Mater., 2000, 12, 362–365 CrossRef CAS.
- R. Abbel, A. P. H. J. Schenning and E. W. Meijer, Macromolecules, 2008, 41, 7497–7504 CrossRef CAS.
- H.-Z. Tang, M. Fujiki and M. Motonaga, Polymer, 2002, 43, 6213–6220 CrossRef CAS.
- G. Lakhwani and S. C. J. Meskers, Macromolecules, 2009, 42, 4220–4223 CrossRef CAS.
- B. Nowacki, H. Oh, C. Zanlorenzi, H. Jee, A. Baev, P. N. Prasad and L. Akcelrud, Macromolecules, 2013, 46, 7158–7165 CrossRef CAS.
- G. Lakhwani and S. C. J. Meskers, J. Phys. Chem. Lett., 2011, 2, 1497–1501 CrossRef CAS.
- H. S. Oh, H. Jee, A. Baev, M. T. Swihart and P. N. Prasad, Adv. Funct. Mater., 2012, 22, 5074–5080 CrossRef CAS.
- S. J. George, T. F. A. de Greef, R. Bovee, J. L. J. van Dongen, A. P. H. J. Schenning and E. W. Meijer, Chem.–Asian J., 2009, 4, 910–917 CrossRef CAS PubMed.
- A. R. A. Palmans and E. W. Meijer, Angew. Chem., Int. Ed., 2007, 46, 8948–8968 CrossRef CAS PubMed.
- K. Watanabe, Y. Koyama, N. Suzuki, M. Fujiki and T. Nakano, Polym. Chem., 2014, 5, 712–717 RSC.
- Y. D. Park, D. H. Kim, J. A. Lim, J. H. Cho, Y. Jang, W. H. Lee, J. H. Park and K. Cho, J. Phys. Chem. C, 2008, 112, 1705–1710 CrossRef CAS.
Footnotes |
| † Electronic supplementary information (ESI) available: DSC curves, PL spectra, absorption and CD spectra, and torsional potential energies of all polymers. See DOI: 10.1039/c5ra26523a |
| ‡ These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2016 |