Hydrolytic stability of end-linked hydrogels from PLGA–PEG–PLGA macromonomers terminated by α,ω-itaconyl groups†
Abstract
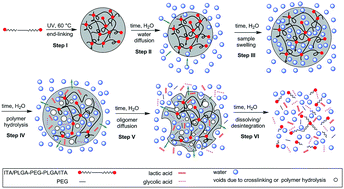
Biodegradable amphiphilic PLGA–PEG–PLGA triblock copolymers end-terminated with itaconic acid (ITA) having reactive double bonds were synthesized by ring opening polymerization. The prepared α,ω-itaconyl-PLGA–PEG–PLGA telechelic macromonomers were additionally covalently crosslinked under an inert atmosphere by blue light irradiation without the use of a further cross-linker resulting in end-linked polymeric networks. The effects of the ITA amount attached to the α,ω-itaconyl-PLGA–PEG–PLGA copolymers and the crosslinking time on swelling behaviours and hydrolytic stability of the prepared well-defined polymeric network were investigated. Physicochemical properties were characterized by proton and carbon nuclear magnetic resonance spectroscopy (1H NMR, 13C NMR), proton nuclear magnetic resonance relaxometry, attenuated total reflectance Fourier transformed infrared spectroscopy (ATR-FTIR) and thermogravimetric analysis (TGA). It was found that the hydrolytic stability of ITA modified PLGA–PEG–PLGA end-linked hydrogels enhances with both increasing the time of crosslinking and the amount of double bonds attached to α,ω-itaconyl-PLGA–PEG–PLGA polymer chains. In comparison with the original un-crosslinked α,ω-itaconyl-PLGA–PEG–PLGA copolymer, the hydrolytic stability of the end-linked hydrogels significantly increased. Three kinds of water fractions (unbound, weakly and strongly bonded) were determined by proton NMR relaxometry in hydrogels containing 63 mol% of ITA crosslinked for 40 minutes. Even for hydrogels surviving 32 days in water the NMR relaxometry showed structural collapse of the hydrogel probably due to breaking of end-linked nodes followed by hydrolysis faster than water diffusion after day 15 of immersion. End-linked α,ω-itaconyl-PLGA–PEG–PLGA hydrogels can be used in medical, biological or tissue engineering applications.

 Please wait while we load your content...
Please wait while we load your content...