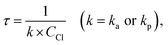Mechanisms and kinetic parameters for the gas-phase reactions of anthracene and pyrene with Cl atoms in the presence of NOx†
Juan Dang and
Maoxia He*
Environment Research Institute, Shandong University, Jinan 250100, P. R. China. E-mail: hemaox@sdu.edu.cn; Fax: +86-531-8836-1990
First published on 27th January 2016
Abstract
Due to their global dispersion and toxicity, polycyclic aromatic hydrocarbons (PAHs) in the atmosphere have become a serious environmental concern. Atmospheric reactions of PAHs with Cl atoms are of critical importance in specific areas such as the marine boundary layer and coastal regions. In this study, the mechanisms of the Cl radical-initiated atmospheric oxidation of anthracene (Ant) and pyrene (Pyr) were investigated using quantum chemistry calculations. The rate constants for the crucial elementary reactions were estimated. The oxidation products of the gas-phase reactions of Ant and Pyr with Cl atoms are chloro-Ants, chloro-Pyrs, 2-chloroanthracen-1-one, 1-chloropyren-2-one, 1-chloropyren-3-one, 4-chloropyren-5-one, 1-chloro-2-hydroperoxyanthracene, 2-chloro-1-hydroperoxyanthracene, 1-chloro-2-hydroperoxypyrene, 4-chloro-5-hydroperoxypyrene, epoxides, dialdehydes, 9-nitroanthracene, 1-nitroanthracene and nitropyrenes. 9-Nitroanthracene can be more easily produced by the gas-phase reaction of Ant with Cl atoms than that of Ant with OH radicals. Water plays a vital role in the formation of 9-nitroanthracene, resulting from the reactions with NO2. This comprehensive mechanistic study is the first one reported for the Cl radical-initiated atmospheric oxidation of PAHs. The calculated overall rate constants for the Cl addition reactions of Ant and Pyr are 5.87 × 10−12 and 2.81 × 10−12 cm3 per molecule per s, respectively, at 298 K and 1 atm.
1. Introduction
Polycyclic aromatic hydrocarbons (PAHs) are a class of ubiquitous environmental contaminants that mainly arise from incomplete combustion of fossil fuels and biomass,1–3 and they have been constantly detected in the atmosphere throughout the world.4–7 The annual atmospheric emissions of 16 priority PAHs in Asian countries accounted for 53.5% of the total global emissions (504 Gg), with the highest emissions being from China (106 Gg) and India (67 Gg) in 2007.8 Because of their prevalent occurrence, persistence in the environmental matrices and extremely hazardous properties to human health,9–11 the fate of these compounds in the atmosphere is of high public concern.In the troposphere, PAHs can undergo gas-phase reactions with OH radicals, NO3 radicals, O3 and chlorine atoms.12,13 Although the reactions with OH radicals, NO3 radicals, and O3 are generally considered to be the main sinks for gaseous PAHs, the reactions with Cl atoms may be significant in the marine boundary layer and in coastal regions, especially in the Arctic lower troposphere during spring.12–14 The peak concentration of Cl atoms is 105 molecules per cm3 or more in the marine boundary layer at dawn.13,15,16 In addition, chlorine atoms are highly reactive towards organic substances, and are 103 times more reactive than OH radicals;17 therefore, even at relatively low concentrations, chlorine atoms have the ability to enhance the oxidation of organics in the atmosphere. Previous studies showed that the rate constants for the reactions of many volatile organic compounds with Cl atoms are ∼102 higher than those for reactions with OH radicals;13 therefore, the Cl atom reactions could play an important role in the atmospheric oxidation of PAHs in specific areas such as the marine boundary layer and coastal regions. In particular, some oxidation products (chlorinated PAHs included) of atmospheric reactions of PAHs with Cl atoms have been identified, with enhanced toxicities, mutagenicities and aryl hydrocarbon receptor activities, compared with the corresponding parent PAHs.18–20 Therefore, it is necessary to investigate the reaction mechanisms of PAHs with Cl atoms to clarify their atmospheric transformations. To date, only two studies (Wang et al., 2005 and Long et al., 2007) have reported the rate constants for the reactions of naphthalene and alkylnaphthalenes with Cl atoms, and the reaction mechanisms have not been fully elucidated.13,21,22 Clearly, more study is needed to obtain more comprehensive knowledge.
Anthracene (Ant) and pyrene (Pyr) are members of the 16 USEPA priority PAHs, which have been detected at high concentrations in certain areas.12,23,24 During the summer of 2003, the field measurements in the Shing Mun Tunnel of Hong Kong showed that the average ambient air concentrations of Ant ranged from 30.5 to 15.0 ng m−3.23 In 2008, air samples collected from the Oporto metropolitan area in northern Portugal, revealed that the maximum concentration of Pyr was up to 9.44 ng m−3.25 Considering their widespread occurrence in air, it is critical to clarify the gas-phase reactions of Ant and Pyr with Cl atoms. Due to the scarcity of efficient detection schemes for intermediate radicals in the experimental studies, quantum chemical calculations can be of considerable help to illuminate the reaction mechanisms. In this study, we carried out a theoretical study on the Cl-initiated atmospheric oxidation of Ant and Pyr in the presence of NOx using density functional theory (DFT).26 Modeling of the fate of PAHs in the atmosphere places a high demand on kinetic parameters; therefore, the rate constants of key elementary reactions involved in the Cl-initiated oxidation of Ant and Pyr were also calculated.
2. Computational method
The quantum chemical calculations were performed with the Gaussian 09 software package27 on a supercomputer. The geometries of the reactants, intermediates, transition states and products were optimized at the BB1K/6-31+G(d,p) level. The vibrational frequencies were calculated to identify the structures obtained as true minima or first-order saddle points. The intrinsic reaction coordinate (IRC) analysis was carried out to confirm that each transition state connects to the right minima along the reaction path. A more flexible basis set, 6-311+G(3df,2p), was used to determine the single point energies of various species. The overall energetic profiles were constructed to locate the energetically favorable reaction pathways.The MESMER program, a Master Equation Solver for Multi-Energy Well Reactions, uses matrix techniques to formulate and solve the energy grained master equation (EGME) for reaction systems composed of an arbitrary number of wells, transition states, sinks, and reactants. The master equation is as follows:28
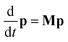 | (1) |
The grain size used was 100 cm−1, and the maximum grain energy was 25 kT in the calculation process. The classical method was applied to calculate the density of states. The exponential down model was implemented for describing collisional transfer probabilities. Simple RRKM or the MesmerILT method was provided for the calculation of rate constants.
For reactions with a well-defined transition state, the common way of obtaining rate constants from a particular energy grain, k(E) is by using Rice–Ramsperger–Kassel–Marcus (RRKM) theory. The RRKM expression is given by the equation as follows:28
 | (2) |
If no transition state is specified, an inverse Laplace transform (ILT) provides a mathematical formalism for deriving k(E)s from an Arrhenius fit to a set of k(E)s. The basis of the ILT is the standard Boltzmann average, which may be expressed as follows:28
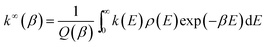 | (3) |
3. Results and discussion
3.1 Reactions with Cl atoms
The atmospheric reactions of volatile organic compounds with Cl atoms proceed via Cl addition to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds, and H abstraction by Cl atoms. Vereecken et al. showed that H abstraction by Cl atoms is less important, even for abstraction of the highly reactive allylic hydrogen atoms.29 According to our simulation, the H abstraction from Ant and Pyr are endothermic reactions; for example, the H abstractions from positions C1, C2, and C9 of Ant are endothermic by 7.59 kcal mol−1, 7.85 kcal mol−1, and 8.33 kcal mol−1, respectively. However, the Cl addition reactions are highly exothermic and therefore only the Cl addition reactions are discussed in this study. For convenience of description, the C atoms in Ant and Pyr are numbered, as presented in Fig. 1. The Ant molecule at ground state is of D2h symmetry, and C atoms in Ant fall into four groups: C1, C4, C5 and C8 atoms belong to one equivalent group; C2, C3, C6 and C7; C9 and C10; C11, C12, C13 and C14 belong to the other three equivalent groups. Three different Cl–Ant adduct isomers can be formed via Cl addition to C1, C2 and C9 atoms. In particular, the corresponding Cl–Ant adduct cannot be formed from the Cl addition to the C11 position. Analysis of the molecular structure of Pyr shows that there are four different types of C atoms theoretically leading to four Cl addition processes. Because the Cl addition to C2 and C3a positions cannot occur, only two Cl–Pyr adducts are generated. The reaction schemes of the Cl additions embedded with reaction heats (ΔH, 0 K) are depicted in Fig. 1. The configuration of the Cl–Ant adducts and Cl–Pyr adducts (IM1–IM5) are shown in Fig. S1 of ESI.†
C bonds, and H abstraction by Cl atoms. Vereecken et al. showed that H abstraction by Cl atoms is less important, even for abstraction of the highly reactive allylic hydrogen atoms.29 According to our simulation, the H abstraction from Ant and Pyr are endothermic reactions; for example, the H abstractions from positions C1, C2, and C9 of Ant are endothermic by 7.59 kcal mol−1, 7.85 kcal mol−1, and 8.33 kcal mol−1, respectively. However, the Cl addition reactions are highly exothermic and therefore only the Cl addition reactions are discussed in this study. For convenience of description, the C atoms in Ant and Pyr are numbered, as presented in Fig. 1. The Ant molecule at ground state is of D2h symmetry, and C atoms in Ant fall into four groups: C1, C4, C5 and C8 atoms belong to one equivalent group; C2, C3, C6 and C7; C9 and C10; C11, C12, C13 and C14 belong to the other three equivalent groups. Three different Cl–Ant adduct isomers can be formed via Cl addition to C1, C2 and C9 atoms. In particular, the corresponding Cl–Ant adduct cannot be formed from the Cl addition to the C11 position. Analysis of the molecular structure of Pyr shows that there are four different types of C atoms theoretically leading to four Cl addition processes. Because the Cl addition to C2 and C3a positions cannot occur, only two Cl–Pyr adducts are generated. The reaction schemes of the Cl additions embedded with reaction heats (ΔH, 0 K) are depicted in Fig. 1. The configuration of the Cl–Ant adducts and Cl–Pyr adducts (IM1–IM5) are shown in Fig. S1 of ESI.†
 | ||
| Fig. 1 The Cl addition reaction scheme of Ant and Pyr embedded with reaction heat ΔH (kcal mol−1). ΔH is calculated at 0 K. | ||
All the Cl addition pathways are highly exothermic with no potential barriers, which indicate that they can occur readily under general atmospheric conditions. According to the previous study, there is also no potential barrier on the addition pathway for the reaction of Cl with benzene.30 At the BB1K/6-311+G(3df,2p) level, the reaction heats of the Cl addition reactions are distributed between −19.83 and −16.31 kcal mol−1. The resulting Cl–Ant and Cl–Pyr adducts will further react with O2/NOx for their removal.
3.2 Secondary reactions
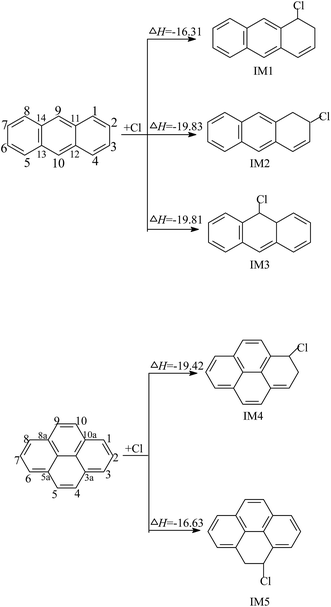
 | ||
| Fig. 2 The formation scheme of chloro-Ants and chloro-Pyrs, embedded with the potential barrier ΔE (kcal mol−1) and reaction heat ΔH (kcal mol−1). ΔH is calculated at 0 K. | ||
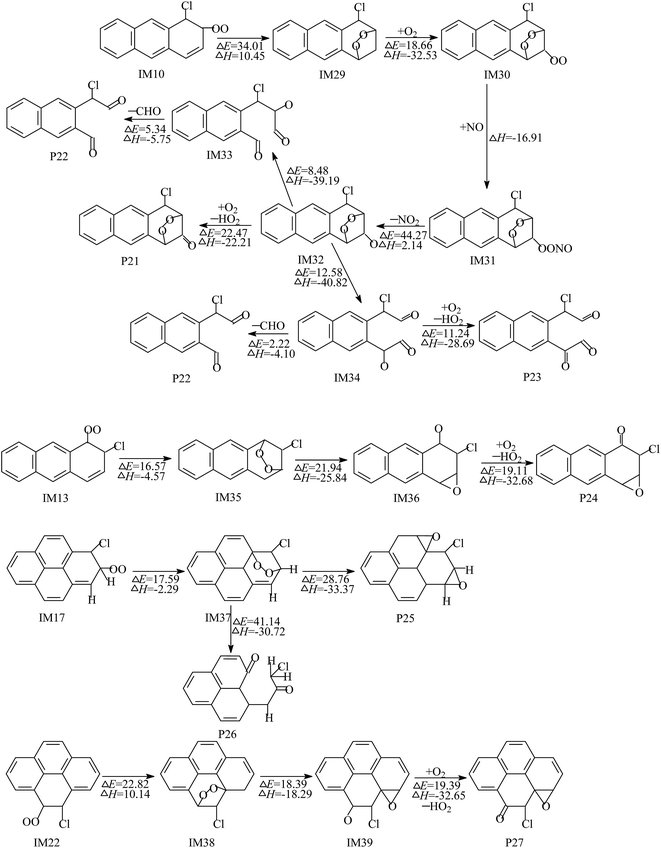
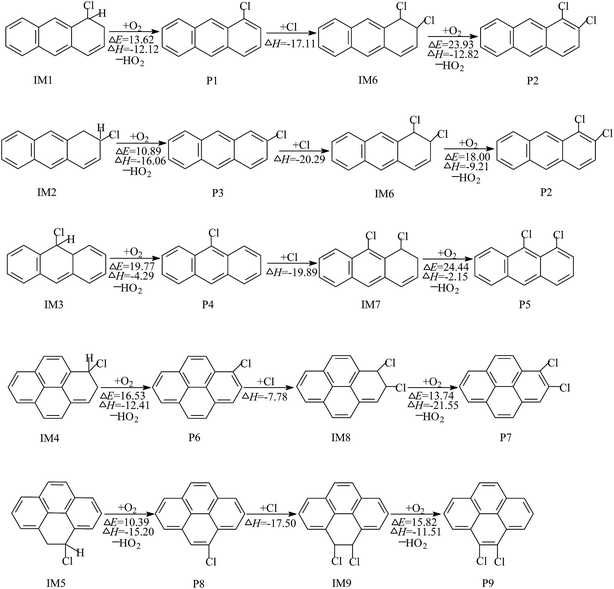
The reaction of the Cl–Ant and Cl–Pyr adducts with O2/NO are displayed in Fig. 3. O2 can attack the C atoms, using an unpaired electron in Cl–Ant and Cl–Pyr, from two different directions: the cis- or trans-position of the –Cl group with respect to the aromatic ring (Fig. S2 of ESI†). Therefore, six O2–Cl–Ant adducts and four O2–Cl–Pyr adducts are expected to be formed. By comparison of the potential barriers and reaction heats of these reaction pathways, the O2 addition from the trans-position of the –Cl group is favored over those of the other ones; therefore, only the subsequent reactions of the favorable adducts are depicted in Fig. 3. As shown, the O2–Cl–Ant adducts IM10 and IM13 have similar reaction channels, which include barrierless association of NO, rupture of the O–ONO bond, elimination of HCl and H abstraction by O2. Calculations show that 2-chloroanthracen-1-one and the radical P12 are more easily obtained because of the lower potential barrier and strong exothermicity. The intermediate IM16 is difficult to be formed by O2 addition. The potential barrier of this reaction is calculated to be 65.38 kcal mol−1 and the process is endothermic by 41.58 kcal mol−1. The intermediate IM17 formed by O2 addition can undergo NO addition without a potential barrier, followed by cleavage of the O–ONO bond, H abstraction by O2 and rupture of the C–O bond, leading to the formation of 1-chloropyren-2-one and 1-chloropyren-3-one. The subsequent reactions of IM22 are similar to those of IM17, resulting in the formation of 4-chloropyren-5-one. The calculated profiles of the potential energy surface show that the cleavage of the O–ONO bond is the rate-determining process, due to its high potential barrier.
 | ||
| Fig. 3 The O2/NO addition reaction scheme of Cl–Ant and Cl–Pyr adducts, embedded with the potential barrier ΔE (kcal mol−1) and reaction heat ΔH (kcal mol−1). | ||
As shown in Fig. 4, the intermediate IM10 produced by O2 addition can also proceed with H migration from the C atom to the O atom, leading to the formation of IM25. The subsequent reaction of IM25 involves two elementary reactions: elimination of the HO2 radical and H abstraction by O2. These processes ultimately result in the generation of 1-chloroanthracene and 1-chloro-2-hydroperoxyanthracene. Through the analysis and comparison, the reaction of the H migration is the rate-determining step, due to the high potential barrier. The potential barrier of the H migration is calculated to be 26.50 kcal mol−1 and the process is exothermic by 3.94 kcal mol−1. Similarly, 2-chloroanthracene, 2-chloro-1-hydroperoxyanthracene, 1-chloropyrene, 1-chloro-2-hydroperoxypyrene, 4-chloropyrene, and 4-chloro-5-hydroperoxypyrene can be formed by the subsequent reactions of IM13, IM17 and IM22, via an analogous mechanism. The potential barriers of these H migration processes are 28.44–25.26 kcal mol−1 at the BB1K/6-311+G(3df,2p) level, and the processes are exothermic by 1.60–8.39 kcal mol−1.
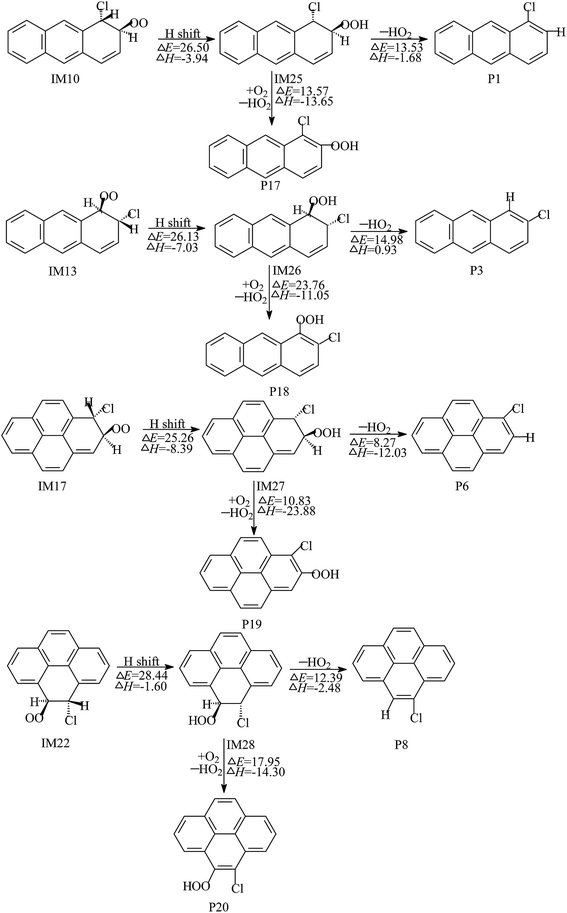 | ||
| Fig. 4 The reaction scheme of the intramolecular H shifts of O2–Cl–Ant and O2–Cl–Pyr adducts, embedded with the potential barrier ΔE (kcal mol−1) and reaction heat ΔH (kcal mol−1). | ||
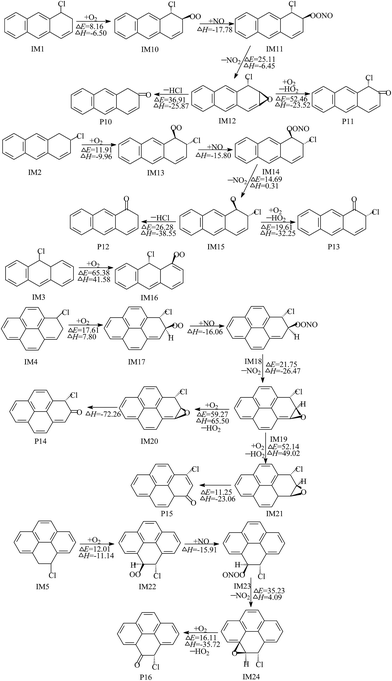
As presented in Fig. 5, the intermediate IM10 undergoes intramolecular cyclization to produce bicyclic peroxy radicals (IM29). IM29 can further proceed with O2 addition, NO barrierless addition and elimination of NO2, resulting in the formation of the important intermediate IM32. The subsequent reaction of IM32 involves three reaction channels: H abstraction by O2 and two different ways of synchronous cleavage of the O–O bond and C–C bond. Obviously, compared with the H abstraction, the ring-opening reactions have lower barriers and release more heat. Therefore, the ring-opening reactions are the energetically more favorable reaction pathways. These processes ultimately result in the formation of a bridge-ring compound (P21) and two dialdehydes (P22 and P23). The reaction channel of IM13 contains three elementary reactions: ring-closure, cleavage of the O–O bond and H abstraction by O2. These reactions are thermodynamically favorable under general atmospheric conditions and lead to the generation of an epoxide (P24). Similarly, IM17 and IM22 can also proceed with ring-closure, ring-opening and H abstraction by O2 to yield epoxides (P25 and P27) and phenalenone radical (P26).
 | ||
| Fig. 6 The formation scheme of nitro-Ants and nitro-Pyrs, embedded with the potential barrier ΔE (kcal mol−1) and reaction heat ΔH (kcal mol−1). ΔH is calculated at 0 K. | ||
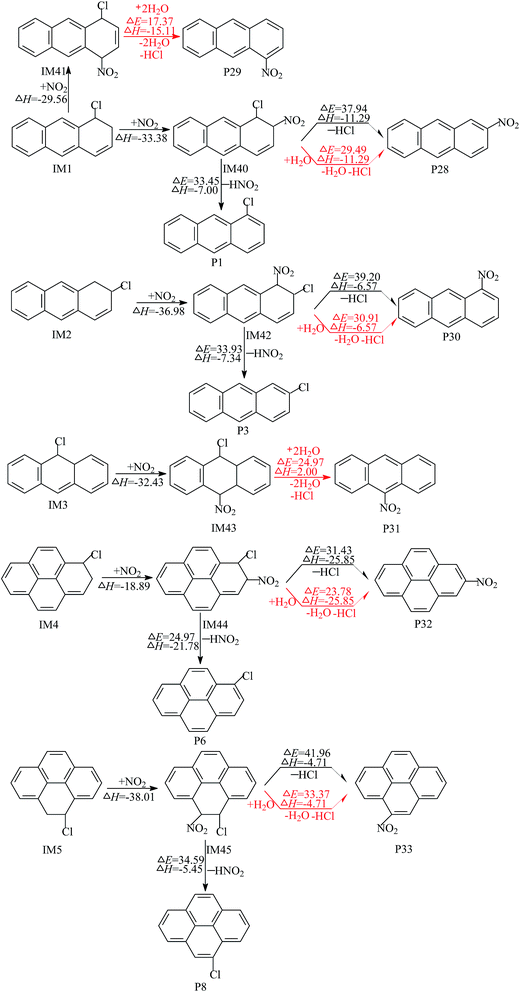
As we all know, water is one of the massive atmospheric constituents and can participate in the reactions through forming hydrogen bonded complexes in the atmosphere.33 With the participation of water, the elimination of HCl from the Cl–NO2–Ant and Cl–NO2–Pyr adducts can become bimolecular reactions (Cl–NO2–Ant + H2O and Cl–NO2–Pyr + H2O). A six-membered ring transition state (Fig. S3 of ESI†) can be formed, in which the water molecule accepts the hydrogen from the aromatic ring and simultaneously donates another hydrogen atom to the Cl atom. The potential barriers of eliminations of HCl and H2O via the bimolecular reactions are 7.65–8.59 kcal mol−1 lower than those of the direct loss of HCl.
Particularly, when Cl atoms add to the C1 atom of Ant, the corresponding Cl–NO2–Ant adducts are hybrids of several resonance structures (shown in Fig. S4 of ESI†). The unpaired electron can be distributed to the ortho and para positions of the –Cl group, therefore NO2 can add to these positions to form the intermediates IM40 and IM41. It was reported that the atmospheric concentration of water dimer is up to 6 × 1014 molecules per cm3 at 292 K.34 As described in Fig. 6 (red arrows), with the aid of the water dimer, 1-nitroanthracene and 9-nitroanthracene can be formed from the elimination of HCl and H2O. The potential barrier of the formation of 1-nitroanthracene is calculated to be 17.37 kcal mol−1 and the process is exothermic by 15.11 kcal mol−1; 9-nitroanthracene is one of the most abundant nitro-PAHs in the atmosphere. Our previous study confirmed that 9-nitroanthracene can be generated by the OH or NO3 radical-initiated gas-phase reactions of Ant, and does not just originate from the primary emissions and heterogeneous reactions.35 As a supplement to our previous study, 9-nitroanthracene also can be produced by the gas-phase reactions of Ant with Cl atoms. Compared with our previous study, the formation of nitroanthracenes and nitropyrenes from the Cl-initiated gas-phase reactions are easier than from the reactions of Ant and Pyr with OH radicals because of the relatively lower barriers.35 For instance, the potential barrier of the formation of 9-nitroanthracene from the OH-initiated reactions of Ant is 30.75 kcal mol−1,35 and the barrier of the corresponding reaction of Ant with OH radicals is 24.97 kcal mol−1.
3.3 Rate constant calculations
For clarifying the atmospheric fates of Ant and Pyr, it is essential to determine accurate rate constants for the elementary reactions involved in the Cl-initiated atmospheric oxidations of Ant and Pyr. On the basis of the BB1K/6-311+G(3df,2p)//BB1K/6-31+G(d,p) energies, the rate constants of the key elementary reactions involved in the Cl-initiated oxidation of Ant and Pyr were evaluated at 298 K and 1 atm using MESMER, which has been successfully applied in the calculation of several similar reactions.28,36 The calculated rate constants for the crucial elementary reactions are listed in Table 1. The individual rate constants for the Cl additions to the C1–H, C2–H and C3–H bonds of Ant are denoted as k1, k2, and k3, respectively. Similarly, the rate constants of the Cl additions to the C1–H and C3–H bonds of Pyr are denoted as k4 and k5, respectively. Considering the molecular symmetries of Ant and Pyr, the overall rate constants for the Cl addition reactions of Ant and Pyr are depicted as ka and kp, where ka = (k1 + k2) × 4 + k3 × 2 and kp = (k4 + k5) × 4, respectively. The calculated overall rate constants of Ant and Pyr (ka and kp) are 5.87 × 10−12 and 2.81 × 10−12 cm3 per molecule per s, respectively, at 298 K and 1 atm. Due to the absence of available experimental information, it is difficult to make a direct comparison of the calculated rate constants with the experimental values for the reactions of Ant and Pyr with Cl atoms. According to the literature, the rate constant for the gas-phase reaction of naphthalene with Cl atoms is ≦0.0091 ± 0.0003 × 10−10 cm3 per molecule per s at 296 K and 735 Torr.13 The calculated rate constants for the reactions of Ant and Pyr with Cl atoms are relatively consistent with the published experimental value of naphthalene. The rate constants of the elementary reactions listed in Table 1 can be of help for the construction of detailed kinetic models describing the atmospheric fates of Ant and Pyr.| Reactions | Rate constants |
|---|---|
| Ant + Cl → Cl–Ant adducts | (ka) 5.87 × 10−12 |
| Ant + Cl → IM1 | (k1) 1.01 × 10−13 |
| Ant + Cl → IM2 | (k2) 8.69 × 10−13 |
| Ant + Cl → IM3 | (k3) 9.93 × 10−13 |
| Pyr + Cl → Cl–Pyr adducts | (kp) 2.81 × 10−12 |
| Pyr + Cl → IM4 | (k4) 5.06 × 10−13 |
| Pyr + Cl → IM5 | (k5) 1.97 × 10−13 |
Based on the overall rate constants for the reactions of Ant and Pyr with Cl atoms (ka and kp), and a detected Cl concentration (CCl) of 4.0 × 105 molecule per cm3 in the marine boundary layer,37 using the expressions as follows,
4. Conclusions and environmental implications
This study presented a comprehensive theoretical study on the reaction mechanisms of the Cl atom-initiated atmospheric oxidations of Ant and Pyr. The individual rate constants were determined. Several conclusions can be drawn from this study as follows:(1) All the Cl addition pathways are highly exothermic with no potential barriers, and they can readily occur under general atmospheric conditions.
(2) The products of the Cl atom-initiated atmospheric oxidations of Ant and Pyr include monochloro-Ants, monochloro-Pyrs, dichloro-Ants, dichloro-Pyrs, 2-chloroanthracen-1-one, 1-chloropyren-2-one, 1-chloropyren-3-one, 4-chloropyren-5-one, 1-chloro-2-hydroperoxyanthracene, 2-chloro-1-hydroperoxyanthracene, 1-chloro-2-hydroperoxypyrene, 4-chloro-5-hydroperoxypyrene, epoxides, dialdehydes, 9-nitroanthracene, 1-nitroanthracene and nitropyrenes. In particular, water plays a crucial role in the gas-phase formation of 9-nitroanthracene, which can be more easily formed by the gas-phase reaction of Ant with Cl atoms than that of Ant with OH radicals.
(3) The calculated overall rate constants of Ant and Pyr (ka and kp) are 5.87 × 10−12 and 2.81 × 10−12 cm3 per molecule per s, respectively, at 298 K and 1 atm. In the marine boundary layer, the atmospheric lifetimes of Ant and Pyr determined by Cl atoms are calculated to be 4.93 and 7.27 days, respectively.
In the troposphere, Ant and Pyr can be transformed by the chemical processes of reactions with OH radicals during the daytime, reactions with NO3 radicals during evening and nighttime, and reactions with Cl atoms in coastal areas. According to the literature, reactions of Ant and Pyr with O3 can be of negligible importance in the atmosphere, due to slow reactions with the aromatic ring.12 The lifetimes for Ant and Pyr, determined by reactions with OH, are in the range of 2–6 hours,12 which are much shorter than that of reaction with Cl atoms in the atmosphere. Although the reaction with OH radicals plays the most important role in the atmospheric oxidations of Ant and Pyr, the reaction with Cl also needs to be understood, due to the high concentration of Cl atoms in the marine boundary layer and in coastal regions. The formation pathway of chlorinated PAHs (more dangerous compounds) in the atmosphere can be obtained from the reaction with Cl atoms, but the simulated results need more experimental data to prove this. To thoroughly clarify the potential risk, further studies are required, with the combination of experimental and theoretical analyses.
Acknowledgements
The study was financially supported by the NSFC (National Natural Science Foundation of China, project No. 21337001, 21377073).References
- F. Chen, W. Hu and Q. Zhong, Atmos. Res., 2013, 124, 53–60 CrossRef CAS.
- S. Christie, D. Raper, D. S. Lee, P. I. Williams, L. Rye, S. Blakey, C. W. Wilson, P. Lobo, D. Hagen and P. D. Whitefield, Environ. Sci. Technol., 2012, 46, 6393–6400 CrossRef CAS PubMed.
- J. H. Seinfeld and S. N. Pandis, Atmospheric Chemistry and Physics, Wiley-Interscience, New York, 2006, ISBN 9471720186 Search PubMed.
- A. Bozlaker, A. Muezzinoglu and M. Odabasi, J. Hazard. Mater., 2008, 153, 1093–1102 CrossRef CAS PubMed.
- M. Nadal, M. Schuhmacher and J. L. Domingo, Environ. Pollut., 2004, 132, 1–11 CrossRef CAS PubMed.
- J. W. Li, X. Shang, Z. X. Zhao, R. L. Tanguay, Q. X. Dong and C. J. Huang, J. Hazard. Mater., 2010, 173, 75–81 CrossRef CAS PubMed.
- T. T. T. Dong and B. K. Lee, Chemosphere, 2009, 74(9), 1245–1253 CrossRef CAS PubMed.
- H. Z. Shen, Y. Huang, R. Wang, D. Zhu, W. Li, G. F. Shen, B. Wang, Y. Y. Zhang, Y. C. Chen, Y. Lu, H. Chen, T. C. Li, K. Sun, B. G. Li, W. X. Liu, J. F. Liu and S. Tao, Environ. Sci. Technol., 2013, 47, 6415–6424 CAS.
- C. E. Boström, P. Gerde, A. Hanberg, B. Jernström, C. Johansson, T. Kyrklund, A. Rannug, M. Törnqvist, K. Victorin and R. Westerholm, Environ. Health Perspect., 2002, 110, 451–488 CrossRef.
- W. L. Xue and D. Warshawsky, Toxicol. Appl. Pharmacol., 2004, 206, 73–93 CrossRef PubMed.
- N. Ramírez, A. Cuadras, E. Rovira, R. M. Marcé and F. Borrull, Environ. Health Perspect., 2011, 119, 1110–1116 CrossRef PubMed.
- I. J. Keyte, R. M. Harrison and G. Lammel, Chem. Soc. Rev., 2013, 42, 9333–9391 RSC.
- L. Wang, J. Arey and R. Atkinson, Environ. Sci. Technol., 2005, 39, 5302–5310 CrossRef CAS PubMed.
- B. Praña, R. Gomperts and J. A. Sordo, Chem. Phys. Lett., 2004, 392, 236–241 CrossRef.
- A. K. Croft, H. M. Howard-Jones, C. E. Skates and C. C. Wood, Org. Biomol. Chem., 2011, 9, 7439–7447 CAS.
- A. Saiz-Lopez and R. V. Glasow, Chem. Soc. Rev., 2012, 41, 6448–6472 RSC.
- P. S. Monks, Chem. Soc. Rev., 2005, 34, 376–395 RSC.
- K. Kakimoto, H. Nagayoshi, Y. Konishi, K. Kajimura, T. Ohura, K. Hayakawa and A. Toriba, Chemosphere, 2014, 111, 40–46 CrossRef CAS PubMed.
- A. Kitazawa, T. Amagai and T. Ohura, Environ. Sci. Technol., 2006, 40(15), 4592–4598 CrossRef CAS PubMed.
- K. Sankoda, K. Nomiyama, T. Yonehara, T. Kuribayashi and R. Shinohara, Chemosphere, 2012, 88(5), 542–547 CrossRef CAS PubMed.
- X. X. Long and J. F. Niu, Chemosphere, 2007, 67(10), 2028–2034 CrossRef CAS PubMed.
- M. Riva, R. M. Healy, P. M. Flaud, E. Perraudin, J. C. Wenger and E. Villenave, J. Phys. Chem. A, 2015, 119(45), 11170–11181 CrossRef CAS PubMed.
- K. F. Ho, S. S. H. Ho, S. C. Lee, Y. Cheng, J. C. Chow, J. G. Watson, P. K. K. Louie and L. W. Tian, Atmos. Environ., 2009, 43, 6343–6351 CrossRef CAS.
- W. L. Ma, Y. F. Li, H. Qi, D. Z. Sun, L. Y. Liu and D. G. Wang, Chemosphere, 2010, 79(4), 441–447 CrossRef CAS PubMed.
- K. Slezakova, D. Castro, A. Begonha, C. Delerue-Matos, M. Alvim-Ferraz, S. Morais and M. Pereira, Microchem. J., 2011, 99, 51–59 CrossRef CAS.
- G. P. Robert, Density functional theory, Annu. Rev. Phys. Chem., 1983, 34, 631–656 CrossRef.
- M. Frisch, G. Trucks, H. B. Schlegel, G. Scuseria, M. Robb, J. Cheeseman, G. Scalmani, V. Barone, B. Mennucci and G. Petersson, Gaussian 09, revision A. 02, Gaussian. Inc., Wallingford, CT, 2009, pp. 270–271 Search PubMed.
- D. R. Glowacki, C. H. Liang, C. Morley, M. J. Pilling and S. H. Robertson, J. Phys. Chem. A, 2012, 116, 9545–9560 CrossRef CAS PubMed.
- L. Vereecken and J. S. Francisco, Chem. Soc. Rev., 2012, 41, 6259–6293 RSC.
- O. Sokolov, M. D. Hurley, T. J. Wallington, E. W. Kaiser, J. Platz, O. J. Nielsen, F. Berho, M.-T. Rayez and R. Lesclaux, J. Phys. Chem. A, 1998, 102(52), 10671–10681 CrossRef CAS.
- G. Löfroth, L. Nilsson, E. Agurell and T. Sugiyama, Mutat. Res., Genet. Toxicol., 1985, 155, 91–94 CrossRef.
- T. Ohura, Sci. World J., 2007, 7, 372–380 CrossRef PubMed.
- V. Vaida and J. D. Simon, Science, 1995, 268, 1443–1448 CAS.
- K. Pfeilsticker, A. Lotter, C. Peters and H. Bösch, Science, 2003, 300, 2078–2080 CrossRef CAS PubMed.
- Q. Z. Zhang, R. Gao, F. Xu, Q. Zhou, G. B. Jiang, T. Wang, J. M. Chen, J. T. Hu, W. Jiang and W. X. Wang, Environ. Sci. Technol., 2014, 48(9), 5051–5057 CrossRef CAS PubMed.
- J. Zhou, J. W. Chen, C.-H. Liang, Q. Xie, Y.-N. Wang, S. Y. Zhang, X. L. Qiao and X. H. Li, Environ. Sci. Technol., 2011, 45, 4839–4845 CrossRef CAS PubMed.
- C. B. Faxon and D. T. Allen, Environ. Chem., 2013, 10, 221–233 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Configuration of the Cl–Ant adducts and Cl–Pyr adducts (IM1–IM5). The O2 addition reaction schemes of Ant and Pyr embedded with the potential barrier ΔE (kcal mol−1) and reaction heat ΔH (kcal mol−1). Configuration of the transition states for the elimination of water and HCl from the Cl–NO2–Ant adducts. TS1: Cl–NO2–Ant → nitro-Ant + HCl, TS2: Cl–NO2–Ant + H2O → nitro-Ant + HCl + H2O, TS3: Cl–NO2–Ant + 2H2O → nitro-Ant + HCl + 2H2O. Resonance structures of the Cl–Ant adduct. See DOI: 10.1039/c5ra25959b |
| This journal is © The Royal Society of Chemistry 2016 |