Improving the flame retardancy and mechanical properties of poly(lactic acid) with a novel nanorod-shaped hybrid flame retardant
Yu Caoa,
Yaqing Jua,
Fenghui Liaoa,
Xiaoxun Jinb,
Xiu Daia,
Jiawei Lia and
Xinlong Wang*a
aSchool of Chemical Engineering, Nanjing University of Science & Technology, Nanjing 210094, China. E-mail: wxinlong323@163.com; Tel: +86 25 8431 5949
bShanghai Institute of Space Propulsion Technology, Huzhou 313000, China
First published on 29th January 2016
Abstract
A novel nanorod-shaped hybrid flame retardant was synthesized by the reaction of benzenephosphinic acid (BPA) with powdery aluminium hydroxide and was analyzed by SEM, XRD, XPS and contact angle tests. Nanocomposites of poly(lactic acid) (PLA) with the flame retardant were prepared. The prepared nanocomposites had good mechanical properties and the tensile strength of the nanocomposite containing 10 wt% nanorods reached 82.51 MPa. Besides, the storage modulus increased about 39.2% for the composites with 10 wt% loading of the flame retardant compared with neat PLA at 25 °C. Meanwhile, the LOI of the nanocomposite was about 25.0% and a V-0 rating in UL-94 was attained in the case of 30 wt% of the flame retardant compared to 20.0% and no rating for pure PLA. SEM observations of the fractured surfaces showed that the nanocomposites had good interfacial interaction, which resulted in the improvement of mechanical properties. The addition of the hybrid flame retardant led to the formation of a coherent and tight char layer as well as the production of PO2˙ radicals and water, which retarded the combustion process of PLA.
Introduction
Poly(lactic acid) (PLA), one of the most promising and important biodegradable candidate polymers, is produced by fermentation from renewable resources such as corn, sugar beets, wheat, and other starch-rich products.1–3 Initially, PLA was mainly applied in packaging, agriculture and biomedical fields for its biodegradable properties. At present, as a valuable biosourced polymer alternative in long-term applications such as electronics and automobile, PLA is gaining more and more attention.4–6 When used in these fields, some properties of PLA such as good mechanical properties and flame retardancy are usually required by different end-use sectors.7–10Adding nanofillers represents an interesting and effective way to improve the properties of PLA. The nanocomposites of PLA have been reported with various nanoparticles, including carbon nanotubes,11 layered silicates or clays,5,9 silica,12 graphite,13 etc. Ray et al. first prepared PLA/layered silicate nanocomposites by incorporating the organically modified montmorillonite to improve the material's properties.14 Kim et al. discussed the influence of exfoliated graphite (EG) on the thermal stability, mechanical, and electrical properties of PLA/EG nanocomposites. They discovered that Young's modulus increased noticeably with the increase of EG content up to 2 wt% and the thermal degradation temperature of nanocomposite with only 0.5 wt% EG was improved by 10 K over neat PLA.15 Serge Bourbigot prepared the PLA nanocomposites containing multi-wall carbon nanotube (MWNT) by reactive extrusion and the reaction to fire of PLA nanocomposite shows the improvement of the flame retardancy.16 Ton Peijs developed a PLA composite with improved flame retardancy utilizing the unique properties of sepiolite nanoclay and multiwalled nanotubes.17
Up to now, the flame retardant aluminum hydroxide (ATH) has gained much attention because of its environmental friendliness, low cost, low toxicity and flame retardancy.18,19 However, the large limitation of ATH is difficult to disperse uniformly in the organic polymers due to the incompatibility in surface characteristics between the hydrophobic polymer and hydrophilic filler surface.20 Therefore, the physical and mechanical properties of the polymer will be affected by adding too much flame retardant. To improve this issue, modifying ATH with some organic coupling agents becomes a common choice, but the procedures involve the use of coupling agents and chemical reactions which can bring about the enhancement of cost and environmental problems. Besides, many coupling agents are combustible and lead to the difficulty in the enhancement of the flame retardance. Recently, there has been considerable interest in the development of organic–inorganic hybrid materials in the chemistry and physics fields. A hybrid material is obtained by combining at least two components, commonly inorganic and organic, at the nanometer or molecular level. Mixing at the microscopic scale leads to an intriguing class of materials that either shows characteristics in between the two original phases or even new properties. The hybrid materials have broad potential applications as nano fillers.21,22 Methods to synthesize inorganic–organic hybrid materials are often based on soft chemistry approaches, such as sol–gel process, intercalation, exchange, or grafting. Benzenephosphinic acid (BPA) is not applied in PLA as an additive flame retardant because the carboxyl group in BPA could react with PLA during melt processing and in use, which results in the depolymerization of PLA molecules and the deterioration to the performances of the PLA composites such as mechanical properties.23 Therefore, BPA is often used as a reactive flame retardant. Because of the limitations of ATH and inspired by the organic–inorganic hybrid materials, we used grafting techniques to prepare a novel nano rod-shaped hybrid flame retardant composed of ATH and BPA components by ATH reacting with BPA in this study. The flame retardant was compounded into PLA matrix to prepare the nanocomposites of PLA. The prepared nanocomposites showed good mechanical properties and flame retardancy compared with neat PLA.
Experimental
Materials
Poly(lactic acid) (PLA 290, injection molding grade) was purchased from Zhejiang Haizheng Biological Materials Co. Ltd (Zhejiang, China) in the form of pellets. Aluminium hydroxide was obtained from Aladdin Industrial Corporation (Shanghai, China). Benzenephosphinic acid was supplied by Tokyo Kasei Kogyo Co. Ltd. (TCI, Japan). Other reagents were all of analytical grade.Preparation of the novel nano rod-shaped hybrid flame retardant (NRH-FR)
A certain amount of ATH and acetic acid were added into the beaker, followed by ultrasonic shaking for 30 min. The mixture was transferred into round flask. Then benzenephosphinic acid was added into the above flask and stirred at 80 °C under nitrogen for 9 h. After the reaction, the white nano organic–inorganic hybrid rods were collected by centrifuging, washed with water and ethanol, and dried at 50 °C for 12 h in vacuum.Preparation of the PLA nanocomposites
The NRH-FR was compounded with PLA in a RM-200A torquerheometer (Hapro Electric Technology Co. Ltd. Harbin, China) with three heating zones of 175, 175 and 175 °C, respectively. The obtained mixture was injection molded into appropriate specimens for tests by a SZ-15 micro injection molding machine (Ruiming Plastics Machinery Co. Ltd. Wuhan, China) at 185 °C. The formulations under investigation were listed in Table 1.| Samples | PLA (wt%) | NRH-FR (wt%) | LOI (%) | UL-94 | |
|---|---|---|---|---|---|
| Dripping | Rating | ||||
| a NRa not rated. | |||||
| P0 | 100 | 0 | 20.0 | Yes | NRa |
| PBA1 | 90 | 10 | 21.5 | No | V-1 |
| PBA2 | 85 | 15 | 22.5 | No | V-1 |
| PBA3 | 80 | 20 | 23.0 | No | V-0 |
| PBA4 | 70 | 30 | 25.0 | No | V-0 |
| PBA5 | 60 | 40 | 25.5 | No | V-0 |
Measurement and characterization
The morphology of the samples was analyzed by transmission electron microscopy (TEM, JEOL 2100, Japan). X-ray photoelectron spectroscopy (XPS) was obtained using a PHI5300 spectrometer (ELMER, USA) with Al Kα as the excitation radiation. X-ray diffraction (XRD) measurements were performed on a D8 ADVANCE diffractometer (Bruker, Germany) using Cu Kα generator (λ = 0.15418 nm) at 40 kV. Thermogravimetric analysis (TGA) was carried out from ambient temperature to 600 °C or 800 °C on a DGT-60 thermo-analyzer instrument (Shimadzu, Japan) in nitrogen or oxygen flow (20 mL min−1) at a heating rate of 20 °C min−1. Contact angle measurement was conducted on a SL200B goniometer (Kino, America). It was investigated by the sessile drop method with water as the solvent. Dynamic mechanical analysis (DMA) (storage modulus and tan delta (δ) as a function of temperature) was performed on a DMA Q800 dynamic mechanical analyzer (TA Corporation, America) equipped with a dual-cantilever clamp and the samples with dimensions of 60 mm × 10 mm × 4 mm were used for testing at temperatures ranging from −20 to 150 °C at a heating rate of 3 °C min−1 and a frequency of 1 Hz. The mechanical properties of the nanocomposites were measured on a CMT tensile tester (Sans, China) with the dumbbell-shaped test specimens molded in accordance with ASTM D638. Limiting oxygen index (LOI) tests were conducted with the sample dimension of 100 mm × 6.5 mm × 3 mm according to ASTM D2863-97 on an HC-2 oxygen index meter (Jiangning Analysis Instrument Co., China). UL 94 burning tests were performed on the samples of 130 mm × 13 mm × 3 mm in size with CFZ-3 instrument (Jiangning Analysis Instrument Co., China) according to ASTM D3801-96.Results and discussion
Synthesis and characterization of NRH-FR
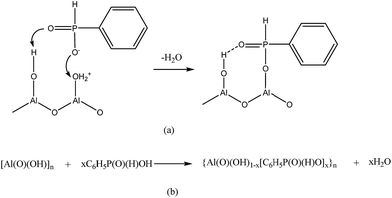
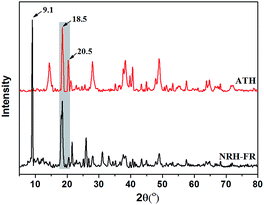
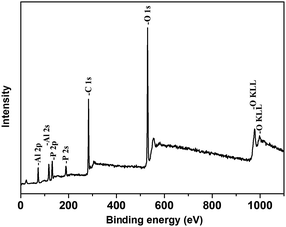
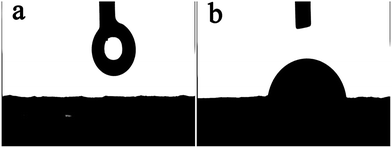
Fig. 1(a) shows the TEM image of raw ATH and it can be seen that the raw materials are powdery without the specific shape. After ATH reacts with BPA, the formed nano organic–inorganic rods can be seen in Fig. 1(b). These nano rods have a uniform length of 0.6 μm and a diameter of less than 100 nm. The surface modification of ATH with phosphinic acids has a balance between grafting and the formation of bulk aluminium phosphinite phases. However, the grafting reaction is markedly favoured in organic media such as in acetic acid. The grafting involves both the formation of hydrogen bond between the phosphoryl oxygen and surface Al–OH groups, and the condensation of the P–OH functions with surface Al–OH groups as in Scheme 1(a).24 The reaction of ATH with BPA could also leads to the cleavage of hydrogen bonds between octahedral layers of ATH and to the replacement of hydroxyl groups by bidentate benzenephosphinite ligands, causing exfoliation of the ATH layers without damaging the Al–O–Al linkages as in Scheme 1(b). Therefore, the formed rods consist of the ATH-like core decorated with benzenephosphinite groups.25 This can be further examined by X-ray diffraction. The XRD results are shown in Fig. 2. We can see that the two patterns all have two peaks at 2θ of about 18.5°, 20.5° corresponding to the characteristic diffraction adsorptions of ATH. However, compared with raw ATH, the spectrum of the nano rods is found to have the new peak appeared at about 9.1° and the peak at 20.5° is near-disappeared, thus supporting that a significant change has happened in crystal structure of ATH because of hybridization.26The hybrid structure can be further investigated by XPS and contact angle tests. Fig. 3 shows XPS of NRH-FR and all the peaks on the curves can be ascribed to Al, P, O and C elements. The results of XPS indicate the presence of BPA unit in NRH-FR. The contact angle results for raw ATH and NRH-FR are displayed in Fig. 4. We cannot identify the contact angle of neat nano ATH pallets because of its water-absorbing nature. After grafting with BPA, we can clearly see that the contact angle of NRH-FR reaches 45°, indicating that NRH-FR becomes more hydrophobic than raw ATH because of the organic groups in BPA. The prepared nano rods could be more compatible with PLA.27
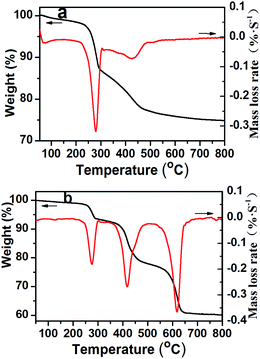
Fig. 5 shows the TGA and DTG results of ATH and NRH-FR. The endothermic decomposition of ATH occurs at 196.2 °C and leads to the release of water and the formation of alumina. As shown in Fig. 5, this reaction tends to occur in two stages, which corresponds to two endothermic transitions. The intermediate product formed is known as boehmite “AlOOH”, which attributes to the much lower endothermic process energy.28 For NRH-FR, its initial decomposition temperature is increased to 220.5 °C and this means that NRH-FR is more stable than ATH. The improvement in thermal stability of ATH is beneficial to its application in polymers because the compounding of fillers and polymers is usually performed by melt blending methods. Compared with the two-stage decomposition processes, the reaction of NRH-FR occurs in three stages. The first and second processes are due to the decomposition of ATH and partially decomposition of BPA. The third process is resulted from the further decomposition of the residues of BPA produced in the first and second processes.
SEM of the PLA nanocomposites
The cross section scan of the PLA nanocomposites is observed under SEM and the micrographs are demonstrated in Fig. 6. In the present case, both in Fig. 6(a) and (b), the micrographs show dense planes and there are no micro cavities or agglomerated particles existed in the matrix. As mentioned earlier, the NRH-FR possesses a lot of organic groups that can increase interfacial strength between the fillers and PLA matrix.15Mechanical properties of the PLA nanocomposites
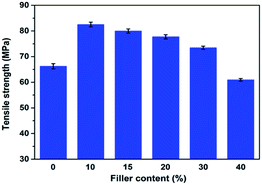
The results of tensile strength for the prepared nanocomposites are shown in Fig. 7. It can be seen that when appropriate amount of NRH-FR is added, the nanocomposites show better tensile strength than that of neat PLA. For instance, the PBA1, with the addition of 10 wt% NRH-FR, shows the tensile strength of 83 MPa, increased by 25.8% compared with the pure PLA, 66 MPa. First, the hydrophobicity of the organic groups in NRH-FR has an impact on the improvement of compatibility between PLA and NRH-FR, which leads to good dispersion of NRH-FR in PLA. Second, the high aspect ratio of NRH-FR can entrap a great amount of PLA chains on the surface, which ultimately endows the good filler/matrix interactions and the improved properties to the nanocomposites. With the increasing content of NRH-FR in the nanocomposites further, the prepared nanocomposite such as PBA4 and PBA5 show lower tensile strength, which can be owing to the fact that the filler aggregation could be caused by the large amount of junctions existing in the matrix, making the micro cracks develop into macro cracks. The aggregation of fillers and the macro cracks cause the prepared nanocomposites to rupture easily.10,29Dynamic mechanical analysis of the PLA nanocomposites
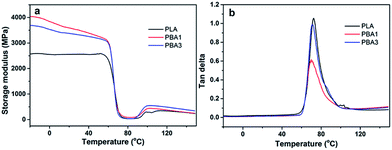
The temperature dependence of storage modulus (a) and tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ (b) from DMA experiments are presented in Fig. 8. Storage modulus is related to the load-bearing capacity of the materials. As indicated in Fig. 8(a), the storage modulus of pure PLA and the PLA nanocomposites shows a gradual decrease from −10 to 60 °C, and then decreases rapidly due to the glass transition. There is an obvious increase in storage modulus after the addition of NRH-FR into the PLA matrix below 60 °C. For instance, the storage modulus increases up to 39.2% and 31.4% in the case of 10 wt% and 20 wt% NRH-FR loading, respectively, compared with neat PLA at 25 °C. This could be attributed to the fact that NRH-FR has high specific surface area and hydrophobicity which lead to strong interfacial interactions between PLA matrix and fillers. It is also observed that the storage modulus is a little bit lower when the loading amount increases and this may be due to the agglomeration effect of NRH-FR. As can be observed from Fig. 8(b), the tan
δ (b) from DMA experiments are presented in Fig. 8. Storage modulus is related to the load-bearing capacity of the materials. As indicated in Fig. 8(a), the storage modulus of pure PLA and the PLA nanocomposites shows a gradual decrease from −10 to 60 °C, and then decreases rapidly due to the glass transition. There is an obvious increase in storage modulus after the addition of NRH-FR into the PLA matrix below 60 °C. For instance, the storage modulus increases up to 39.2% and 31.4% in the case of 10 wt% and 20 wt% NRH-FR loading, respectively, compared with neat PLA at 25 °C. This could be attributed to the fact that NRH-FR has high specific surface area and hydrophobicity which lead to strong interfacial interactions between PLA matrix and fillers. It is also observed that the storage modulus is a little bit lower when the loading amount increases and this may be due to the agglomeration effect of NRH-FR. As can be observed from Fig. 8(b), the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ of pure PLA shows the highest damping. The incorporation of NRH-FR decreases the tan
δ of pure PLA shows the highest damping. The incorporation of NRH-FR decreases the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ peak intensity compared to that of pure PLA, which reflects an increased stiffness of the material at the glass transition region. The glass transition temperature itself is nearly unaffected by the presence of NRH-FR, which may be due to the existence of organic moieties in NRH-FR.30
δ peak intensity compared to that of pure PLA, which reflects an increased stiffness of the material at the glass transition region. The glass transition temperature itself is nearly unaffected by the presence of NRH-FR, which may be due to the existence of organic moieties in NRH-FR.30
Thermogravimetric analysis of the PLA nanocomposites
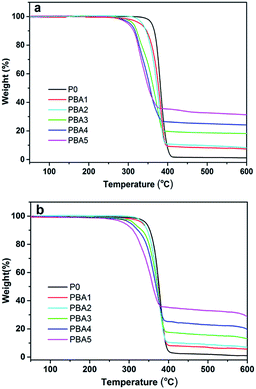
Thermogravimetric analysis of the PLA nanocomposites is performed in nitrogen and air conditions. Thermal degradation behaviors of pure PLA and the PLA nanocomposites in nitrogen condition are presented in Fig. 9(a). It is observed that PLA decomposes in one stage process and T10 (the temperature of 10% weight loss) of PLA is about 346.5 °C. As shown in Fig. 9(a), all the nanocomposites also show a one-step weight loss with the lower T10s than that of PLA. The reduction in the thermal stability of nanocomposites with addition of NRH-FR is owing to the lower stability of P–C bonds in BPA compared to common C–C bonds.31 The TGA in air condition is shown in Fig. 9(b). Similar phenomena are also found in the nanocomposites. It is interesting to find that in both the nitrogen and air conditions, the addition of NRH-FR could significantly increase the char residues. For example, the residual char of PBA3 is 18.16 wt%, while the value of pure PLA is only 1.20 wt% in nitrogen, indicating the char formed at elevated temperature has better thermal stability.32 This may be attributed to that the presence of NRH-FR which could promote the char formation.Flammability behavior of the PLA nanocomposites and the mechanism consideration
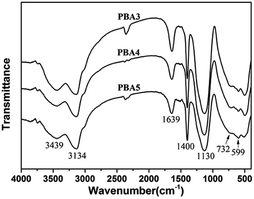
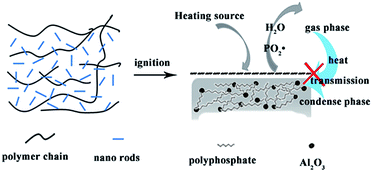
Underwriters Laboratory (UL-94) and limited oxygen index (LOI) tests are two of the most widely used tests to characterize the fire performance of polymer composites. The LOI test is one of the most important screening and quality control methods used in the plastics industry. The UL-94 tests, developed by the Underwriters Laboratories, is the most common procedure to check the regulatory compliance of plastic materials for parts in devices and appliances.33 The LOI and UL-94 results are summarized in Table 1. PLA is a flammable polymeric material and the melt dripping is serious during the combustion of PLA. As we know, melt dripping usually causes another fire accident. The LOI values of the nanocomposites show improvement with addition of NRH-FR. In the case of 30 wt% fillers (PBA4), the LOI of the nanocomposite reaches 25.0%. Furthermore, in UL-94 tests, the rating reaches V-0 without dripping, which is very satisfactory for PLA composites when they are applied in electronic or automobile industry.The morphology of char residues for the nanocomposites is examined by SEM and shown in Fig. 10. The SEM image at 50 μm shows that the char surface is coherent and tight, indicating the formation of a dense and coherent char layer which shields the underlying polymers from attack by oxygen and radiant heat during the burning process.34 Fig. 11 is the FTIR spectra of char residues left by the burning of the nanocomposites. It can be observed that the peak at 1400 cm−1 belongs to aromatic ring, and the absorption peak observed at 1130 cm−1 could be assigned to PO2−. The peak centred at 732 cm−1 and 599 cm−1 is assigned to the stretching and deformation vibrations of Al–O structures, respectively. It can be concluded that the char residues of the nanocomposites are composed of the polyaromatic char, Al2O3 particles and pyrophosphate or phosphate structure, which has good stability during the combustion process.35 The probable flame retardant mechanism is presented in Fig. 12. On one hand, the BPA units in NRH-FR releases PO2˙ radical which can inhibit free radical reactions in gas phase and forms polyphosphates remaining in condensed phase which reduces the heat transferring.36 On the other hand, the ATH units in NRH-FR give off water vapor during burning which lowers temperature of the condensed phase and dilutes the concentration of combustible specimens in the gas phase. ATH can also be decomposed into Al2O3 particles and the polyphosphates from the decomposition of BPA crosslink the Al2O3 particles to form coherent char layer in condense phase. All these are beneficial to the flame retardancy of the PLA nanocomposites.
Conclusions
The nano organic–inorganic rod flame retardant with a uniform diameter of less than 100 nm was synthesized by the reaction of benzenephosphinic acid (BPA) with ATH. The NRH-FR had good thermal stability and hydrophobic nature. As an inorganic–organic hybrid, the NRH-FR had good dispersion in PLA matrix owing to the presence of organic units, which induced the reinforcement of NRH-FR crystals to the nanocomposites and increased the storage modulus and tensile strength of the nanocomposites. Besides, it could also improve the flame retardancy of the nanocomposites because the formed char residue could effectively prevent the heat and oxygen approaching the PLA matrix and the produced PO2˙ radical and water could inhibit combustion process. These made the PLA nanocomposites attractive for the manufacture of electrical/electronic devices or automotive parts.Acknowledgements
This work was subsidized by a project funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions and the Science and Technology Support Program (Social Development) of Jiangsu Province of China (BE 2013714).Notes and references
- J. R. Jambeck, R. Geyer, C. Wilcox, T. R. Siegler, M. Perryman, A. Andrady, R. Narayan and K. L. Law, Science, 2015, 347, 768–771 CrossRef CAS PubMed.
- B. U. Nam, K. D. Min and Y. Son, Mater. Lett., 2015, 150, 118–121 CrossRef CAS.
- Y. Dong, A. Ghataura, H. Takagi, H. J. Haroosh, A. N. Nakagaito and K. T. Lau, Composites, Part A, 2014, 63, 76–84 CrossRef CAS.
- G. Tang, X. Wang, W. Xing, P. Zhang, B. Wang, N. Hong, W. Yang, Y. Hu and L. Song, Ind. Eng. Chem. Res., 2012, 51, 12009–12016 CrossRef CAS.
- L. Jiang, J. Zhang and M. P. Wolcott, Polymer, 2007, 48, 7632–7644 CrossRef CAS.
- F. J. Li, S. D. Zhang, J. Z. Liang and J. Z. Wang, Polym. Adv. Technol., 2015, 26, 465–475 CrossRef CAS.
- N. A. Isitman, M. Dogan, E. Bayramli and C. Kaynak, Polym. Degrad. Stab., 2012, 97, 1285–1296 CrossRef CAS.
- H. Zhao, Z. Cui, X. Wang, L. S. Turng and X. Peng, Composites, Part B, 2013, 51, 79–91 CrossRef CAS.
- A. González, A. Dasari, B. Herrero, E. Plancher, J. Santarén, A. Esteban and S. H. Lim, Polym. Degrad. Stab., 2012, 97, 248–256 CrossRef.
- K. Das, S. Sinha Ray, S. Chapple and J. Wesley-Smith, Ind. Eng. Chem. Res., 2013, 52, 6083–6091 CrossRef CAS.
- T. Villmow, P. Pötschke, S. Pegel, L. Häussler and B. Kretzschmar, Polymer, 2008, 49, 3500–3509 CrossRef CAS.
- Y. Li, C. Han, J. Bian, L. Han, L. Dong and G. Gao, Polym. Compos., 2012, 33, 1719–1727 CrossRef CAS.
- H. Zhu, Q. Zhu, J. Li, K. Tao, L. Xue and Q. Yan, Polym. Degrad. Stab., 2011, 96, 183–189 CrossRef CAS.
- S. Sinha Ray, P. Maiti, M. Okamoto, K. Yamada and K. Ueda, Macromolecules, 2002, 35, 3104–3110 CrossRef.
- I. H. Kim and Y. G. Jeong, J. Polym. Sci., Part B: Polym. Phys., 2010, 48, 850–858 CrossRef CAS.
- S. Bourbigot, G. Fontaine, A. Gallos and S. Bellayer, Polym. Adv. Technol., 2011, 22, 30–37 CrossRef CAS.
- T. D. Hapuarachchi and T. Peijs, Composites, Part A, 2010, 41, 954–963 CrossRef.
- H. Nishida, Y. Fan, T. Mori, N. Oyagi, Y. Shirai and T. Endo, Ind. Eng. Chem. Res., 2005, 44, 1433–1437 CrossRef CAS.
- J. Z. Liang, Y. Chen and X. H. Jiang, Polym.-Plast. Technol. Eng., 2012, 51, 439–445 CrossRef CAS.
- J. C. Liu, J. H. Jean and C. C. Li, J. Am. Ceram. Soc., 2006, 89, 882–887 CrossRef CAS.
- D. S. Achilleos and M. Vamvakaki, Materials, 2010, 3, 1981–2026 CrossRef CAS.
- N. Kojima, M. Enomoto, N. Kida and K. Kagesawa, Materials, 2010, 3, 3141–3187 CrossRef CAS.
- H. J. Lin, S. R. Liu, L. J. Han, X. M. Wang, Y. J. Bian and L. S. Dong, Polym. Degrad. Stab., 2013, 98, 1389–1396 CrossRef CAS.
- G. Gilles, M. P. Hubert and V. Andre, J. Mater. Chem., 2001, 11, 3161–3165 RSC.
- F. Zbigniew, L. Anna, W. Andrze and Z. Janus, Chem. Mater., 2006, 18, 1995–2003 CrossRef.
- G. Lefevre and M. Fedoroff, Mater. Lett., 2002, 56, 978–983 CrossRef CAS.
- Y. Xie, C. A. Hill, Z. Xiao, H. Militz and C. Mai, Composites, Part A, 2010, 41, 806–819 CrossRef.
- T. R. Hull, A. Witkowski and L. Hollingbery, Polym. Degrad. Stab., 2011, 96, 1462–1469 CrossRef CAS.
- F. Zheng, S. Wang, S. Wen, M. Shen, M. Zhu and X. Shi, Biomaterials, 2013, 34, 1402–1412 CrossRef CAS PubMed.
- M. S. Huda, L. T. Drzal, A. K. Mohanty and M. Misra, Compos. Sci. Technol., 2008, 68, 424–432 CrossRef CAS.
- N. Tian, J. Gong, X. Wen, K. Yao and T. Tang, RSC Adv., 2014, 4, 17607–17614 RSC.
- S. P. Liu, J. Ind. Eng. Chem., 2014, 20, 2401–2408 CrossRef CAS.
- R. Dupretz, G. Fontaine, S. Duquesne and S. Bourbigot, Polym. Adv. Technol., 2015, 26, 865–873 CrossRef CAS.
- Z. Wang, P. Wei, Y. Qian and J. Liu, Composites, Part B, 2014, 60(341), 341–349 CrossRef CAS.
- A. Marchal, R. Delobel, M. Le Bras, J. M. Leroy and D. Price, Polym. Degrad. Stab., 1994, 44, 263–272 CrossRef CAS.
- S. Takayama, N. Mathubara, T. Arai and K. Takeda, Polym. Degrad. Stab., 1995, 50, 277–284 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2016 |