Pseudo-first order reaction kinetics and thermodynamic properties study of neem oil esterification using MgO grafted natural hydroxyapatite
Himadri Sahu and
Kaustubha Mohanty*
Department of Chemical Engineering, Indian Institute of Technology Guwahati, Guwahati 781039, India. E-mail: kmohanty@iitg.ernet.in; Fax: +91-361-2582291; Tel: +91-361-2582267
First published on 15th January 2016
Abstract
In this work, waste fish bone was used as a source of natural hydroxyapatite which was later used for the preparation of a metal grafted catalyst. Calcium phosphate is the main component of fish bone which has a relatively high catalytic activity, good thermal and chemical stability, and can be used for various applications. Magnesium oxide (MgO) was used for grafting due to its high specific surface area. It can be used as the active phase during catalysis which influences the selectivity and activity of catalytic reactions. In the present investigation, the waste fish bone-derived catalysts were characterized in detail and used for the esterification of neem oil (Azadirachta indica). The highest methyl ester yield of 96.7% was obtained at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 oil to methanol ratio, 70 °C reaction temperature and 6 wt% catalyst loading. The catalytic effect on yield and reusability of the catalyst was also studied. Subsequently, a kinetic model was developed and the thermodynamic properties of the process are illustrated. Overall the whole work gives a new direction towards the development of heterogeneous catalysts from bio wastages.
15 oil to methanol ratio, 70 °C reaction temperature and 6 wt% catalyst loading. The catalytic effect on yield and reusability of the catalyst was also studied. Subsequently, a kinetic model was developed and the thermodynamic properties of the process are illustrated. Overall the whole work gives a new direction towards the development of heterogeneous catalysts from bio wastages.
1. Introduction
Globally in excess of ninety-one million tons of fish are caught annually and around 55% of these are used for human consumption, while the rest are discarded.1 Most of the fish used for human consumption are deboned and exported. The bones are usually discarded without any further use and thus create an undesirable impact on the environment. These bones are a rich source of calcium phosphate, popularly also known as hydroxyapatite (HAp), with a stoichiometric formula Ca10(PO4)6(OH2). It is therefore advisable to use this low-cost source of HAp for some meaningful purpose which will address the waste management problem also.There are few differences between synthetic and natural hydroxyapatite. Natural HAp has better dynamic response to the environment, high catalytic activity and good thermo-chemical stability than the synthetic HAp.2,3 As far as solubility and activity is concerned, crystallographic structure plays an important role. In line to synthetic HAp, the natural HAp has perturbed nanostructures and nonstoichiometric composition with low hydroxyl content. As physical and chemical properties depend on structure, natural HAp will lead to good results.4 Because of these properties HAp can be used as a heterogeneous catalyst. Although natural HAp has favorable structure but the BET surface area is bit low, which can be increased by grafting metal ions on to it. Venkatesan et al. developed nano-hydroxyapatite from salmon fishbone and was characterized using various techniques. From the analysis it is clearly shown that it has potential to give a metal support on it. The SAED pattern showed the crystalline nature of the material.5 Sasaki and Goto also extracted hydroxyapatite from four types of fish bone and compared with commercial grade hydroxyapatite. They showed ion-exchange efficiency, but however application in reaction field was missing.6 Chakraborty and RoyChowdhury tried to dope Cu2+ on hydroxyapatite derived from waste fishbone and the heterogeneous catalyst was used for esterification of oleic acid. However, the catalyst has very low surface area, pore volume as well as very poor thermal stability. In the literature it was mentioned that there are uniform pores but the SEM image shows flat surface at 5 μm range which is contradictory. Although above 90% conversion of oleic acid was shown but the effect on mixture of fatty acid i.e. vegetable oil was not discussed.7 Considering all these aspects, the objective of the present work is decided.
In the present work, natural HAp collected from waste fish bone was used as a base material for metal grafting to carry out esterification of neem (Azadirachta indica) oil. From literature, it was found that MgO alone was proved to be a very good catalyst in esterification reaction.8–11 Therefore Mg metal ion was grafted on to the natural HAp by wet impregnation method followed by calcination to increase the surface area of HAp as well as it's catalytic activity. In this grafted catalyst both the HAp and MgO will act as an active source for the adsorption of reactants during the reaction.
2. Materials and detailed methodology
2.1 Materials
From IIT Guwahati hostel messes cooked waste fish bone was collected. Few chemicals viz. 25% aqueous ammonia solution, methanol and magnesium sulfate (MgSO4·7H2O) were procured from Sigma Aldrich, India and were used as it is without any further treatment.2.2 Preparation methodology
Fish bone was washed properly to remove proteinaceous part and sun dried for 2 days. After that oily matters was removed completely by dipping in acetone for 24 h and dried at 60 °C. The fishbone was crushed and natural hydroxyapatite (NHAp) was obtained. Magnesium sulfate was used as the precursor for metal ions during wet impregnation.12 Following the method, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 wt proportions of NHAp and MgO were prepared. 30 g of MgSO4·7H2O was added to 100 mL of water to get a solution of magnesium sulfate. To this solution, 30 g of fine powder of NHAp was added gradually and blended rigorously using a mechanical agitator under aggregate reflux time of 30 min. 25% aqueous ammonia solution was used to maintain pH at 11. The entire solution was mechanically stirred for 2 h at 700 rpm at 60 °C. The final solution was kept for 24 h at room temperature so as to allow the deposition of magnesium ions on NHAp. After that mother liquor was vacuum filtered and the sticky precipitate obtained was dried in hot air oven. The dried mass was then calcined at 650 °C for 3 h and final Mg grafted NHAp (Mg-NHAp) was sent for characterization.
1 wt proportions of NHAp and MgO were prepared. 30 g of MgSO4·7H2O was added to 100 mL of water to get a solution of magnesium sulfate. To this solution, 30 g of fine powder of NHAp was added gradually and blended rigorously using a mechanical agitator under aggregate reflux time of 30 min. 25% aqueous ammonia solution was used to maintain pH at 11. The entire solution was mechanically stirred for 2 h at 700 rpm at 60 °C. The final solution was kept for 24 h at room temperature so as to allow the deposition of magnesium ions on NHAp. After that mother liquor was vacuum filtered and the sticky precipitate obtained was dried in hot air oven. The dried mass was then calcined at 650 °C for 3 h and final Mg grafted NHAp (Mg-NHAp) was sent for characterization.
2.3 Catalyst, raw neem oil (RNO) and esterified neem oil (TNO) characterization
Surface analyzer device (Beckman-Coulter; model: SA3100) was used to measure BET surface area, average pore measurement and pore volume (t-plot strategy) by physical adsorption–desorption of N2 at the boiling point (77 K). Preceding the examination, Mg-NHAp and NHAp were subjected to a preheating for 90 min at 423 K under vacuum.
A thermal analyzer (NETZSCH: STA449F3 Jupiter) was used for the thermogravimetric analysis of the powder sample. Argon was passed through the instrument with a heating rate of 10 K min−1 using a ceramic crucible up to 1000 °C.
A scanning electron microscope (JEOL-JSM-6390LV) furnished with an EDAX PV 9760 detector for Energy Dispersive X-ray spectroscopy (EDX) was used to investigate nearby chemical arrangement. The samples were dispersed in methanol and deposited on an aluminium foil before mounting on sample holder. It was then coated with gold–palladium to make the specimen fit for microstructures investigations.
Fourier transform infrared spectroscopy (FTIR) provides information with respect to various functional groups present in the sample. DRS connected with Excalibur Bio-Rad spectrophotometer (model FTS 3500 GX) FTIR analyzer was utilized for the same. The IR range was between 400 and 4000 cm−1 at a scan rate of 40 and at a step size of 4 cm−1.
2.4 Procedure for esterification of neem oil using Mg-NHAp
Activity of Mg-NHAp was studied by esterification of neem oil in a batch reactor having conventional heating framework. For a standard analysis, 30 g of neem oil, methanol and catalyst (2 wt%) were taken in a 3-necked glass reactor of 1 L volume and was heated up to 70 °C and then agitated for a particular time period. After that the entire reaction mixture was transferred to a rotavapor for removing excess methanol. The reaction mixture was then centrifuged to separate the catalyst and the mixture was kept in a separating funnel overnight to separate the glycerol produced during the reaction. The methyl ester yield was calculated by gas chromatography. Following the standard method, effect of oil to methanol ratio and catalyst loading on methyl ester yield with respect to reaction time was also studied. To study the kinetics during the reaction progress, small volume of samples was withdrawn negligible to the whole reaction mixture. The methyl ester yield was noted at regular interval of time.2.5 Reaction kinetics study of the process
The esterification reaction is reversible and the stoichiometric relation between reactant and product is presented below.| fa + me ⇔ fame + w | (1) |
To fit the data in the kinetic model few assumptions were made, as given below.
• In the whole reactant mixture, fatty acid content was limiting and methanol content was in excess.
• The esterification reaction is a heterogeneous process, however the stirring rate is adequate to overcome the diffusion limitation in between reactive species and catalyst.15
Considering the above, the generalized rate equation can be written as,
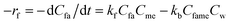 | (2) |
So, according to 1st assumption, Cme ≈ Cme0
Thus, eqn (2) can be written as,
 | (3) |
If χfa is the fatty acid conversion, Cfa0, Cfame0, Cw0 are initial concentrations of respective reactants then Cfa, Cfame, Cw can be expressed as,
| Cfa = Cfa0(1 − χfa) | (4) |
| Cfame = Cfame0 + Cfa0χfa | (5) |
| Cw = Cw0 + Cfa0χfa | (6) |
As initially Cfame0 = Cw0 = 0, Cfame and Cw can be written as Cfame = Cfa0χfa and Cw = Cfa0χfa.
Now eqn (3) can be expressed as,
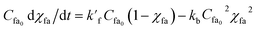 | (7) |
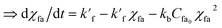 | (8) |
When equilibrium state is reached, then 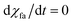 .
.
Eqn (8) thus becomes,
| k′f(1 − χfae) = kbCfa0χfae2 | (9) |
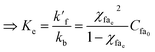 | (10) |
Now, eqn (8) is rearranged as
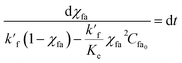 | (11) |
 | (12) |
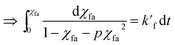 | (13) |

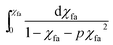 vs. t is plotted and by linear fit the k′f can be calculated.
vs. t is plotted and by linear fit the k′f can be calculated.
The rate of esterification reaction is higher at lower activation energy. Hence the reaction temperature effect on kinetic model was studied by using Arrhenius equation which is listed as:
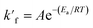 | (14) |
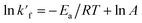 | (15) |
3. Results and discussions
3.1 Characterization of Mg-NHAp catalyst
XRD patterns of NHAp and Mg-NHAp are delineated in Fig. 1. Well-resolved characteristic peak of most elevated intensity for HAp was acquired at 2θ value of 31.77° corresponding to 211 planes. The phase formed was clear and matches well with standard pattern reported in literature.16 The standard corresponding plane for HAp (viz. 100, 101, 200, 002, 211, 202, 301, 130, 131, 113, 203, 222, 132, 321, 004, 240, 241, 502, 323, and 511) were well observed in case of the synthesized catalyst. The diffraction peak at 2θ = 62.97° with plane 502 corresponds to the Mg support as registered on the XRD patterns (Fig. 1).The principal evidence for framing of NHAp was in the form of a strong complex broad FTIR band (Fig. 2) focused at about 1000–1120 cm−1 because of asymmetric stretching mode of vibration for PO4 group.17 The band at 570 cm−1 corresponds to P–O stretching vibration of the PO4 group. As a major peak of phosphate group, the vibration peak could be distinguished in the region between 1120–960 cm−1 for both NHAp and Mg-NHAp, which were due to P–O asymmetric stretching of PO43−. Many of the compounds can be acknowledged to be analogs of ethers, especially when an alkoxy group was available, featuring the P–O–C linkage at 1240 cm−1. In case of Mg-NHAp the bands at 780–980 cm−1 and the peak in the range of 900–1200 cm−1 were resulted due to the peroxide formation (M–O–O–M) and the M–O–M bonding respectively.18
Fig. 3 shows a typical nitrogen adsorption–desorption isotherm of NHAp and Mg-NHAp. The isotherms display a type IV curve with a hysteresis loop which corresponds to mesoporous materials. NHAp and Mg-NHAp have BET surface areas of 10.865 m2 g−1 and 134.1 m2 g−1 respectively. Different types of surface area analysis are classified in Table 1. Fig. 3 (in side) shows a plot of varying pore volume distribution with reference to pore diameter (nm). The total pore volumes were found to be 0.0894 cc g−1 and 0.45 cc g−1 for NHAp and Mg-NHAp respectively. It can be seen from the Table 1 that, the BJH pore volumes were close to the BET pore volumes for both the samples. Further, it was confirmed that the grafting of Mg on NHAp has significantly increased the BET surface area as well as pore volume.
| Parameter | Surface area & pore volume | Co-relation co-efficient | ||
|---|---|---|---|---|
| NHAp | Mg-NHAp | NHAp | Mg-NHAp | |
| Langmuir surface area (m2 g−1) at PS/P0 = 0.9814 | 5.708 | 128.45 | 0.99 | 0.99 |
| One point BET surface area (m2 g−1) at PS/P0 = 0.3 | 8.879 | 131.24 | — | — |
| Adsorbed BET surface area (m2 g−1) | 10.865 | 134.1 | 0.97 | 0.99 |
| Adsorbed t-plot surface area (m2 g−1) | 27.261 | 136.376 | 0.98 | 0.99 |
| Total pore volume (cc g−1) at PS/P0 = 0.9814 | 0.0894 | 0.4529 | — | — |
| Total BJH pore volume (cc g−1) | 0.10882 | 0.46863 | — | — |
The morphologies of NHAp and Mg-NHAp are presented in Fig. 4(a)–(d) respectively. These SEM micrographs gave understanding into the structure with respect to pore sizes and distribution. The surface of NHAp is almost plane and less porous. However, grafting of Mg followed by calcination at 650 °C led to surface roughness and porous formation. As can be seen from the micrographs, surface morphologies of the Mg-NHAp appears as a polycrystalline material. Fig. 4(d) shows the porous structure is uniform however, the it was observed to be slit like. The calcination at 650 °C resulted in decomposition of carbonate which helped in forming uniform pores. Hence, the surface area as well as the pore volume was higher in case of Mg-NHAp than NHAp which was evident from the BET results. The EDX analysis of Mg-NHAp showed the presence of C (7.89%), O (43.83%), Mg (19.77%), P (14.13%), Ca (17.37%). The wt% analysis confirmed the oxide formation of Ca and Mg.
Fig. 5 demonstrates the TGA analysis of NHAp and Mg-NHAp. In case of NHAp, initial weight loss of 6.84% occurred in between the temperature regime of 90 °C to 150 °C because of the evaporation of water molecules. After that the weight loss was found to be about 35% which was due to the decomposition of carbonate as mentioned below.
| CaCO3 → CaO + CO2 | (16) |
However, in case of Mg-NHAp, the initial weight loss was only about 3.48% (within 150 °C). Moreover, no crest was found within a temperature region of 150 °C to 1000 °C and the reported weight loss was only 7.5%. The TGA curve within this specified temperature range was thought to be the aftereffect of continuous de-hydroxylation of Mg-NHAp powder. It is clear from the TGA that the total weight loss at 1000 °C was 10.98% for Mg-NHAp compared to 41.7% for NHAp. Thus, it was confirmed that Mg grafting resulted in a more stable Mg-NHAp.
3.2 Parametric effects on methyl ester yield
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, 1
5, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, 1
10, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15, 1
15, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, and 1
20, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 as shown in Fig. 6. It shows the effect of neem oil to methanol molar ratio on methyl ester yield with reference to reaction time. Significant effect was clearly observed affecting the methyl ester yield. The time required to reach highest yield at lower concentration of Mg-NHAp (2 weight% of oil) and lower oil to methanol ratio (1
25 as shown in Fig. 6. It shows the effect of neem oil to methanol molar ratio on methyl ester yield with reference to reaction time. Significant effect was clearly observed affecting the methyl ester yield. The time required to reach highest yield at lower concentration of Mg-NHAp (2 weight% of oil) and lower oil to methanol ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5), was approximately 250 minutes. But with increase in oil to methanol ratio the time required to reach highest conversion gradually decreased to 135 minutes.
5), was approximately 250 minutes. But with increase in oil to methanol ratio the time required to reach highest conversion gradually decreased to 135 minutes.
The concentration of methanol has dominant role during esterification reaction. The methyl ester yield was increased significantly with the addition of methanol. Nevertheless, higher concentration of methanol may adsorb on the catalyst active sites because of which the esterification process inhibited. As shown in the Fig. 6 the oil to methanol molar ratio was increased by keeping catalyst loading constant in the reaction system. It can be seen that after 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 oil to methanol ratio the reduction in conversion was clearly observed.
15 oil to methanol ratio the reduction in conversion was clearly observed.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15) and reaction temperature (70 °C) are presented in Fig. 7. It shows the percentage increase in methyl ester yield with and without catalyst. In the figure, the 0% Mg-NHAp shows that the whole reaction was conducted without catalyst. Further, the catalyst loading was gradually increased by 1% to see the catalytic effect on the yield obtained. The interaction between catalyst and reactant was more obvious at early stage of the reaction. It was evident from the analysis that without using catalyst the equilibrium conversion was achieved after three and half hour. Also it can be clearly noted that the time required to reach equilibrium conversion has gradually been decreased with increase in Mg-NHAp weight%. There is increase in reaction rate perhaps due to the increase in number of active sites inside the reaction chamber. Highest yield of 96.7% was observed when the Mg-NHAp loading increased to 6%. Methyl ester yield was increased from 48.7% (0% Mg-NHAp) to 95% (6% Mg-NHAp) in 135 minutes. The gradual increase in yield can be attributed to the increase in number of active catalytic sites available in the reaction chamber.
15) and reaction temperature (70 °C) are presented in Fig. 7. It shows the percentage increase in methyl ester yield with and without catalyst. In the figure, the 0% Mg-NHAp shows that the whole reaction was conducted without catalyst. Further, the catalyst loading was gradually increased by 1% to see the catalytic effect on the yield obtained. The interaction between catalyst and reactant was more obvious at early stage of the reaction. It was evident from the analysis that without using catalyst the equilibrium conversion was achieved after three and half hour. Also it can be clearly noted that the time required to reach equilibrium conversion has gradually been decreased with increase in Mg-NHAp weight%. There is increase in reaction rate perhaps due to the increase in number of active sites inside the reaction chamber. Highest yield of 96.7% was observed when the Mg-NHAp loading increased to 6%. Methyl ester yield was increased from 48.7% (0% Mg-NHAp) to 95% (6% Mg-NHAp) in 135 minutes. The gradual increase in yield can be attributed to the increase in number of active catalytic sites available in the reaction chamber.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 oil to methanol ratio. A significant and rapid rise in methyl ester yield was clearly observed with increase in reaction temperature. This is due to the higher molecular activity and mass transfer which eventually lead to faster reaction rate.
15 oil to methanol ratio. A significant and rapid rise in methyl ester yield was clearly observed with increase in reaction temperature. This is due to the higher molecular activity and mass transfer which eventually lead to faster reaction rate.
3.3 Evaluation of kinetic rate constant and activation energy of neem oil esterification reaction
Eqn (13) shows the reaction kinetic model for the pseudo-first-order reaction. This equation was used for regression analysis of the obtained experimental data at varying catalyst loading and reaction temperature so as to calculate the reaction rate constant. After linearly fitted, the slope i.e. k′f is listed in the Table 2. It's clearly showing the significant rise in rate with increase in reaction temperature.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 oil to methanol ratio
15 oil to methanol ratio
| Reaction temperature (K) | Oil to methanol ratio | k′f (min−1) | |
|---|---|---|---|
| Mg-NHAp = 0% | Mg-NHAp = 6% | ||
| 333 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
0.0135 | 0.0137 |
| 343 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
0.0265 | 0.0179 |
| 353 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
0.037 | 0.0244 |
| 363 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
0.0591 | 0.0364 |
The catalyst loading has also significant effect on the forward rate constant, which is explained by the following equation.
| k′f = kMg-NHApWMg-NHAp | (17) |
The kinetics parameter of esterification viz. rate constant, activation energy (Ea) and pre-exponential factor (A) were calculated from Arrhenius equation. Ea was calculated for two set of reactions separately, first is at 0% Mg-NHAp loading and second at 6% Mg-NHAp. Fig. 10 illustrates a linear correlation between ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k′f and 1/T. The slope of the line gives Ea. The final value of Ea and A is enlisted in Table 3. It's clearly showing that the activation energy decreases from 47.86 kJ mol−1 to 32.35 kJ mol−1 after addition of Mg-NHAp.
k′f and 1/T. The slope of the line gives Ea. The final value of Ea and A is enlisted in Table 3. It's clearly showing that the activation energy decreases from 47.86 kJ mol−1 to 32.35 kJ mol−1 after addition of Mg-NHAp.
| Ea (kJ mol−1) | A | |
|---|---|---|
a All the reaction were carried out with 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 oil to methanol ratio. 15 oil to methanol ratio. |
||
| Mg-NHAp = 0% | 47.86 | 464![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 167.1 167.1 |
| Mg-NHAp = 6% | 32.35 | 1575.9 |
3.4 Thermodynamic properties of Mg-NHAp catalyzed esterification
The thermodynamic properties of esterified product were also studied. As per Van't Hoff equation, the Gibbs free energy (ΔG in kJ mol−1) is expressed as follows:
ΔG = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K K
| (18) |
Considering change in enthalpy and entropy with temperature constant, ΔG can be written as
| ΔG = ΔH − TΔS | (19) |
Now combining both eqn (18) and (19),
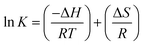 | (20) |
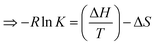 | (21) |
After plotting −R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K vs. 1/T and linearly fitting the values of ΔH, ΔS can be calculated.
K vs. 1/T and linearly fitting the values of ΔH, ΔS can be calculated.
A linear correlation between −R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K vs. 1/T is shown in Fig. 11(a) for both 0% Mg-NHAp and 6% Mg-NHAp loading. The slope and intercept of the corresponding plots represents enthalpy (ΔH) and entropy (ΔS) of the reaction. The enthalpy (ΔH) for 0% and 6% Mg-NHAp loading was found to be 104.06 kJ mol−1 and 49.3 kJ mol−1 at 333 K. Fig. 11(b) shows the Gibbs free energy variation for the esterification reaction with and without catalyst for temperature ranging from 333 K to 363 K. The trend line shows that the reaction is continuous process (Table 4).
K vs. 1/T is shown in Fig. 11(a) for both 0% Mg-NHAp and 6% Mg-NHAp loading. The slope and intercept of the corresponding plots represents enthalpy (ΔH) and entropy (ΔS) of the reaction. The enthalpy (ΔH) for 0% and 6% Mg-NHAp loading was found to be 104.06 kJ mol−1 and 49.3 kJ mol−1 at 333 K. Fig. 11(b) shows the Gibbs free energy variation for the esterification reaction with and without catalyst for temperature ranging from 333 K to 363 K. The trend line shows that the reaction is continuous process (Table 4).
 | ||
Fig. 11 (a) Linear correlation between −R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K vs. 1/T (b) Gibbs free energy variation for the esterification reaction for 0% and 6% Mg-NHAp loading. K vs. 1/T (b) Gibbs free energy variation for the esterification reaction for 0% and 6% Mg-NHAp loading. | ||
| Temperature (K) | ΔH (kJ mol−1) | |
|---|---|---|
| 0% Mg-NHAp loading | 6% Mg-NHAp loading | |
a All the reactions were carried out at oil to methanol molar ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15. 15. |
||
| 333 | 104.06 | 49.2 |
| 343 | 107.1 | 50.7 |
| 353 | 110.03 | 52.2 |
| 363 | 113.4 | 53.7 |
3.5 FTIR results of purified and esterified neem oil
Fig. 12 presents the graph in between wave number (cm−1) and transmittance (%) for esterified neem oil produced at constant reaction temperature (70 °C), oil to methanol ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30) and Mg-NHAp weight% (2%). At first it is clear from the figure that alkane is present without phosphorous and sulfur. This confirms that leaching of catalyst didn't occur and it was separated clearly. Also a C
30) and Mg-NHAp weight% (2%). At first it is clear from the figure that alkane is present without phosphorous and sulfur. This confirms that leaching of catalyst didn't occur and it was separated clearly. Also a C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group is identified at 1742 cm−1 which refers to ester. Other wavenumbers and corresponding functional groups are marked in the figure.
O group is identified at 1742 cm−1 which refers to ester. Other wavenumbers and corresponding functional groups are marked in the figure.
3.6 Comparison of earlier reported work with present work using hydroxyapatite based catalyst for esterification
Table 5 reported a comparison of different natural and synthetic hydroxyapatite based heterogeneous catalysts and their methyl ester yield at different operating parameters reported in literature.2,7,20–22 It can be observed from the data that all the catalyst developed possess very less BET surface area in comparison to present developed catalyst. But Al2O3/H3PO4 (synthetic hydroxyapatite) has very good surface area i.e. 120 m2 g−1. Jazie et al. tried to produce methyl ester from peanut oil using animal bone powder and observed more than 80% of yield with gradual increase in reaction time period. However, reaction time was bit higher than present study. They also have studied methyl ester yield of rapeseed oil using the same catalyst and found above 85% yield. In this case also reaction time period as well as methanol to oil ratio was extensively high.20 Chakraborty and RoyChowdhury tried to produce oleic acid ethyl ester using Cu grafted fishbone derived hydroxyapatite. The catalyst has very low surface area. In spite of having lower surface area the yield obtained was good. However, the effect of yield with and without metal grafting was not discussed.7 Xie et al. also produces methyl ester from rapeseed oil using biont shell without metal support. The results were much promising considering all the parameters except catalyst loading.21 Obadiah et al. used palm oil to produce methyl ester, for which the yield was above 80% at 15 wt% catalyst loading and 4 h reaction time period. They observed above 95% conversion when catalyst wt% loading was 20%, which is very high if the same process is considered in a large scale.2 Araujo et al. used synthetic hydroxyapatite with Al metal support for esterification of oleic acid. They observed highest conversion of 95% within 1 h, but only demerit in the work was reaction temperature i.e. 350 °C.22 Thus, observing all the positive and negative aspects of the proclaimed works, the present experiments were conducted. It is confirmed from the comparison that such types of reactions can give promising results using the present developed catalyst.| Fatty acid/vegetable oil | Catalyst | BET surface area (m2 g−1) | Reaction process parameter | Methyl ester yield |
|---|---|---|---|---|
| Peanut20 | Animal bone powder | 4.01 | 4 h, 60 °C, 600 rpm | <80% |
| Oleic acid7 | Cu/hydroxyapatite (fishbone) | 16.78 | 1 h, 70 °C, 800 rpm, 0.4 mL min−1 ethanol flow rate | <83.57% |
| Rapeseed21 | Biont shell | 12.64 | 3 h, 70 °C, 600 °C (calcination), 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 methanol to oil 1 methanol to oil |
<80% |
| Palm2 | Waste animal bone | 88.53 | 4 h, 65 °C, 800 °C (catalyst calcination), 200 rpm, 15 wt% catalyst loading | 80% |
| Rapeseed20 | Animal bone powder | 4.01 | 4 h, 60 °C, 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 methanol to oil, 600 rpm 1 methanol to oil, 600 rpm |
85% |
| Oleic acid22 | Al2O3/H3PO4 (synthetic) | 120 | 1 h, 350 °C, 0.15 (mmol acid per g product) | 95.5% |
| Present work (neem) | Mg/hydroxyapatite (fishbone) | 131.24 | 2.25 h, 70 °C, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 (oil to methanol), 6 wt% catalyst loading, 650 °C (calcination) 15 (oil to methanol), 6 wt% catalyst loading, 650 °C (calcination) |
95% |
3.7 Reusability study of Mg-NHAp during esterification
Reusability of Mg-NHAp was studied by multiple repetition (5 times) of esterification experiments at constant oil to methanol ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15), reaction temperature (70 °C) and catalyst loading (6 wt%). After each reaction, the final mixture was centrifuged to remove the catalyst. Then it was regenerated by washing with methanol several times and then dried for 2 h at 110 °C. The regeneration process was continued for 5 times after each run. From Fig. 13, it can be inferred that the yield after regeneration doesn't vary significantly up to 5th regeneration. After 5th regeneration the yield was decreased to 91.5% from 96.7% after 160 min. The low variation in yield after regeneration thus affirms the stability of Mg-NHAp.
15), reaction temperature (70 °C) and catalyst loading (6 wt%). After each reaction, the final mixture was centrifuged to remove the catalyst. Then it was regenerated by washing with methanol several times and then dried for 2 h at 110 °C. The regeneration process was continued for 5 times after each run. From Fig. 13, it can be inferred that the yield after regeneration doesn't vary significantly up to 5th regeneration. After 5th regeneration the yield was decreased to 91.5% from 96.7% after 160 min. The low variation in yield after regeneration thus affirms the stability of Mg-NHAp.
4. Conclusion
An active, reusable solid catalyst was prepared from cooked waste fish bone. Grafting of Mg ion by impregnation followed by calcination resulted in high surface area and pore volume. The method of reusing fishbone waste to prepare catalyst could help waste recycling, contaminants minimization, cost reduction and making the catalyst environmentally viable. This high efficient and low-cost fishbone catalyst could make the process of esterification economic also. Thus, it can be anticipated that this low-cost catalyst if utilized in a large-scale industrial process of biofuel production, will make the entire process sustainable as well as low cost. Also this work gives a new horizon towards the use of various bio-waste products for heterogeneous catalyst development.Acknowledgements
Authors acknowledge the analytical facilities i.e. FESEM, EDX, BET, TGA analysis provided by Central Instruments Facility (CIF), IIT Guwahati. Use of XRD facility (procured through FIST Grant No. SR/FST/ETII–028/2010 from Department of Science and Technology, Government of India) at Department of Chemical Engineering, IIT Guwahati is also acknowledged.References
- T. Rustad, Electron. J. Environ., Agric. Food Chem., 2003, 2, 458–463 Search PubMed.
- A. Obadiah, G. A. Swaroopa, S. V. Kumar, K. R. Jeganathan and A. Ramasubbu, Bioresour. Technol., 2012, 116, 512–516 CrossRef CAS PubMed.
- S. M. Best, A. E. Porter, E. S. Thian and J. Huang, J. Eur. Ceram. Soc., 2008, 28, 1319–1327 CrossRef CAS.
- T. Leventouri, Biomaterials, 2006, 27, 3339–3342 CrossRef CAS PubMed.
- J. Venkatesan, B. Lowe, P. Manivasagan, K. H. Kang, E. P. Chalisserry, S. Anil, D. G. Kim and S. K. Kim, Materials, 2015, 8, 5426–5439 CrossRef.
- K. Sasaki and T. Goto, Ceram. Int., 2014, 40, 11649–11656 CrossRef CAS.
- R. Chakraborty and D. RoyChowdhury, Chem. Eng. J., 2013, 215, 491–499 CrossRef.
- L. B. Sun, X. Q. Liu and H. C. Zhou, Chem. Soc. Rev., 2015, 44, 5092–5147 RSC.
- M. Verziu, B. Cojocaru, J. Hu, R. Richards, C. Ciuculescu, P. Filipc and V. I. Parvulescu, Green Chem., 2008, 10, 373–381 RSC.
- L. B. Sun, W. H. Tian and X. Q. Liu, J. Phys. Chem. C, 2009, 113, 19172–19178 CAS.
- C. Xu, D. I. Enache, R. Lloyd, D. W. Knight, J. K. Bartley and G. J. Hutchings, Catal. Lett., 2010, 138, 1–7 CrossRef CAS.
- J. Wang, T. Nonami and K. Yubata, J. Mater. Sci.: Mater. Med., 2008, 19, 2663–2667 CrossRef CAS PubMed.
- A. Baig and F. T. Ng, J. Am. Oil Chem. Soc., 2011, 88(2), 243–253 CrossRef CAS.
- D. T. Englis and J. E. Reinschreiber, Anal. Chem., 1949, 21.5, 602–605 CrossRef.
- J. I. Choi, W. H. Hong and H. N. Chang, Int. J. Chem. Kinet., 1996, 28, 37 CrossRef CAS.
- J. H. G. Rocha, A. F. Lemos, S. Kannan, S. Agathopoulosa and J. M. F. Ferreira, J. Mater. Chem., 2005, 15, 5007–5011 RSC.
- H. K. Varma and S. S. Babu, Ceram. Int., 2005, 31, 109–114 CrossRef CAS.
- S. Maensiri, P. Laokul and V. Promarak, J. Cryst. Growth, 2006, 289, 102–106 CrossRef CAS.
- M. G. Kulkarni, R. Gopinath, L. C. Meher and A. K. Dalai, Green Chem., 2006, 8, 1056–1062 RSC.
- A. A. Jazie, H. Pramanik and A. S. K. Sinha, Materials for Renewable and Sustainable Energy, 2013, 2, 1–10 CrossRef.
- J. Xie, X. Zheng, A. Dong, Z. Xiao and J. Zhang, Green Chem., 2009, 11, 355–364 RSC.
- L. R. R. D. Araujo, C. F. Scofield, N. M. R. Pastura and W. D. A. Gonzalez, Mater. Res., 2006, 9, 181–184 CrossRef.
| This journal is © The Royal Society of Chemistry 2016 |












