Mixing control by frequency variable magnetic micropillar†
Hao Yua,
Thien-Binh Nguyenab,
Sum Huan Ngb and
Tuan Tran *a
*a
aSchool of Mechanical & Aerospace Engineering, Nanyang Technological University, 50 Nanyang Avenue, 639798, Singapore. E-mail: ttran@ntu.edu.sg
bSingapore Institute of Manufacturing Technology, 71 Nanyang Drive, 638075, Singapore
First published on 21st January 2016
Abstract
Mixing of fluids in microchannels is a crucial yet challenging task in lab-on-a-chip devices. Here we demonstrate a mixing enhancement method of two streams of fluids based on actuation of a single micropillar embedded with paramagnetic nanoparticles. The actuation is controlled by an external rotating magnetic field and has variable frequency up to ≈160 Hz. We systematically study the effects of actuation frequency and flow rate on the mixing efficiency, which can almost reach the highest value corresponding to the completely mixed state. We find that the resulting concentration profile of the mixed fluids downstream of the actuated pillar can be described by the convection–diffusion equation with an effective coefficient that depends only on one control parameter: the number of actuation cycles of the micropillar during the fluid's transit time. Our results indicate that the proposed approach may be adapted and scaled up to enhance the mixing efficiency with high degree of control in other lab-on-chip devices.
1 Introduction
The emergence of microfluidic technology has allowed numerous analytic tasks to be performed on minute quantities of fluids.1–4 One of the most basic tasks that needs to be carried out rapidly and efficiently in microfluidic devices is mixing of different fluids.5 Yet this is also one of the most challenging tasks because the dominant mechanism for mixing at such inherently small scales is diffusion, which is significantly slow compared to mixing by convection. Various microfluidic modules designated for mixing have thus been proposed. These so-called micromixers can be mainly divided into two categories: passive and active ones.6,7 Passive micromixers rely on elaborate channel geometries to enhance convective fluid motion and hence mixing efficiency.8–10 Though passive micromixers normally require long mixing length or elaborate in-channel microstructures, they are useful in a variety of applications because their working principles are simple and no external stimulus is required.In contrast, active micromixers generate flow disturbances by utilizing auxiliary stimuli such as acoustic, temperature, electric, and magnetic fields.11 As the names suggest, they allow active control of the mixing process, generally by applying external fields on moving elements that are specially designed and fabricated in microfluidic channels. Given the small size, active micromixers often involve complicated fabrication process and requirement for integration of various on-chip components, e.g., current carrying wires12 or piezoelectric actuators.13,14 The external stimuli may also cause undesirable effects such as unnecessary and even harmful heating of samples. Nonetheless, the capability of controlling the mixing performance in active micromixers is an attractive feature. Thus, active micromixers with minimum obstacles in the fabrication process and maximized usability are increasingly explored in the microfluidics community.
Recently, magnetism has been used to manipulate magnetic objects in microfluidic devices. This enables realization of various functions such as transportation, positioning, separation, sorting, propulsion, and mixing.15 Using magnetic particles moved by external magnetic stimuli is one of the approaches to mixing static or moving fluids in integrated lab-on-chip diagnostic assays.16 However, it should be noted that fluid purification and separation of particles are required in this method due to inevitable contact between the magnetic objects and the handling fluids. External magnetic stimuli can also be used to actuate integrated components, which are usually made of polymer composite doped with magnetic particles. One of the major benefits of this approach is its high compatibility with biological fluids.17 Most notably, micro- and nano-scaled artificial cilia, which are inspired by biological cilia or flagella, have been increasingly utilized as active on-chip components.18,19 During the past decade, several groups have developed bio-inspired artificial cilia for propulsion, pumping, and mixing in microfluidic devices. Most of the proposed artificial cilia are lying film cantilevers, which can be actuated by a rotating or swinging magnetic field.20–25 As for upright artificial cilia, Khatavkar et al.26 numerically simulated fluid mixing using two artificial cilia positioned perpendicularly to the substrate and studied the system's optimal configuration for mixing. Shields et al.27 reported simultaneous pumping and mixing processes driven by biomimetic nano-rod arrays, which were actuated by a steel-tipped magnet. Chen et al. incorporated arrays of micropillars in microchannels and studied the mixing enhancement numerically and experimentally.28,29 The micropillars were made of polydimethylsiloxane (PDMS) embedded with microparticles and were actuated in several different beating patterns using an elaborate electromagnet system with actuation frequency up to 30 Hz. In a recent study, Zhou et al.30 proposed a disordered arrangement of magnetic micropillars in a microchannel that caused significant distortion of the flow field leading to enhancement of the mixing performance. The focus therefore was on the arrangement of micropillars; a two-phase (on/off or bending/straight) beating pattern of micropillars was implemented by a linear movement of a permanent magnet. Thus, it is observed that mixing enhancement in the aforementioned active micromixers was achieved either by increasing the quantity of artificial cilia or by imposing elaborate beating schemes to the cilia.
In this paper, we demonstrate an active micromixer consisting of a single magnetic micropillar that can be actuated with frequency as high as 161.6 Hz. We present a systematic study and analysis of the mixing performance on the actuation frequency and the flow rate. We also show that the resulting mixing performance can be described by the convection–diffusion equation with an effective diffusion coefficient.
2 Experimental
2.1 Chip fabrication
We fabricate PDMS micromixers using a silicon mold made by standard UV photolithography and deep reactive-ion etching. The silicon mold has an inverted channel with width and height of 200 μm, as well as a hole with diameter of 40 μm and depth of 150 μm. Prior to PDMS chip fabrication, the silicon mold was coated by trichloro(1H,1H,2H,2H-perfluorooctyl)silane (Sigma-Aldrich) by vapor deposition for four hours in a vacuum desiccator. This step was to ensure easy detachment of PDMS chips from the mold.In order to fabricate the magnetic micropillar for each micromixer, we use dry iron oxide nanoparticles (NPs) with a proprietary hydrophobic surfactant (EMG 1400, Ferrotec). The particle mixture consists of 50% Fe3O4 and 50% γ-Fe2O3 with an average particle size of 10 nm. Magnetic NPs were then poured over the silicon mold with a permanent neodymium magnet (magnetic field induction 0.4 T) underneath to attract NPs into the microhole. The filling process was repeated several times until a cluster of NPs in the microhole was observed under a microscope. Excessive NPs were then removed carefully from the mold surface using Kimwipes wipers. It is crucial in this step to make sure NPs are not taken away from the microhole due to electrostatic force from the wipers. A mixture of PDMS base and curing agent at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixing ratio was poured onto the silicon mold in a Petri dish, which subsequently was placed in the vacuum desiccator for degassing. During gas evacuation, a permanent magnet was attached to the backside of the Petri dish in order to facilitate formation of the NP cluster allowing PDMS to flow into the microhole. The PDMS chip containing the micromixer was cured in an oven at 80 °C for 3 hours. After the chip was peeled off from the mold, access holes were made with a 1 mm diameter puncher. The chip was then bonded to a PDMS-coated glass slide by air-plasma surface activation and post-baking at 80 °C for 5 minutes.
1 mixing ratio was poured onto the silicon mold in a Petri dish, which subsequently was placed in the vacuum desiccator for degassing. During gas evacuation, a permanent magnet was attached to the backside of the Petri dish in order to facilitate formation of the NP cluster allowing PDMS to flow into the microhole. The PDMS chip containing the micromixer was cured in an oven at 80 °C for 3 hours. After the chip was peeled off from the mold, access holes were made with a 1 mm diameter puncher. The chip was then bonded to a PDMS-coated glass slide by air-plasma surface activation and post-baking at 80 °C for 5 minutes.
2.2 Actuation scheme
In Fig. 1(a), we show the experimental setup used to evaluate the mixing performance of our micromixer. The magnetic field was generated in the gap between two countersunk permanent neodymium magnets (ϕ 20 mm, Titan Magnetics). The gap was kept at 12 mm, and the magnetic field induction, measured at the center of the gap by a gauss meter (GM07, Hirst Magnetics), was uniform within the instrumental accuracy: B = 0.28 T (see Fig. 1(b)). We then fixed the pair of magnets on a U-shaped mounting bracket, which can be rotated using a motor with the frequency fB. The resulting magnetic field rotates around the x-axis of the microchannel with frequency identical to that of the motor. | ||
| Fig. 1 (a) Three dimensional schematic of the experimental setup used to evaluate the micromixer's performance. Inset: SEM micrograph of a Y-type microchannel (200 μm in width and 200 μm in depth) incorporated with a micropillar (40 μm in diameter and 150 μm in height). (b) Magnetic induction B measured in the gap between two magnets (see Fig. S1 of ESI for more information†). The red dashed line indicates the region where the micromixer was placed; the magnetic induction in this region is B = 0.28 T. (c) Upper panel: sequential bright-filed images showing micropillar's one beating cycle. Lower panel: micropillar's beating amplitude δ measured for actuation frequency f = 60.4 Hz, vs. time t and magnetic field rotational angle θ. Inset: definition of δ and θ. (d) Schematics of microchannel and boundary conditions used in the nonlinear diffusive mixing model. | ||
We show a series of images in Fig. 1(c) illustrating the movement of a micropillar in a microchannel. It can be seen that a cluster of magnetic nanoparticles (NPs) is permanently locked in PDMS along the longitudinal direction of the micropillar. Due to the micropillar's geometrical anisotropy and the elongated NP cluster, the micropillar is magnetically anisotropic.12,31 As a result, magnetic anisotropy enables a uniform magnetic field to generate a magnetic torque.31 Subsequently, this magnetic torque causes the micropillar to bend around the anchoring point.
A beating cycle of the micropillar, as shown in Fig. 1(c), starts from its upright position, followed by back-and-forth movements between the two sidewalls and finally completes at its upright position. To quantitatively characterize the actuation, we measured the bending amplitude δ, defined as the deflection of the moving tip of the micropillar (see the inset in Fig. 1(c), lower panel), by high-speed cameras from bottom and side views. In Fig. 1(c), we show the dependence of δ on time t and the rotational angle θ of the magnetic field for actuation frequency f = 60.4 Hz. Notably, the obtained non-reciprocal mode is similar to the asymmetric beating behavior of a propulsive biological cilium, which normally consists of a fast effective stroke and a slow recovery stroke.32,33 There are two types of beats observed here in one beating cycle. In beat I, the amplitude increases as the magnetic torque bends the micropillar following the rotating magnetic field lines until the micropillar touches a sidewall. In beat II, the micropillar rapidly swing backwards to the other sidewall due to the combined effects of magnetic torque and elastic force. In active fluid mixing, both of these transverse beats are considered effective in fluid transportation across the flow.26 It should be noted that the duration of one beating cycle is one half of the revolution of the rotating magnetic field; therefore the actuation frequency f of the micropillar is f = 2fB. Thus we were able to accurately control the actuation frequency and varied it from 0 to 161.6 Hz, which is close to the intrinsic beating frequency of a biological cilium (10–100 Hz).19 Representative videos showing the bottom and side views of the micropillar actuation can be found in the ESI.†
3 Results and discussion
3.1 Diffusive mixing measurement and model
In order to quantify the mixing performance of the active micromixer, we first performed an experiment in which the micropillar was inactivated and mixing was done entirely by molecular diffusion. This serves as a validation for our experimental method and a reference to evaluate the performance of our active micromixer. In Fig. 1(d), we show a schematic of the micromixer: inlet A, hereafter referred to as the solvent side, was supplied with an aqueous glycerol solution (G7757, Sigma-Aldrich) having 50 wt% in DI water, whereas inlet B, referred to as the solute side, was supplied with the same glycerol solution mixed with a fluorescent dye (fluorescein sodium salt, F6377, Sigma-Aldrich) with concentration 100 μM. The flow rates going through the two inlets were kept identical by a duo-syringe pump (Legato 101, KD Scientific).We varied the flow rate Q through the main microchannel in the range 0.88–3.00 μL min−1. The averaged velocity ū = Q/W2 was from 0.37 mm s−1 to 1.25 mm s−1, where W = 200 μm was the height and width of the main microchannel. As a result, the Reynolds number Re = ūWρ/μ was from 1.4 × 10−2 to 4.7 × 10−2, where ρ = 1126.3 kg m−3 and μ = 6.0 mPa s were the density and dynamic viscosity of the DI–glycerol solution respectively. We chose to work with such small Reynolds numbers (Re ≪ 1) in order to demonstrate the efficiency of our micromixer.
We illuminated the micromixer by a mercury lamp through an excitation filter (450–490 nm). The emitted light from the measured area of the microchannel was captured by a high-speed camera (Fastcam SAX2, Photron) attached to a long working-distance microscope and a 10× objective (Optem). A long-pass filter (520 nm) was placed in front of the objective to make sure only emitted light from the fluorescent dye was recorded. The intensity profile of the emitted light in the transverse direction is related to the concentration profile of the solute and is indicative of the mixing performance: a flat intensity profile signifies that the solute is completely mixed across the microchannel. In Fig. 2(a), we show several typical fluorescent images of the flow at different stream-wise locations x = 1, 2, 3, 4 mm while the flow rate was kept at 1.34 μL min−1. At any stream-wise location x0, a grayscale profile I(y) was obtained by taking an averaged profile in a region of interest (ROI) bounded within [x0 − 0.1, x0 + 0.1] mm. For each I, a corresponding normalized profile I* was obtained using the minimum grayscale value Imin and the maximum value Imax, both measured at the inlet, as follows:
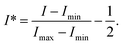 | (1) |
In Fig. 2(b), we show several dimensionless concentration profiles c* versus y* = y/W, the normalized transverse distance from the center line, at different normalized stream-wise locations x* = x/W = 5, 10, 15, 20. It can be seen that as the fluid moves downstream, the difference in solute concentration between the solute side and the solvent side is slightly decreased due to molecular diffusion. In Fig. 2(c), we show the effect of flow rate on the concentration profile at the microchannel's outlet x* = 20. Within the range of the flow rate in our experiment, the mixing effect can be qualitatively considered as insignificant.
The transport of solute across the microchannel can be described by a two-dimensional non-linear diffusive model in which changes in solute concentration are caused by convective and diffusive effects. The governing equation, written in dimensionless form, is as follows:34
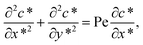 | (2) |
| D = D0[(1 − a)(c* + 0.5) + a]. | (3) |
Together with the governing equation, the dimensionless boundary conditions are (see the lower panel of Fig. 1(d) for a detailed illustration):
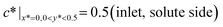 | (4) |
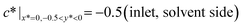 | (5) |
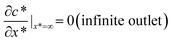 | (6) |
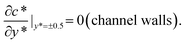 | (7) |
3.2 Mixing evaluation
We now present the performance analysis of our micromixer with the micropillar activated. For each of the flow rate Q and frequency f, the normalized concentration profile c*(y*) at the outlet (x* = 20) was recorded. Generally we observed that the concentration profile were increasingly flattened with increasing actuation frequency (as exemplified in Fig. 3(a) and (b)) and decreasing flow rate, as shown in Fig. 3(c). This indicates that the transverse component of the flow caused by the micropillar's beating was effective in homogenizing the unmixed volumes. We evaluated the mixing effectiveness using the efficiency η, which is defined based on the standard deviation of the concentration distribution 10,38: η = 1 − 〈(c*/〈c*〉 − 1)2〉1/2, where 〈〉 indicates average across the profile. The value of η is 0 for completely separated streams and 1 for completely mixed streams. The dependence of η on f and Q is shown in Fig. 3(d). For relatively lower flow rates, η can reach high values with sufficiently high actuation frequency: η ≳ 0.90 at the outlet was achieved at frequencies f = 138.9 and 161.6 Hz for flow rates Q = 0.88, 1.00, and 1.34 μL min−1, respectively. Note that the mixing length, defined as the distance required for complete diffusive mixing to occur, is estimated as lm = ūW2/D0.10 For these low flow rates, the respective mixing lengths are lm ≈ 122, 139, 186 mm, which are excessively long as opposed to 4 mm, the distance from inlet to outlet in our experiment. For higher flow rates Q = 2.00, and 3.00 μL min−1, the efficiency only reaches 0.67 and 0.45 respectively at the maximum frequency 161.6 Hz. However, we note that generally η first increases linearly with f and subsequently saturates only when it reaches a sufficiently high value for the cases of lower flow rates. Thus, because there is no sign of saturation at higher flow rates, we predict that higher values of η can be achieved with higher actuation frequency.Although η quantitatively measures the mixing performance across the microchannel it is still unclear how the two contributing factors f and Q are related and collectively affect the mixing performance. A qualitative observation can be made from Fig. 3(d) that the increasing rate of η is higher for lower Q. To gain further insight on the effect of f and Q on the mixing performance, we compared the concentration profiles caused by the actuated micropillar with those caused only by diffusive mixing. We used the diffusive model to find the best fit for each of the concentration profile in the case that the micropillar was actuated. The diffusion coefficient D used to calculate the Peclet number in eqn (2) is now
| D = De0[(1 − a)(c* + 0.5) + a]. | (8) |
We take a = 0.8, which is the constant that characterizes molecular interactions for the same pair of solvent and solute as in the case of inactivated micropillar. The fitting parameter De0 represents an effective diffusion coefficient that would cause the same concentration profile at the outlet. The resulting profiles are in remarkable agreement with the experimental data for a wide range of frequency and flow rate, as exemplified in Fig. 3(b) and (c). The concentration profile at the outlet caused by the actuated micropillar is therefore well described by the modified diffusive mixing model. We also infer that the extracted effective diffusion coefficient De0 can be used to characterize the mixing process caused by the actuated micropillar.
In Fig. 4, we show the ratio De*0 = De0/D0 versus the normalized frequency f* = LW2f/Q. Here De*0 characterizes the mixing enhancement, whereas f* is the number of beating cycles of the micropillar during the transit time, defined as the duration for the liquid to travel from the inlet to the outlet: τ = LW2/Q. The collapse of data in Fig. 4 signifies that the mixing enhancement caused by a stimulus such as the actuated micropillar depends only on a control parameter f*. We also note that De*0 first increases linearly with f* and starts saturating at De*0 = 10.13 and f* = 1330. The saturation point corresponds to η = 0.95 ± 0.01 and is the optimum point for mixing enhancement: further increase of f only marginally increases the mixing enhancement. The data collapse also leads us to a prediction that in order to achieve the same mixing enhancement for higher flow rates Q, the frequency f has to be increased linearly.
4 Conclusions
We have demonstrated an effective mechanism for mixing of two streams of liquids using an actuatable micropillar in a microchannel. The micropillar embedded with paramagnetic nanoparticles can be actuated by a rotating magnetic field at tunable frequencies. We conducted a systematic study to evaluate the mixing efficiency for a wide range of actuation frequencies as well as flow rates. We showed that with sufficiently high frequency, it was possible to achieve mixing efficiency up to 0.95 for the tested range of Reynolds number. We found that the concentration distributions can be described by a nonlinear diffusive mixing model for both cases: inactivated micropillar, in which mixing was dominated by diffusion, and activated micropillar, in which the mixing process was enhanced by convective fluid motion caused by the micropillar's beating. In the former case, the nonlinear diffusive mixing model resulted in the molecular diffusion coefficient D0, while in the later case, the model enabled extraction of an effective diffusion coefficient De0, which can be used as an alternative to characterize the mixing enhancement. We showed that the normalized coefficient De*0 = De0/D0 was dependent on only one control parameter, the dimensionless frequency, which represents the number of beating cycles of the micropillar during the transit time of fluid from inlet to outlet. This enables accurate predictions of the required actuation frequency for mixing with varying flow rates and serves as a building block for adapting the micromixer to other integrated lab-on-chip devices. Furthermore, the demonstrated actuation scheme of the micropillar may be useful for other microscopic manipulation purposes such as motion generation and particles transportation.Acknowledgements
This work was supported by the Nanyang Technological University and A*STAR, Singapore. H. Yu wishes to acknowledge the research fellowship and T.-B. Nguyen wishes to acknowledge the PhD scholarship from Nanyang Technological University.References
- D. J. Beebe, G. A. Mensing and G. M. Walker, Annu. Rev. Biomed. Eng., 2002, 4, 261–286 CrossRef CAS PubMed.
- H. A. Stone, A. D. Stroock and A. Ajdari, Annu. Rev. Fluid Mech., 2004, 36, 381–411 CrossRef.
- T. M. Squires and S. R. Quake, Rev. Mod. Phys., 2005, 77, 977–1026 CrossRef CAS.
- G. M. Whitesides, Nature, 2006, 442, 368–373 CrossRef CAS PubMed.
- J. M. Ottino and S. Wiggins, Philos. Trans. R. Soc., A, 2004, 362, 923–935 CrossRef PubMed.
- N. T. Nguyen and Z. Wu, J. Micromech. Microeng., 2005, 15, R1–R16 CrossRef.
- V. Hessel, H. Löwe and F. Schönfeld, Chem. Eng. Sci., 2005, 60, 2479–2501 CrossRef CAS.
- F. G. Bessoth, A. J. DeMello and A. Manz, Anal. Commun., 1999, 36, 213–215 RSC.
- R. H. Liu, M. A. Stremler, K. V. Sharp, M. G. Olsen, J. G. Santiago, R. J. Adrian, H. Aref and D. J. Beebe, J. Microelectromech. Syst., 2000, 9, 190–197 CrossRef.
- A. D. Stroock, S. K. Dertinger, A. Ajdari, I. Mezić, H. A. Stone and G. M. Whitesides, Science, 2002, 295, 647–651 CrossRef CAS PubMed.
- C. Y. Lee, C. L. Chang, Y. N. Wang and L. M. Fu, Int. J. Mol. Sci., 2011, 12, 3263–3287 CrossRef CAS PubMed.
- F. Fahrni, M. W. J. Prins and L. J. van Ijzendoorn, J. Magn. Magn. Mater., 2009, 321, 1843–1850 CrossRef CAS.
- Z. Yang, H. Goto, M. Matsumoto and R. Maeda, Electrophoresis, 2000, 21, 116–119 CrossRef CAS PubMed.
- K. Oh, B. Smith, S. Devasia, J. J. Riley and J. H. Chung, Microfluid. Nanofluid., 2010, 9, 645–655 CrossRef CAS.
- N. Pamme, Lab Chip, 2006, 6, 24–38 RSC.
- A. Van Reenen, A. M. De Jong, J. M. J. Den Toonder and M. W. J. Prins, Lab Chip, 2014, 14, 1966–1986 RSC.
- J. M. J. D. Toonder and P. R. Onck, Trends Biotechnol., 2013, 31, 85–91 CrossRef PubMed.
- R. Dreyfus, J. Baudry, M. L. Roper, M. Fermigier, H. A. Stone and J. Bibette, Nature, 2005, 437, 862–865 CrossRef CAS PubMed.
- B. A. Evans, A. R. Shields, R. L. Carroll, S. Washburn, M. R. Falvo and R. Superfine, Nano Lett., 2007, 7, 1428–1434 CrossRef CAS PubMed.
- E. M. Gauger, M. T. Downton and H. Stark, Eur. Phys. J. E: Soft Matter Biol. Phys., 2009, 28, 231–242 CrossRef CAS PubMed.
- S. N. Khaderi, M. G. H. M. Baltussen, P. D. Anderson, D. Ioan, J. M. J. Den Toonder and P. R. Onck, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys., 2009, 79, 046304 CrossRef CAS PubMed.
- F. Fahrni, M. W. J. Prins and L. J. Van IJzendoorn, Lab Chip, 2009, 9, 3413–3421 RSC.
- J. Belardi, N. Schorr, O. Prucker and J. Rühe, Adv. Funct. Mater., 2011, 21, 3314–3320 CrossRef CAS.
- S. N. Khaderi, C. B. Craus, J. Hussong, N. Schorr, J. Belardi, J. Westerweel, O. Prucker, J. Rühe, J. M. J. Den Toonder and P. R. Onck, Lab Chip, 2011, 11, 2002–2010 RSC.
- J. Hussong, N. Schorr, J. Belardi, O. Prucker, J. Rühe and J. Westerweel, Lab Chip, 2011, 11, 2017–2022 RSC.
- V. V. Khatavkar, P. D. Anderson, J. M. J. den Toonder and H. E. H. Meijer, Phys. Fluids, 2007, 19, 083605 Search PubMed.
- A. R. Shields, B. L. Fiser, B. A. Evans, M. R. Falvo, S. Washburn and R. Superfine, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 15670–15675 CrossRef CAS PubMed.
- C. Y. Chen, C. Y. Chen, C. Y. Lin and Y. T. Hu, Lab Chip, 2013, 13, 2834–2839 RSC.
- C. Y. Chen, C. Y. Lin, Y. T. Hu, L. Y. Cheng and C. C. Hsu, Chem. Eng. J., 2015, 259, 391–396 CrossRef CAS.
- B. Zhou, W. Xu, A. A. Syed, Y. Chau, L. Chen, B. Chew, O. Yassine, X. Wu, Y. Gao, J. Zhang, X. Xiao, J. Kosel, X. X. Zhang, Z. Yao and W. Wen, Lab Chip, 2015, 15, 2125–2132 RSC.
- J. Kim, S. E. Chung, S. E. Choi, H. Lee, J. Kim and S. Kwon, Nat. Mater., 2011, 10, 747–752 CrossRef CAS PubMed.
- M. Murase, The Dynamics of Cellular Motility, John Wiley & Sons, 1992 Search PubMed.
- S. Nonaka, H. Shiratori, Y. Saijoh and H. Hamada, Nature, 2002, 418, 96–99 CrossRef CAS PubMed.
- Z. Wu, N.-T. Nguyen and X. Huang, J. Micromech. Microeng., 2004, 14, 604 CrossRef CAS.
- F. A. Bassi, G. Arcovito and G. D. Abramo, J. Phys. E: Sci. Instrum., 1977, 10, 249 CrossRef CAS.
- M. B. Mustafa, D. L. Tipton, M. D. Barkley, P. S. Russo and F. D. Blum, Macromolecules, 1993, 26, 370–378 CrossRef CAS.
- R. Furukawa, J. L. Arauz-Lara and B. R. Ware, Macromolecules, 1991, 24, 599–605 CrossRef CAS.
- A. Venancio-Marques, F. Barbaud and D. Baigl, J. Am. Chem. Soc., 2013, 135, 3218–3223 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra24996a |
| This journal is © The Royal Society of Chemistry 2016 |



