Rhodamine B-sensitized BiOCl hierarchical nanostructure for methyl orange photodegradation†
Huiping Zhao‡
,
Yafang Zhang‡,
Guangfang Li,
Fan Tian ,
Han Tang and
Rong Chen*
,
Han Tang and
Rong Chen*
School of Chemistry and Environmental Engineering, Wuhan Institute of Technology, Xiongchu Avenue, Wuhan, 430073, PR China. E-mail: rchenhku@hotmail.com; Fax: +86 2787195060; Tel: +86 13659815698
First published on 8th January 2016
Abstract
Loose-packed flower-like BiOCl hierarchical nanostructures (HNs) assembled by thin nanosheets were successfully synthesized via a facile solvothermal method. Based on the matchability analysis of band structure of BiOCl and the molecular orbitals of the dyes, the RhB dye-sensitized heterogeneous BiOCl HNs system was established for MO degradation under visible light irradiation. The mechanism of MO degradation over RhB-sensitized BiOCl was also discussed according to the photocurrent and photoluminescence results. It was found that MO degradation efficiency over RhB-sensitized BiOCl HNs was obviously improved due to the generation of superoxide radicals (˙O2−) through the reduction of surface-adsorbed O2 molecules by the injected electrons on the conduction band of BiOCl HNs from the excited RhB molecules. This work may provide insight on better understanding the organic dye photodegradation process in RhB dye-sensitized BiOCl heterogeneous systems and explore a dye-sensitized semiconductor photocatalyst strategy for environmental remediation.
1. Introduction
Environmental remediation in terms of semiconductor photocatalysis has been reported as a promising strategy for organic wastewater treatment.1–3 However, wide bandgap semiconductors which can only absorb limited UV light were commonly used in organic dye degradation, thus hindering their applications.2,4–8 By introducing suitable visible light responsive organic dyes, the excited electrons generated by dye sensitization flow into conduction band (CB) of the semiconductor while holes localize on organic dyes, which realizes efficient spatial separation of photo-generated electrons and holes, thus resulting in the improvement of photocatalytic efficiency.9–11 Previous works also reported that electrons localized on CB could be further trapped by surface adsorbed oxygen molecules in some semiconductors, which resulted in the production of superoxide radicals.10,12–15 As superoxide radicals are widely considered as destructive reactive oxygen species (ROS) for organic compounds deterioration, the utilization of sensitization process via employing suitable dye sensitizers as sacrifice regent could improve the overall remediation performance of wide-bandgap semiconductor photocatalysts in organic wastewater treatment, which could be a useful strategy for real mixed dye treatment. For example, it has been reported that MO could be easily photo-dissociated in the presence of RhB and MB over CoFe2O4/BiOX (X = Cl, Br, and I),16 Ag/ZnO,17 and graphene, charcoal, ZnO, and ZnS/BiOX (X = Cl, Br, and I) hybrid microspheres.18As a wide-bandgap semiconductor (Eg = 3.1–3.5 eV), BiOCl can only be excited by ultraviolet light.19–22 However, it exhibits excellent visible-light-induced activity through photosensitization pathway owing to the relatively low conduction band, which involves superoxide radical intermediates.10,13 For example, BiOCl nanosheets with tunable thickness exhibit superior visible-light-induced RhB photodegradation efficiency via a photosensitization pathway.23 In this photocatalysis process, superoxide radicals (·O2−) were generated through the reduction of surface adsorbed O2 molecules by the injected electrons on CB of BiOCl from the excited RhB molecules.10,24 It was believed that the yield of ·O2− active species could not only achieve RhB photodegradation under visible light irradiation, but also endow RhB-sensitized BiOCl heterogeneous system with high activity for other organic pollutants degradation. Unfortunately, the application of RhB-sensitized BiOCl heterogeneous system for other organic dye removal and the corresponding degradation mechanism has been seldom reported. Given the fact that hierarchical BiOCl (HNs) semiconductor generally possesses larger surface area and outstanding dye adsorption capacity,14,25 it is reasonable to improve the performance for organic dyes removal via the construction of RhB-sensitized BiOCl HNs.
To verify our hypothesis, in this work, we fabricated flower-like BiOCl hierarchical nanostructures (HNs) with loose-packed structure and self-assembled thin nanosheets by solvothermal method. The visible-light-driven degradation performance for methyl orange (MO) was determined in pure BiOCl HNs and RhB-sensitized BiOCl HNs system. Based on the active species trapping experiment combined with the electrochemical and photoluminescence analysis, the possible mechanism for enhanced visible-light-induced MO degradation efficiency over the RhB dye-sensitized BiOCl HNs was also discussed. These results would provide new insights for better understanding the role of dye-sensitized semiconductor heterogeneous system for the organic pollutants treatment in wastewater.
2. Experimental
2.1 Chemicals
Bismuth nitrate pentahydrate (Bi(NO3)3·5H2O), rhodamine B (RhB) and methyl orange (MO) were purchased from Aladdin (Shanghai, China). Sodium chloride (NaCl), cobalt dichloride (CoCl2) and diethylene glycol (DEG) were purchased from Sinopharm Chemical Reagent Co., Ltd. (Shanghai, China). Citric acid was purchased from Tianjin Kermel Chemical Reagent Co., Ltd. All the reagents were analytical grade and used directly without further purification.2.2 Preparation
In a typical procedure, 0.485 g Bi(NO3)3·5H2O and 0.2 g citric acid were dissolved into 20 mL diethylene glycol (DEG) with vigorous stirring to obtain a transparent solution. Then it was fully mixed with 10 mL deionized water solution containing 0.058 g NaCl, and transferred to a Teflon-lined stainless steel autoclave to perform a solvothermal process at 150 °C for 15 h. After cooling down to room temperature naturally, the solid product was collected by centrifugation and then washed with deionized water and ethanol for five times. The sample (S1) was finally dried in a desiccator for a few days at room temperature for further characterization. To understand the formation of BiOCl hierarchical nanostructures, controlled experiments were also carried out under identical conditions by varying the Cl precursor and the amount of citric acid. The detailed procedure is the same as described above and all the experimental parameters are listed in Table S1 (ESI†).2.3 Characterization
X-ray powder diffraction (XRD) was carried out on Bruker axs D8 Discover (Cu K = 1.5406 Å) at a scan rate of 2° min−1 in the 2θ range from 10° to 80°. Scanning electron microscopy (SEM) images were taken on a Hitachi S4800 scanning electron microscope operating at 5.0 kV. Transmission electron microscopy (TEM) was recorded on a Philips Tecnai G2 20 electron microscope, using an accelerating voltage of 200 kV. Samples for TEM analysis were prepared by drying a drop of nanocrystal dispersion in absolute ethanol on carbon-coated copper grids. Brunauer–Emmett–Teller (BET) specific surface area was analyzed by nitrogen adsorption in a Micromeritics ASAP 2020 nitrogen adsorption apparatus (USA). The as-prepared sample was degassed at 150 °C for 4 h prior to nitrogen adsorption measurements. UV-Vis diffuse reflectance spectrum (DRS) was recorded on a UV-Vis spectrometer (Shimadzu UV-2550) by using BaSO4 as a reference and was converted from reflection to absorbance by the Kubelka–Munk method. The fluorescence spectrum was determined by a Hitachi 850-luminescence spectrophotometer with a Xe lamp at room temperature.2.4 Photodegradation activity evaluation
The photosensitization efficiency of different BiOCl samples was firstly evaluated by single-component dye degradation of RhB under visible light (a 500 W Xe lamp with a 420 nm cut-off filter, with the light intensity of 120 μW cm−2). 0.03 g of BiOCl sample was added into 40 mL mixed solution of RhB (5 × 10−6 mol L−1) and MO (1.5 × 10−5 mol L−1). Before illumination, the suspension was sonicated for 10 min and magnetically stirred for 30 min in the dark to ensure the establishment of an adsorption–desorption equilibrium. Then the suspension was exposed to visible light irradiation under magnetic stirring. At each time interval, 3 mL suspension was sampled and centrifuged to remove the BiOCl powder. The concentration variation of RhB and MO during the degradation was monitored from its characteristic absorption band (generally 554 nm for RhB and 464 nm for MO) using a Shimadzu UV2800 spectrophotometer. For comparison, single-component dye photodegradation of RhB (40 mL, 1 × 10−5 mol L−1) and MO (3 × 10−5 mol L−1) over the as-synthesized BiOCl was also performed under identical conditions. All of the measurements were carried out at room temperature.2.5 Active species trapping and ·O2− quantification experiment
In order to detect the active species during the visible-light-induced degradation process, 2 mM benzoquinone (BQ, a quencher of ·O2−), 2 mM KI (a quencher of h+) and 2 mM tert-butanol (t-BuOH, a quencher of ·OH) were applied to investigate the superoxide radicals (·O2−), the photo-induced holes (h+) and the hydroxyl radicals (·OH), respectively.26,27 In addition, N2 purging experiment was carried out to indirectly verify the role of dissolved oxygen in the dye degradation process. Nitrotetrazolium blue chloride (NBT, 2 × 10−5 M, with a maximum absorption at 259 nm) was utilized to determine the amount of ·O2− production by monitoring the NBT concentration change with a Shimadzu UV2800 spectrophotometer.28 The procedure for active species trapping and ·O2− quantification experiments were similar to the method of degradation activity evaluation.2.6 Electrochemical measurements
Cyclic voltammetry (CV) curves were measured at proper potential ranges with a scanning rate of 50 mV s−1, using glassy carbon as the working electrode, Pt wire as the counter electrode and Ag/Ag+ electrode as the pseudo-reference electrode, respectively. Samples were dissolved in CHCl3 solution with tetrabutylammonium hexafluorophosphate (TBAPF6, 0.1 mol L−1) as the supporting electrolyte. The ferrocenium/ferrocene (Fc/Fc+) redox couple was used as an internal standard.The photocurrent tests of BiOCl samples before and after RhB adsorption were performed in a three-electrode system on CHI660E electrochemical workstation (Shanghai Chenhua Instrument Co., Ltd., Shanghai, China). The prepared series photoelectrodes (1 cm × 1 cm), standard calomel electrode (SCE), and Pt electrode were respectively used as the working, reference, and counter electrodes. Na2SO4 aqueous solution (0.5 mol L−1) was used as the electrolyte. A 500 W xenon lamp was applied as light source with a 420 nm cut-off filter. The working electrode of BiOCl after RhB adsorption was prepared according to the following procedure: 10 mg BiOCl sample was well suspended in 1 mL of H2O containing 20 μL of 5% Nafion solution and 10 μL of RhB solution (1 × 10−3 mol L−1) under sonication for 4 h. Then it was evenly coated onto the conductive side of an indium–tin oxide (ITO) glass (1 cm × 1 cm). Finally, the ITO glass deposited with the suspension was annealed at 60 °C for 2 h. The working electrode of pure BiOCl was prepared with the same method with no addition of RhB solution.
3. Results and discussion
Fig. 1a shows the typical powder X-ray diffraction (XRD) pattern of the as-synthesized sample, which illustrates that all the diffraction peaks could be well indexed to pure tetragonal phase BiOCl (JCPDS no. 06-249) with lattice constants of a = 3.819 Å and c = 7.369 Å. No other phase and impurity peak were detected, indicative of high-purity of tetragonal BiOCl product. The detailed morphology and structure of the sample were characterized by SEM and TEM images. Large quantities of flower-like hierarchical BiOCl nanostructures composed of loosely packed nanoflakes with a thickness of about 8 nm were obtained (Fig. 1b and c). TEM image further provided information regarding the interior structures of the BiOCl hierarchical nanostructures (Fig. 1d). It was also observed that loose-packed flower-like BiOCl hierarchical nanostructures were fabricated. The specific surface area of the flower-like BiOCl nanostructure was also determined by using nitrogen adsorption and desorption isotherms. As shown in Fig. 1e, the representative N2 adsorption–desorption isotherm could be nearly categorized as type IV with a distinct hysteresis loop observed in the range of 0.5–1.0 P/P0,29 indicative of mesoporous structure. The BET surface area of the flower-like BiOCl hierarchical nanostructures calculated from the results of N2 isotherm was 37.1 m2 g−1. The corresponding average pore diameter of the BiOCl product was 22 nm, which was determined by the Barret–Joyner–Halenda (BJH) method (inset of Fig. 1e). The band gap of the as-prepared BiOCl HNs (S1) was determined to be 3.33 eV derived from the diffuse reflectance spectrum (DRS) of the sample (Fig. 1f). | ||
| Fig. 1 XRD pattern (a), SEM images (b and c), TEM image (d), N2 adsorption/desorption isotherm (e) and UV-Vis diffuse reflection spectra (f) of the flower-like BiOCl hierarchical nanostructures (S1). | ||
In this fabrication, citric acid acted as both a capping agent for inducing the crystal growth and a structure-directing agent for producing different structures of BiOCl. In the absence of citric acid (0 g, S2), Bi(NO3)3 reacted with deionized water and NaCl to form BiOCl nanoflakes (Fig. S1, ESI†). In the presence of small amount of citric acid (0.05 g, S3), citric acid molecules could be selectively adsorbed on the surface of BiOCl nanocrystals, and hydrogen bonds could be formed between the carboxyl of citric acid and the hydroxyl ions in [C6O7H5]3−Bi3+, as well as between the hydroxyl ions in [C6O7H5]3−Bi3+.30–32 However, only a few hydrogen bonds were formed, allowing the BiOCl crystallites to grow along one direction to form nanoplates (Fig. S1, ESI†). With increasing the [C6O7H5]3− concentration (0.2 g, S1), the strong hydrogen bonds between the contacting surfaces among the nanoplates could lead the primal nanoplates to densely pack with each other to form flower-like hierarchical nanostructures through self-assembly for minimizing the surface tension (Fig. 1b–d). However, the crucial role of citric acid in the formation of BiOCl HNs is a complicated issue and would be investigated by first principle calculation in the future work. Moreover, the chlorine precursor also had a strong influence on the structure of BiOCl HNs. When CoCl2 was used as chlorine precursor instead of NaCl, BiOCl hierarchical nanostructures were also obtained (S4, Fig. S2, ESI†). However, the BiOCl HNs displayed a closed-packed structure assembled by thicker nanosheets, which also possessed smaller BET surface areas than that of BiOCl HNs prepared from NaCl. This result is consistent with previous reported study.23
Based on the experimental results, it is believed that the flower-like BiOCl hierarchical nanostructures (S1) with higher BET surface area and loose-packed structure should be the best candidate for the dye-sensitized photodegradation, as confirmed by the corresponding degradation tests under identical conditions (Fig. S3, ESI†). Hence, the visible-light-driven photodegradation efficiency of RhB and MO over flower-like BiOCl HNs (S1) were evaluated, respectively. Fig. 2a and b show the changes of RhB absorption spectra and RhB concentration (C/C0) variation with irradiation time over the as-synthesized BiOCl HNs (S1) sample, respectively. For comparison, direct photolysis of RhB was also investigated under the identical condition. It illustrates that the flower-like BiOCl HNs could completely degrade RhB dye within 15 min upon visible light irradiation, while the RhB concentration hardly changed with the increase of irradiation time in the absence of BiOCl HNs. The results indicate that the as-synthesized BiOCl HNs (S1) were active visible-light-induced photocatalyst for RhB degradation. However, as shown in Fig. 2c and d, no direct photolysis or indirect BiOCl-induced photodegradation of MO was observed under the identical conditions, suggesting that the prepared BiOCl HNs (S1) exhibited poor efficiency for MO degradation under visible light.
 | ||
| Fig. 2 Photocatalytic activity of the as-synthesized flower-like BiOCl HNs (S1) for RhB (a and b) and MO (c and d) degradation under visible light irradiation. | ||
The DRS spectrum (Fig. 1f) of the as-synthesized flower-like BiOCl hierarchical nanostructure (S1) shows the absorption edge ranging from 310 nm to 360 nm, indicating the majorly UV light response property of the BiOCl sample. It illustrates that BiOCl HNs could hardly degrade organic dyes under visible-light irradiation. Therefore, the synthesized flower-like BiOCl HNs exhibits poor visible-light-induced photodegradation efficiency towards MO. However, the excellent adsorption capacity of BiOCl hierarchical microstructures for RhB molecules endows efficient photosensitized degradation performance for RhB, which was also reported in previous studies.23,25 Inspired by the previous work, we try to construct RhB dye-sensitized BiOCl photosensitization system for the degradation of MO dye. In order to further analyze the feasibility of utilizing RhB photosensitized BiOCl HNs for MO degradation, the matchability of band structure of the as-prepared BiOCl HNs and the lowest unoccupied molecular orbit (LUMO) and highest occupied molecular orbit (HOMO) levels of RhB molecules were demonstrated. Fig. 3a provides the detailed information about valence band (VB) and conduction band (CB) potentials of the as-synthesized BiOCl HNs by using cyclic voltammetry (CV) methods.33 The onset reduction potential (Ered) of the flower-like BiOCl HNs was −1.08 V relative to the Fc/Fc+ potential. Correspondingly, the conduction band potential (ECB) and valence band potential (EVB) were calculated according to the following formula (1) and (2), where Eg is the band gap energy of the semiconductor and 4.8 eV is the static energy difference value of Fc/Fc+ relative to vacuum level.
| ECB = −(Ered + 4.8) eV | (1) |
| EVB = −(Eg − ECB) eV | (2) |
 | ||
| Fig. 3 Cyclic voltammograms of BiOCl hierarchical nanostructure ((a), inset is band position of BiOCl HNs), RhB (b) and MO (c). | ||
Based on the eqn (1) and (2), the ECB and EVB of the BiOCl product are calculated to be −3.72 eV and −7.05 eV versus vacuum, respectively. The HOMO level energy of RhB was also determined by using cyclic voltammetry measurement.34,35 The CV curve of RhB in Fig. 3b shows that the onset oxidation potential (Eoxi) of RhB versus Fc/Fc+ is about 0.17 V. Therefore, the HOMO and LUMO level energies of RhB are calculated to be −4.97 and −2.73 eV versus vacuum level, respectively, according to the following eqn (3) and (4), where h, c and λ are the Planck constant, the speed of light in vacuum and the maximum absorption light wavelength of RhB (554 nm), respectively.
| EHOMO = −(Eoxi + 4.8) eV | (3) |
| ELUMO = EHOMO + hc/λ | (4) |
The results demonstrated that the conduction band (ECB = −3.72 eV) of the prepared BiOCl HNs located between the LUMO orbit (ELUMO = −2.73 eV) and the HOMO orbit (EHOMO = −4.97 eV) of RhB, indicative of the good matchability of the band structure of BiOCl and the LUMO and HOMO orbits of RhB. In contrast with RhB, the corresponding onset oxidation potential of MO was determined to be 0.05 V versus Fc/Fc+ as shown in Fig. 3c. Together with the maximum absorption light wavelength of MO molecules (464 nm), the HOMO and LUMO levels of MO could be estimated to be −4.85 and −2.18 eV versus vacuum, respectively. It also illustrates that the conduction band of BiOCl HNs located between the LUMO and HOMO orbits of MO molecules. Theoretically, electrons transfer from excited dye molecules to BiOCl HNs are allowed both for RhB and MO. Unfortunately, MO molecules couldn't be degraded under the condition due to the low MO adsorption efficiency over BiOCl HNs, which resulted in spatial separation of dye and material, further inhibiting the electron transfer. Therefore, the excellent RhB adsorption capacity of BiOCl HNs would also greatly contribute to the feasibility of constructing an RhB dye-sensitized BiOCl photosensitization system for MO degradation under visible light irradiation. The improved photodegradation efficiency of MO via RhB dye-sensitized BiOCl heterogeneous system is expected.
MO photodegradation over RhB-sensitized BiOCl HNs was evaluated under visible light irradiation. Fig. 4a shows the temporal evolution of UV-Vis spectra of RhB and MO. The two absorption bands which centered at 464 and 554 nm are ascribed to the electrons transition from highest occupied molecular orbital (HOMO) to lowest unoccupied molecular orbital (LUMO) of MO and RhB, respectively. Both of the two peaks decrease with the illumination time prolonging, demonstrating the dyes degradation process. Together with the variation of RhB and MO concentrations (C/C0) with the irradiation time in MO/RhB/BiOCl heterogeneous system (Fig. 4b), it was clearly observed that MO photodegradation efficiency was obviously enhanced in heterogeneous MO/RhB/BiOCl system, while RhB still remained high degradation efficiency in this process. For comparison, dyes photodegradation in homogeneous MO/RhB solution was also performed under identical conditions (Fig. 4b), which demonstrates that almost no MO or RhB dyes photodegradation occurred in the absence of BiOCl HNs. It indicates that MO photodegradation is mediated by RhB via photosensitization pathway over BiOCl HNs. It is well known that pH values of the solution can greatly influence the surface charges of a semiconductor and the photodegradation efficiency of the dyes. Consequently, we investigated the pH effect on the surface charges of the synthesized BiOCl HNs and the degradation efficiency. As illustrated Fig. S4 (ESI†), the as-synthesized BiOCl shows negative zeta potential in wide pH range from 2 to 11, and positive zeta potential in a narrow pH range (pH < 2), indicating that the as-synthesized BiOCl in RhB solution is net negative charge, and could selectively adsorb more anions. Fig. S5 (ESI†) illustrates the photodegradation efficiency of the RhB-sensitized BiOCl HNs at pH 3.36 and pH 11.08, respectively. Both RhB and MO displays efficient degradation within 5 min upon visible light irradiation when pH value is 3.36 (Fig. S5a, ESI†). However, under alkalic solution with pH 11.08 (Fig. S5b, ESI†), the degradation efficiency for both RhB and MO obviously decreased. It was proposed that more cationic ions dissociated from RhB would be pushed into the Stern layer (compact layer) with increasing the concentration of H+ ions, due to the electrostatic repulsion, thus facilitating the electrons transfer from RhB to BiOCl, and improving the photodegradation efficiency of system. However, with increasing the concentration of OH− ions, more cationic ions would be kept in the diffusion layer due to the electrostatic attraction and hindered the electrons transfer from RhB to BiOCl. As a result, it can be reasonably inferred that more acidic condition would be benefic for efficient degradation.
In order to clarify the mechanism for enhanced MO photodegradation efficiency in RhB dye-sensitized BiOCl system, the involving active species in the photocatalysis were firstly investigated through trapping experiment. Benzoquinone (BQ), KI and tert-butanol (t-BuOH) were utilized as scavengers to determine the presence of superoxide radicals (·O2−), photo-generated holes (h+) and the hydroxyl radicals (·OH), respectively. As shown in Fig. 5, only BQ obviously inhibits both RhB and MO photodegradation. Almost no change in RhB and MO photodegradation efficiency was observed in the presence of KI and t-BuOH, demonstrating that the major active specie was ·O2−, instead of h+ and ·OH. In N2 purging experiment, it was also found MO photodegradation efficiency slightly decreased (Fig. 5, left side of the dotted line). It suggests that the decolorization process of RhB would be followed by O2− independent de-ethyl pathway, as previously reported work.12 And the MO photodegradation process strongly depended on ·O2− production, which further illustrates the photosensitization process. The production of ·O2− over pure BiOCl and RhB dye-sensitized BiOCl system was also quantitatively determined by monitoring the concentration variation of nitrotetrazolium blue chloride (NBT). After 2 h visible light irradiation, the NBT transformation percentage in RhB/BiOCl system was 20%, which was ten times higher than that of 2% in pure BiOCl system. The result illustrates that more ·O2− was generated in RhB-sensitized BiOCl HNs, which significantly contributes to the enhanced MO degradation efficiency.
The results demonstrate that MO degradation over RhB-sensitized BiOCl HNs undergoes via photogenerated electrons process, which come from excited RhB dye molecules. To reveal the interfacial electron transfer dynamics, photocurrent responses as well as photoluminescence of BiOCl HNs and RhB-sensitized BiOCl HNs were evaluated accordingly. As shown in Fig. 6a, RhB-sensitized BiOCl HNs display distinctly higher photocurrent response than pure BiOCl HNs under identical conditions, suggesting more photo-induced electrons were generated in RhB-sensitized BiOCl system, which might be derived from the electrons transfer from excited RhB dye molecules to the surface of BiOCl HNs. In accord with the electron injection process, electrons transfer from RhB to conduction band of BiOCl would cause the quenching of RhB photoluminescence. Fig. 6b shows the photoluminescence spectroscopy of RhB and RhB/BiOCl with an excitation wavelength of 500 nm. A strong and luminous broad peak ranged from 560 to 600 nm was observed in the PL spectrum of RhB. However, the light-emitting broad peak was quenched in the PL spectrum of RhB/BiOCl system. It illustrates that the photo-generated electrons could transfer from RhB molecules to BiOCl HNs, which subsequently reduce the surface adsorbed O2 molecules on BiOCl to generate ·O2− radicals. The interfacial electrons transfer between RhB dye and BiOCl HNs greatly facilitated the efficiency of RhB-sensitized BiOCl HNs for MO degradation.
 | ||
| Fig. 6 Transient photocurrent response of RhB-sensitized BiOCl and pure BiOCl system under visible light irradiation (a) and photoluminescence spectra of RhB and RhB/BiOCl system (b). | ||
According to the experimental results, the proposed mechanism for enhanced MO photodegradation efficiency via heterogeneous RhB dye-sensitized BiOCl HNs is schematically illustrated in Scheme 1. Firstly, once RhB dye molecules were excited upon visible light irradiation, the electrons were injected from higher locating LUMO orbit of RhB (ELUMO = −2.73 eV vs. vacuum) to the lower locating conduction band (CB) of BiOCl semiconductor (ECB = −3.72 eV vs. vacuum). Due to the higher location of BiOCl conduction band compared with the redox level of O2/·O2− (E0(O2/·O2−) = −4.45 eV vs. vacuum), electrons on CB of BiOCl semiconductor could subsequently reduce the surface adsorbed O2 to ·O2− radicals. Then organic dyes pollutants were efficiently degraded through ·O2− oxidation. However, pure BiOCl semiconductor could not be excited by visible light due to the wide band gap and showed extremely low MO adsorption capacity, which inhibited the electron transfer from excited MO molecules to CB of BiOCl. As a result, no MO degradation occurred over pure BiOCl catalyst. Consequently, electron transfer from excited RhB to CB of BiOCl yielded the active species generation of ·O2− and resulted in the enhanced MO photodegradation efficiency in RhB-sensitized BiOCl system.
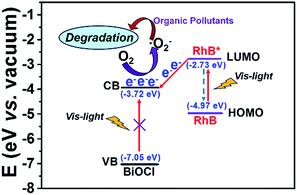 | ||
| Scheme 1 Proposed mechanism for the enhanced MO photodegradation efficiency in RhB-sensitized BiOCl heterogeneous system under visible light irradiation. | ||
4. Conclusions
In summary, flower-like BiOCl hierarchical nanostructures assembled by thin nanosheets have been synthesized via a facile solvothermal method. A high efficient BiOCl-based photosensitization system has been developed by using RhB as sacrifice photosensitizer. The matchability between band structure of BiOCl HNs and molecular orbit of RhB endows the RhB dye-sensitized BiOCl HNs possess highly enhanced photodegradation efficiency for MO dye, which could be hardly decomposed over pure BiOCl semiconductor catalyst. The electrons transfer from LUMO orbit of RhB to CB of BiOCl HNs facilitated the yield of superoxide radicals, which was the main active species for MO degradation and resulted in enhanced MO photodegradation efficiency over RhB-sensitized BiOCl. This study not only provides insight into the photodegradation mechanism in a dye-sensitized BiOCl heterogeneous system, but also develops a strategy for environmental pollutants removal via dye-sensitized semiconductor heterogeneous catalysts.Acknowledgements
This work was supported by the National Natural Science Foundation of China (21471121, 21171136), and High-Tech Industry Technology Innovation Team Training Program of Wuhan Science and Technology Bureau (2014070504020243).References
- C. Chen, W. Ma and J. Zhao, Chem. Soc. Rev., 2010, 39, 4206–4219 RSC.
- J. Schneider, M. Matsuoka, M. Takeuchi, J. Zhang, Y. Horiuchi, M. Anpo and D. W. Bahnemann, Chem. Rev., 2014, 114, 9919–9986 CrossRef CAS.
- H. Zhao, F. Tian, R. Wang and R. Chen, Reviews in Advanced Sciences and Engineering, 2014, 3, 3–27 CrossRef.
- H. Fang, Y. Gao, G. Li, J. An, P. K. Wong, H. Fu, S. Yao, X. Nie and T. An, Environ. Sci. Technol., 2013, 47, 2704–2712 CrossRef CAS.
- G. Li, Y. Ding, Y. Zhang, Z. Lu, H. Sun and R. Chen, J. Colloid Interface Sci., 2011, 363, 497–503 CrossRef CAS.
- J. Li, L. Zhang, Y. Li and Y. Yu, Nanoscale, 2014, 6, 167–171 RSC.
- F. Qin, G. Li, R. Wang, J. Wu, H. Sun and R. Chen, Chem.–Eur. J., 2012, 18, 16491–16497 CrossRef CAS PubMed.
- J. Jiang, K. Zhao, X. Xiao and L. Zhang, J. Am. Chem. Soc., 2012, 134, 4473–4476 CrossRef CAS.
- M. Yin, Z. Li, J. Kou and Z. Zou, Environ. Sci. Technol., 2009, 43, 8361–8366 CrossRef CAS PubMed.
- J. Xiong, G. Cheng, G. Li, F. Qin and R. Chen, RSC Adv., 2011, 1, 1542–1553 RSC.
- D. Wang, G. Gao, Y. Zhang, L. Zhou, A. Xu and W. Chen, Nanoscale, 2012, 4, 7780–7785 RSC.
- C. Chen, W. Zhao, P. Lei, J. Zhao and N. Serpone, Chem.–Eur. J., 2004, 10, 1956–1965 CrossRef CAS.
- K. Zhao, L. Zhang, J. Wang, Q. Li, W. He and J. Yin, J. Am. Chem. Soc., 2013, 135, 15750–15753 CrossRef CAS.
- J. Xiong, G. Cheng, F. Qin, R. Wang, H. Sun and R. Chen, Chem. Eng. J., 2013, 220, 228–236 CrossRef CAS.
- M. Guan, C. Xiao, J. Zhang, S. Fan, R. An, Q. Cheng, J. Xie, M. Zhou, B. Ye and Y. Xie, J. Am. Chem. Soc., 2013, 135, 10411–10417 CrossRef CAS.
- Y. I. Choi, Y.-I. Kim, D. W. Cho, J.-S. Kang, K. T. Leung and Y. Sohn, RSC Adv., 2015, 5, 79624–79634 RSC.
- Y. I. Choi, H. J. Jung, W. G. Shin and Y. Sohn, Appl. Surf. Sci., 2015, 356, 615–625 CrossRef CAS.
- H. J. Yoon, Y. I. Choi, E.-S. Jang and Y. Sohn, J. Ind. Eng. Chem., 2015, 32, 137–152 CrossRef CAS.
- L. Ye, K. Deng, F. Xu, L. Tian, T. Peng and L. Zan, Phys. Chem. Chem. Phys., 2012, 14, 82–85 RSC.
- X. Zhang, X. Wang, L. Wang, W. Wang, L. Long, W. Li and H. Yu, ACS Appl. Mater. Interfaces, 2014, 6, 7766–7772 CAS.
- G. Li, F. Qin, R. Wang, S. Xiao, H. Sun and R. Chen, J. Colloid Interface Sci., 2013, 409, 43–51 CrossRef CAS PubMed.
- K. Zhang, J. Liang, S. Wang, J. Liu, K. Ren, X. Zheng, H. Luo, Y. Peng, X. Zou, X. Bo, J. Li and X. Yu, Cryst. Growth Des., 2012, 12, 793–803 CAS.
- F. Tian, Y. Zhang, G. Li, Y. Liu and R. Chen, New J. Chem., 2015, 39, 1274–1280 RSC.
- J. Xiong, G. Cheng, R. Wang, H. Yang, S. Xiao and R. Chen, Sci. Adv. Mater., 2013, 5, 1024–1031 CrossRef CAS.
- F. Tian, H. Zhao, Y. Liu, S. Xiao and R. Chen, CrystEngComm, 2014, 16, 4298–4305 RSC.
- Y. Tian, B. Chang, J. Lu, J. Fu, F. Xi and X. Dong, ACS Appl. Mater. Interfaces, 2013, 5, 7079–7085 CAS.
- S. Wang, D. Li, C. Sun, S. Yang, Y. Guan and H. He, Appl. Catal., B, 2014, 144, 885–892 CrossRef CAS.
- L. Ye, J. Liu, Z. Jiang, T. Peng and L. Zan, Appl. Catal., B, 2013, 142, 1–7 Search PubMed.
- J. Zhang, F. Shi, J. Lin, D. Chen, J. Gao, Z. Huang, X. Ding and C. Tang, Chem. Mater., 2008, 20, 2937–2941 CrossRef CAS.
- J. A. Parkinson and H. Sun, Chem. Commun., 1998, 881–882 RSC.
- Y. Huo, Y. Jin and Y. Zhang, J. Mol. Catal. A: Chem., 2010, 331, 15–20 CrossRef CAS.
- K. Zhang, J. Liang, S. Wang, J. Liu, K. Ren, X. Zheng, H. Luo, Y. Peng, X. Zou and X. Bo, Cryst. Growth Des., 2012, 12, 793–803 CAS.
- Y. Myung, F. Wu, S. Banerjee, J. Park and P. Banerjee, Chem. Commun., 2015, 51, 2629–2632 RSC.
- Y. Kamei, N. Robertson, Y. Shichibu and K. Konishi, J. Phys. Chem. C, 2015, 119, 10995–10999 CAS.
- W. Sharmoukh, K. C. Ko, S. Y. Park, J. H. Ko, J. M. Lee, C. Noh, J. Y. Lee and S. U. Son, Org. Lett., 2008, 10, 5365–5368 CrossRef CAS PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra24887f |
| ‡ These authors make equal contribution to this work. |
| This journal is © The Royal Society of Chemistry 2016 |


