Modeling solvent-activated shape-memory behaviors based on an analogy between solvent and temperature
Rui Xiao
Institute of Soft Matter Mechanics, College of Mechanics and Materials, Hohai University, Nanjing, Jiangsu 210098, China. E-mail: rxiao@hhu.edu.cn
First published on 22nd December 2015
Abstract
For amorphous polymers, the thermally-activated and solvent-activated shape-memory effect share the same physical mechanism. The programmed shape can be recovered either by increasing the environment temperature above the activation temperature (thermally-activated mechanism) or decreasing the activation temperature below the ambient temperature (solvent-activated mechanism). An equivalent role exists for solvent and heat in activating shape recovery. Based on this assumption, we presented a method to simulate solvent-activated shape-memory behaviors through the widely available models developed for the thermally-activated mechanism. The recovery in the solvent was treated as increasing the temperature of the specimen, while the diffusion of the solvent into the polymer matrix was analogous to heat conduction. The model was employed to simulate the solvent-activated temperature memory effect of Nafion in acetone and ethanol. The model predictions showed good agreement with the experimental results.
1 Introduction
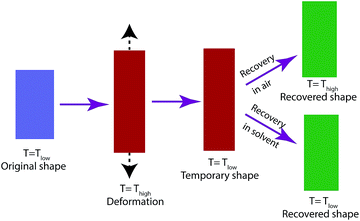
Shape-memory polymers (SMPs) are soft active materials that have greatly attracted researcher interest due to their broad applications in biomedical and industrial areas.1–5 Among all the different types of SMPs, amorphous SMPs are most widely investigated due to their simplicity of synthesis and mechanical characterization.6–9 The physical mechanism underlying the shape-memory effect of amorphous SMPs is the tremendous mechanical properties change through glass transition.10,11 In a typical shape-memory cycle, the polymers are deformed above the glass transition temperature (Tg) and cooled down below Tg to fix the temporary shape. To achieve shape recovery, the polymers can either be heated above Tg (ref. 12–14) or be immersed in solvent to reduce Tg below the ambient temperature15–17 (Fig. 1). Thus the underlying mechanism is the same for both activation methods.Early experimental characterization of solvent-activated shape-memory behaviors focused on water-polymer systems.16,18–22 Huang and coworkers found that the Tg of polyurethane SMPs decreased with increasing immersion time in water.18 Chen and coworkers19 synthesized novel polyurethane SMPs, which showed the good absorption of moisture. The corresponding moisture-activated shape recovery depended on the relative humidity and ambient temperature.19 Xiao and Nguyen20 demonstrated that programmed acrylate copolymers can achieve shape recovery in water. The equilibrium concentration of water or moisture is typically small (less than 10%) in the above polymer systems. In contrast, large amounts of organic solvent can diffuse into the polymer matrix and activate shape recovery.23–26 The recovery ratio and recovery rate depends on the solvent types24,25 as well as the deformation temperature.25
Compared with the continuously growing work on experimental characterization, the efforts to model solvent-activated shape-memory behaviors have lagged. Lu et al.27 developed a phenomenological model on the basis of a permeation transition model. The model did not incorporate the kinetics of the diffusion process. Xiao and Nguyen20 developed a coupled chemical–mechanical model and implemented the model into finite element software including the diffusion process. However, the model can only be applied to low concentration solvent–polymer systems. There still lacks a model that can describe the time-dependent recovery of polymers in organic solvents. In contrast, the amount of work modeling thermally-activated shape-memory behaviors has greatly expanded.28–33 Numerous three-dimensional finite deformation constitutive models have been developed30–33 and can be implemented into finite element software. These models are capable of simulating complex three dimensional shape recovery performance under various thermo-mechanical programming and recovery conditions.
In this paper, we demonstrated that the model developed for thermally-activated SMPs can also be applied to describe the solvent-activated shape-memory behaviors. We made the following assumptions: increasing the solvent concentration in polymers has the same effect on viscosity as increasing the temperature; the diffusion process can be approximated as the heat conduction process. To validate this method, we adopted the constitutive model reported by Xiao et al.34 which was previously developed for studying the temperature memory and multiple shape-memory effect of Nafion, to simulate the shape recovery performance of Nafion in solvent. Experimentally we measured the influence of the programmed temperature on the shape recovery of Nafion films in acetone and ethanol. We further performed a parameter study to investigate the effect of the diffusion coefficient, equivalent temperature of solvent and specimen thickness on the shape recovery performance.
2 Experimental methods
The experimental methods were described in detail by Xiao et al.25 Here we briefly describe the experimental procedures. The material adopted in this study is a Dupont Nafion® PFSA membrane with an equivalent molecular weight of 1100 (Dupont, Wilmington, DE, USA). Before all the tests, the specimens were annealed at 160 °C for half an hour to get rid of the bound water in the specimens. The specimen size used in the following tests was 10 mm × 10 mm × 0.25 mm if not otherwise specified.2.1 Swelling tests
The film specimens with size 4.0 × 4.0 × 0.25 mm3 were used to measure the swelling rate of Nafion in solvents. The specimens were placed in acetone or ethanol, removed periodically and weighted using a digital balance with 10−4 g resolution. The swelling ratio is defined as:
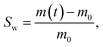 | (1) |
2.2 Solvent-induced temperature memory effect
The film specimens were stretched to 60% engineering strain at 160, 140, 120, 100 or 80 °C in two minutes on a Dynamic Mechanical Analyzer (DMA) Q800 (TA Instruments, New Castle, DE, USA). The specimens were then cooled to 20 °C at 10 °C min−1 and annealed for 5 minutes before unloading. The programmed specimens were unmounted from the DMA and immersed either in acetone or ethanol at room temperature. A digital camera was used to record the temporary shape of the specimens. Nafion can absorb 37 ± 4% acetone and 116 ± 10% ethanol25 of its own weight. The size of the specimens can be changed by the solvent so it is not possible to define the shape recovery ratio through the length change. Thus, a square mark was labeled on the undeformed specimen in the length and width direction. We used the aspect ratio between the width W and length L of the marker to represent the shape recovery ratio as: D = W/L, where D = 1 indicates fully recovery.2.3 Dual programming shape recovery
A dual programming shape recovery test was performed on a cruciform specimen. The specimen was first stretched in one direction to 60% engineering strain at 160 °C on the DMA Q800. The specimen was then cooled to 20 °C and unmounted from the DMA. The specimen was remounted on the DMA in the perpendicular direction. The specimen was heated to 80 °C and annealed for 10 minutes. The specimen was stretched another 60% engineering strain and cooled to 20 °C. The specimen was then transferred into acetone or ethanol at room temperature to achieve shape recovery.3 Numerical methods
3.1 Constitutive model
To model the 3D finite deformation of polymers, we first define the deformation gradient F mapping the material lines in the undeformed reference configuration to the spatial lines in the current configuration. To model the broad viscoelastic response, the deformation gradient is multiplicatively split into N pairs of elastic and viscous parts, F = FeiFvi, i = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) N. To represent the inherent difference of polymers in response to the shear and volumetric deformation, the deformation gradient is decomposed into distortional and volumetric parts, as F = J1/3
N. To represent the inherent difference of polymers in response to the shear and volumetric deformation, the deformation gradient is decomposed into distortional and volumetric parts, as F = J1/3![[F with combining macron]](https://www.rsc.org/images/entities/b_char_0046_0304.gif) and Fei = Je1/3i
and Fei = Je1/3i![[F with combining macron]](https://www.rsc.org/images/entities/b_char_0046_0304.gif) ei, where J = det(F) and Jei = det(Fei) are the total and elastic part of the volumetric deformation ratio. We then define the total and elastic deviatoric left Cauchy-Green deformation tensors
ei, where J = det(F) and Jei = det(Fei) are the total and elastic part of the volumetric deformation ratio. We then define the total and elastic deviatoric left Cauchy-Green deformation tensors ![[b with combining macron]](https://www.rsc.org/images/entities/b_char_0062_0304.gif) =
= ![[F with combining macron]](https://www.rsc.org/images/entities/b_char_0046_0304.gif)
![[F with combining macron]](https://www.rsc.org/images/entities/b_char_0046_0304.gif) T and
T and ![[b with combining macron]](https://www.rsc.org/images/entities/b_char_0062_0304.gif) ei =
ei = ![[F with combining macron]](https://www.rsc.org/images/entities/b_char_0046_0304.gif) ei
ei![[F with combining macron]](https://www.rsc.org/images/entities/b_char_0046_0304.gif) eTi. We also define the rate of viscous deformation tensor and its symmetric part as, Lvi = ḞviFv−1i and
eTi. We also define the rate of viscous deformation tensor and its symmetric part as, Lvi = ḞviFv−1i and 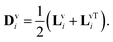
Stress σ is represented by an equilibrium distortional component, N nonequilibrium distortional components, and a time-independent volumetric component,
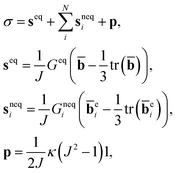 | (2) |
The following nonlinear evolution equation is adopted for Dvi,35
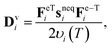 | (3) |
According to time–temperature superposition, υi(T) can be represented as υi(T) = υrefia(T), where υrefi is the characteristic viscosity at the reference temperature and a(T) is the shift factor. We further define the characteristic stress relaxation time as τrefi = υrefi/Gneqi.
3.2 Finite element model
The above constitutive model is implemented into finite element code TAHOE. Fick’s second law was adopted for heat conduction to represent solvent diffusion,
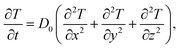 | (4) |
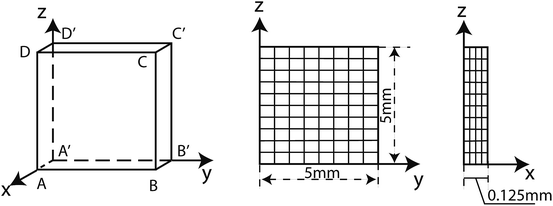
We employed the model to study the shape-memory behaviors of Nafion films as shown in Fig. 2. Due to symmetry, we only simulated one eighth of the whole specimen. The shape was discretized using hexahedral elements. The displacement boundary conditions for the film was set as ux(x = 0, y, z) = 0, uy(x, y = 0, z) = 0, uz(x, y, z = 0) and uz(x, y, z = 5) = u(t). The boundary conditions for displacement and temperature for the programming step were the same as in the experiment setup described in Section 2. At the beginning of the recovery process, the temperature at the surfaces ABCD, BB′C′C and CC′D′D was ramped from room temperature (25 °C) to the equivalent temperature Te in 2 seconds and during the recovery process all the surfaces were set as traction free. At each time step, both the mechanical part and heat conduction part were solved. An iterative staggered scheme was employed to couple the mechanical process and heat conduction process to guarantee that the solution for both processes got converged.
4 Model parameters
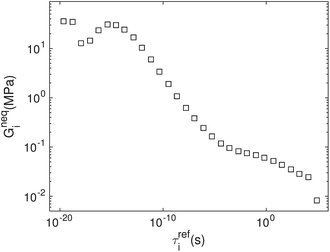
The parameters of the constitutive model for Nafion were obtained through the stress relaxation tests described in detail by Xiao et al.34 In brief, we performed stress relaxation tests at multiple temperatures and constructed the master curve of relaxation modulus at the reference temperature through time–temperature superposition. We then obtained the continuous spectrum from the master curve. The discrete relaxation modulus was obtained by the staircase approximation of the cumulative relaxation spectrum of the discrete model to continuous cumulative relaxation spectrum. The parameters for Geq and κ are listed in Table 1. The discrete relaxation spectrum is shown in Fig. 3. The shift factor a(T) was obtained through fitting a third order polynomial function to the experimentally measured shift factor, which is represented as,| log(a(T)) = 2.76 × 10−6T3 − 4.78 × 10−4T2 − 0.171T + 28.2. | (5) |
| Parameter | Values | Physical significance |
|---|---|---|
| Geq (MPa) | 0.087 | Equilibrium shear modulus |
| κ (MPa) | 744.4 | Bulk modulus |
| Te1 (°C) | 125 | Equivalent temperature of acetone |
| Te2 (°C) | 135 | Equivalent temperature of ethanol |
| D01 (mm2 s−1) | 6 × 10−4 | Thermal diffusivity to represent diffusion coefficient of acetone |
| D02 (mm2 s−1) | 2 × 10−4 | Thermal diffusivity to represent diffusion coefficient of ethanol |
The equivalent temperature Te and the diffusion coefficient (thermal diffusivity) D0 are obtained by fitting to the experimentally measured shape recovery of specimens programmed at 140 °C.
5 Results and discussion
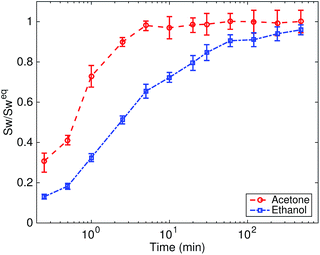
Fig. 4 plots the normalized swelling ratio of Nafion in acetone and ethanol with time. The equilibrium swelling ratio of Nafion is 37 ± 4% in acetone and 116 ± 10% in ethanol. The finite element model was applied to study the recovery behaviors of the programmed Nafion specimens in acetone and ethanol. Fig. 5 compares the experimental results and model predictions of the recovery response of Nafion in acetone and ethanol. The specimens programmed at 80 °C and 100 °C showed a similar recovery response. Thus, only the results of the specimens programmed at 80 °C are presented. In general, the model predictions showed good quantitative agreement with the experimental observations. Specifically, the simulation accurately predicted the final recovery ratio and initial recovery rate for both recovery in acetone and ethanol. For specimens programmed at lower temperature (80 and 120 °C), the simulation predicted a faster recovery rate than the experimental results.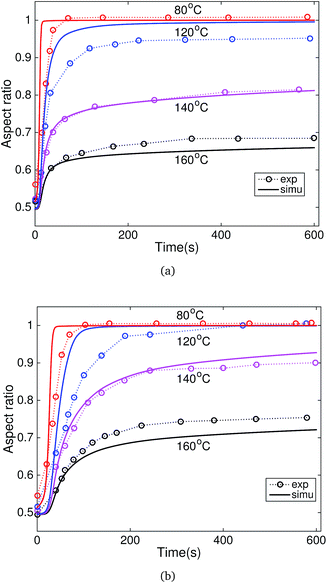 | ||
| Fig. 5 Comparison of the experimental results and model predictions of the recovery response of Nafion in (a) acetone and (b) ethanol (black: 160 °C, mauve: 140 °C, blue: 120 °C, red: 80 °C.). | ||
As shown in Fig. 5, the deformation temperature had a strong effect on the recovery behaviors. The specimens programmed at lower temperatures exhibited a larger recovery ratio. For example, the specimens programmed at 80 °C and 100 °C can achieve full recovery in both acetone and ethanol, while the specimens programmed at 140 °C and 160 °C can only achieve partial recovery. This temperature memory effect originated from the broad distribution of the relaxation time. For specimens programmed at lower temperatures, the temporary shape was fixed by the relaxation processes with a smaller relaxation time. Thus, the specimens programmed at lower temperatures can achieve a larger ratio of shape recovery. In addition to the programming temperature, the solvent type also had a strong influence on recovery behaviors. Compared with acetone, ethanol is a more efficient solvent to activate the shape recovery of the programmed Nafion films, represented as a larger recovery ratio. It can also be seen from the swelling results that Nafion can absorb more ethanol than acetone.
The recovery performance of the specimens programmed with two temporary shapes at both 160 °C and 80 °C is shown in Fig. 6. Both experimental results and model predictions showed that the recovery ratio first decreased to the minimum value, and then gradually increased to the steady state value. The decrease of the recovery ratio represented the recovery of the shape programmed at 80 °C. The recovery of the shape programmed at 160 °C dominated in the late recovery stages since the shape programmed at 80 °C had achieved full recovery. The simulation predicted a higher initial aspect ratio than the experimental data. One possible reason is the inhomogeneous deformation of the specimen during the second loading. We found that the surface of the specimen is not perfectly smooth when remounting onto the machine to program the second temporary shape.
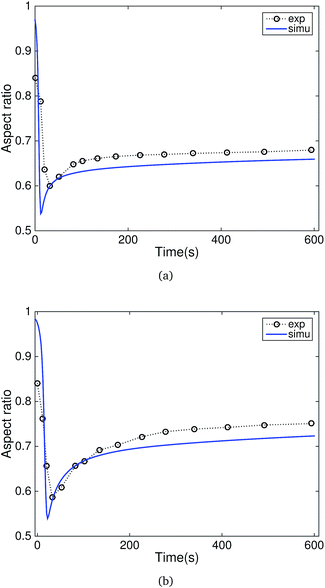 | ||
| Fig. 6 Comparison of the experimental results and simulation of the recovery of Nafion programmed with two temporary shapes in (a) acetone and (b) ethanol. | ||
As shown in Fig. 5 and 6, the solvent type has an effect on the recovery performance. Meanwhile, the diffusion coefficient of the solvent in polymers directly determines the initial recovery time and recovery rate. Thus we performed a parameter study to investigate these effects on the recovery performance. We adopted the test of the Nafion specimen programmed at 160 °C and recovered in acetone as the base experiment. Fig. 7a plots the influence of the diffusion coefficient on the recovery performance. A larger diffusion coefficient resulted in a smaller activation time and a larger initial recovery rate. We then investigated the influence of the specimen thickness on the recovery behaviors. As shown in Fig. 7b, increasing the specimen thickness has a similar effect as decreasing the diffusion coefficient. For the solvent-activated mechanism, two key factors determine the recovery behaviors: viscosity and solvent diffusion. The parameter study on specimen thickness provides a method to separate the effect of viscosity and diffusion on the recovery behaviors. As demonstrated in Fig. 7b, the shape recovery behaviors of the thin specimens are dominated by the influence of viscosity, while the recovery of the thick specimens is mainly controlled by the diffusion process. We then studied the effect of different solvents on viscosity by adjusting the Te as shown in Fig. 7c. The recovery ratio is determined by the equivalent recovery temperature Te, with only a small fraction of recovery at Te = 95 °C and nearly full recovery at Te = 155 °C.
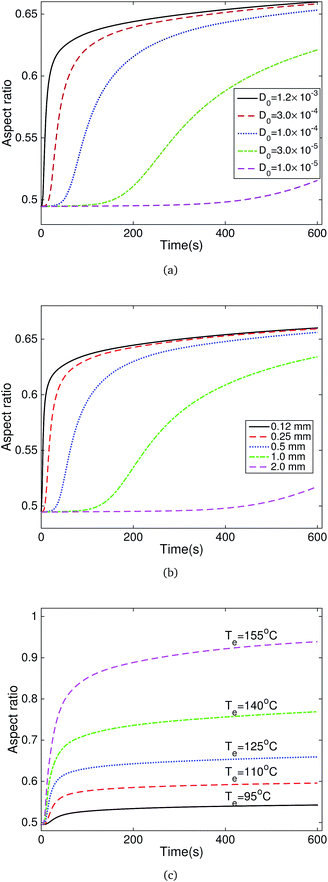 | ||
| Fig. 7 Parameter influence on the recovery behaviors: (a) diffusion constant, (b) specimen thickness and (c) recovery temperature. | ||
From the above results, we can see this method is able to simulate the solvent-activated shape-memory behaviors. However, there are still some limitations of the method which can be improved in the future. First, the current model does not consider the deformation caused by swelling. Thus it can not be adopted to simulate the force response during the constrained recovery. This can be improved by adopting thermal deformation to represent the deformation caused by swelling. Second, a constant diffusion coefficient is adopted for the diffusion process. As discussed in ref. 36, the diffusion coefficient may depend on the solvent concentration. This can also be improved by employing temperature-dependent thermal diffusivity. Third, we performed the shape recovery experiments only at room temperature. The experimental results in ref. 19 and 20 show that the recovery ratio and recovery rate increases with the recovery temperature. Thus the equivalent temperature Te may also be dependent on the recovery temperature. In this work, the specimens did not exhibit mechanical instability such as buckling or wrinkling, which has been observed in previous work.37 In order to apply our method to describe the mechanical instability in the solvent-activated shape recovery processes, the initial perturbation has to be incorporated in the simulation such as geometry imperfection.34,38
6 Conclusions
We presented a method that adopts the constitutive models for thermally-activated SMPs to simulate the solvent-activated shape-memory effect based on the assumption that the solvent plays the same role as temperature during the shape recovery process. The model was adopted to simulate the deformation temperature influence on the Nafion recovery in organic solvent, acetone and ethanol. The model prediction showed good agreement with experimental observations. The parameter study shows that the viscosity and diffusion process are the main factors controlling the recovery response. This method can be easily applied to commercial finite element software and provides an efficient way to simulate the solvent effect on thermomechanical properties and shape-memory performance.Acknowledgements
R. Xiao acknowledges the funding support from the National Natural Science Foundation of China (Grant No. 11502068).References
- K. Gall, C. M. Yakacki, Y. Liu, R. S. Adn, N. Willet and K. S. Anseth, J. Biomed. Mater. Res., Part A, 2005, 73, 339–348 CrossRef PubMed.
- W. Sokolowski, A. Metcalfe, S. Hayashi, L. Yahia and J. Raymond, Mater. Today, 2007, 2, S23–S27 CAS.
- Y. Liu, H. Du, L. Liu and J. Leng, Smart Mater. Struct., 2014, 23, 023001 CrossRef.
- P. T. Mather, X. Luo and I. Rousseau, Annu. Rev. Mater. Res., 2009, 39, 445–471 CrossRef CAS.
- W. Huang, Open Med. Dev. J., 2010, 2, 11–19 CrossRef.
- C. Liu, H. Qin and P. T. Mather, J. Mater. Chem., 2007, 17, 1543–1558 RSC.
- M. Behl and A. Lendlein, Mater. Today, 2007, 10, 20–28 CrossRef CAS.
- C. M. Yakacki, R. Shandas, C. Lanning, B. Rech, A. Eckstein and K. Gall, Biomaterials, 2007, 28, 2255–2263 CrossRef CAS PubMed.
- C. M. Yakacki, R. Shandas, D. Safranski, A. M. Ortega, K. Sassaman and K. Gall, Adv. Funct. Mater., 2008, 18, 2428–2435 CrossRef CAS PubMed.
- T. Nguyen, H. J. Qi, F. Castro and K. N. Long, J. Mech. Phys. Solids, 2008, 56, 2792–2814 CrossRef CAS.
- T. D. Nguyen, Polym. Rev., 2013, 53, 130–152 CrossRef CAS.
- R. Xiao, J. Choi, N. Lakhera, C. Yakacki, C. Frick and T. Nguyen, J. Mech. Phys. Solids, 2013, 61, 1612–1635 CrossRef.
- T. Xie, Nature, 2010, 464, 267–270 CrossRef CAS.
- K. Yu, Q. Ge and H. J. Qi, Nat. Commun., 2014, 5, 1–9 Search PubMed.
- B. Yang, W. Huang, C. Li, C. Lee and L. Li, Smart Mater. Struct., 2003, 13, 191 CrossRef.
- W. Huang, B. Yang, L. An, C. Li and Y. Chan, Appl. Phys. Lett., 2005, 86, 114105 CrossRef.
- H. Lv, J. Leng, Y. Liu and S. Du, Adv. Eng. Mater., 2008, 10, 592–595 CrossRef.
- B. Yang, W. Huang, C. Li and L. Li, Polymer, 2006, 47, 1348–1356 CrossRef CAS.
- S. Chen, J. Hu, C. Yuen and L. Chan, Polymer, 2009, 50, 4424–4428 CrossRef CAS.
- R. Xiao and T. D. Nguyen, Soft Matter, 2013, 9, 9455–9464 RSC.
- X. Gu and P. T. Mather, RSC Adv., 2013, 3, 15783–15791 RSC.
- Q. Bai, G. Zhang, B. Xu, X. Feng, H. Jiang and H. Li, RSC Adv., 2015, 5, 91213–91217 RSC.
- H. Lu, Y. Liu, J. Leng and S. Du, Eur. Polym. J., 2010, 46, 1908–1914 CrossRef CAS.
- H. Du and J. Zhang, Soft Matter, 2010, 6, 3370–3376 RSC.
- R. Xiao, J. Guo, D. L. Safranski and T. D. Nguyen, Soft Matter, 2015, 11, 3977–3985 RSC.
- Y. Zhao, C. C. Wang, W. M. Huang and H. Purnawali, Appl. Phys. Lett., 2011, 99, 131911 CrossRef.
- H. Lu, W. M. Huang and J. Leng, Smart Mater. Struct., 2014, 23, 045038 CrossRef.
- H. Tobushi, K. Okamura, S. Hayashi and N. Ito, Mech. Mater., 2001, 33, 545–554 CrossRef.
- C. P. Buckley, C. Prisacariu and A. Caraculacu, Polymer, 2007, 48, 1388–1396 CrossRef CAS.
- J. Diani, P. Gilormini, C. Frédy and I. Rousseau, Int. J. Solids Struct., 2012, 49, 793–799 CrossRef.
- T. Nguyen, C. M. Yakacki, P. D. Brahmbhatt and M. L. Chambers, et al., Adv. Mater., 2010, 22, 3411–3423 CrossRef CAS PubMed.
- G. Li and W. Xu, J. Mech. Phys. Solids, 2011, 59, 1231–1250 CrossRef CAS.
- K. K. Westbrook, P. H. Kao, F. Castro, Y. Ding and H. Jerry Qi, Mech. Mater., 2011, 43, 853–869 CrossRef.
- R. Xiao, J. Guo and T. D. Nguyen, RSC Adv., 2015, 5, 416–423 RSC.
- S. Reese and S. Govindjee, Int. J. Solids Struct., 1998, 35, 3455–3482 CrossRef.
- P. Neogi, Diffusion in polymers, CRC Press, 1996, vol. 32 Search PubMed.
- W. M. Huang, H. B. Lu, Y. Zhao, Z. Ding, C. C. Wang, J. L. Zhang, L. Sun, J. Fu and X. Y. Gao, Mater. Des., 2014, 59, 176–192 CrossRef.
- J. Li, J. Shim, J. Deng, J. T. Overvelde, X. Zhu, K. Bertoldi and S. Yang, Soft Matter, 2012, 8, 10322–10328 RSC.
| This journal is © The Royal Society of Chemistry 2016 |