Gas adsorption on MoS2/WS2 in-plane heterojunctions and the I–V response: a first principles study†
Abstract
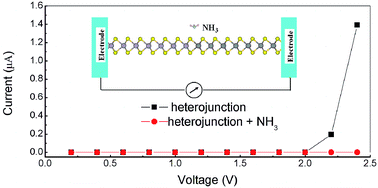
New artificial in-plane heterojunctions based on two-dimensional transition metal dichalcogenides fabricated in recent reports are considered able to offer great scope for applications. Here, we study by first principles calculations the adsorption of CO, H2O, NH3, NO, and NO2 gas molecules on the MoS2/WS2 heterojunction. We have determined the optimal adsorption positions and the adsorption strength, which is driven by charge transfer between the molecules and the heterojunction. Except NH3, which performs as the charge donor, all the other studied molecules act as charge acceptors to the heterojunction. The charge transfer mechanism has been discussed by analyzing the electronic structure of the molecules and the heterojunction. Further calculations show that the molecule adsorption significantly affects the electronic transport properties of the heterojunction. Both the rectification behavior and the value of the passing current can be altered by adsorption, and such sensitivity to adsorption makes the heterojunction a superior gas sensor that promises wide-ranging applications.

 Please wait while we load your content...
Please wait while we load your content...