Extraction and purification of phytol from Abutilon indicum: cytotoxic and apoptotic activity
Parth Thakora,
Japan B. Mehtab,
Ravi R. Patela,
Disha D. Patela,
Ramalingam B. Subramaniana and
Vasudev R. Thakkar*a
aBRD School of Biosciences, Sardar Patel University, Vallabh Vidyanagar, Gujarat, India. E-mail: vasuthakkar@gmail.com; parth7218@gmail.com
bDepartment of Marine and Environmental Sciences, Northeastern University, Boston, USA
First published on 11th May 2016
Abstract
Medicinal plants possess a wide range of secondary metabolites and most of these secondary metabolites have their own importance in the field of medicine. Abutilon indicum is widely used as a medicinal plant in the Indian system of medicine. In the present study, we have evaluated the apoptosis inducing ability of leaf extract of A. indicum. Hydromethanolic leaf extract showed cytotoxicity on Schizosaccharomyces pombe cells by affecting the growth and viability. Phytochemical analysis of the extract revealed the presence of secondary metabolites. A 2D TLC method was used for the separation of the active cytotoxic compound. A HPTLC method was used for the quantification of the bioactive compound. A GC-HRMS method was employed for the identification of the active principle as phytol, which was found to be responsible for inducing ROS mediated apoptosis in S. pombe.
Introduction
Medicinal plants are the reservoir of secondary metabolites. A number of plant derived secondary metabolites have been developed into more effective and less toxic medicines. Screening of plants has revealed many compounds like glycosides, tannins, saponins, steroids, anthraquinones, anthocyanids, alkaloids, flavonoids, terpenoids, phenolics, etc. with pronounced antioxidant, antineoplastic, antiulcer, anti-inflammatory, antimicrobial and immuno-stimulating potential.1 Plants, vegetables and herbs used in folk and traditional medicine have been accepted currently as one of the main sources of cancer prevention drug discovery and development.2 Plant secondary metabolites possess cytotoxic effects and therefore they have been used in the treatment of tumors. Chemoprevention is one of the prominent approaches to treat cancer. In this strategy, synthetic or natural agents (alone or in combination) are used to block the development of cancer in human. Many plants are being studied in contemporary research for chemopreventive drug discovery, from which we have selected Abutilon indicum in the present study.In the literature, A. indicum is ascribed to have a wide range of medicinal applications. A. indicum Linn. (Malvaceae), commonly known as “Atibala” in Sanskrit, is an annual horizontal climbing herb found in plains and wetlands throughout India. Every part of the plant possesses medicinal properties, but leaves are found to have more medicinal properties.3 In folk medicine, A. indicum has been used in treating fever, cough, lung disease, urine output, deafness, ringing in the ears, high fever, mumps, pulmonary tuberculosis, haemorrhoids, diabetes and menorrhoea.4 The juice from the leaves of A. indicum has also been used for quick ulcer healing, relieving thirst, diarrhea, gonorrhoea, inflammation of the bladder, in cleaning wounds and ulcers, treating vaginal infections; and can also be used as an enema.5 The plant contains mucilage tannins, aspergines, gallic acid, sesqueterpenes, flavonoids, saponins, alkaloids and phenolic compounds.5,6 A number of reports have been made on different compounds present in the different plant parts of A. indicum.7–12 Although, A. indicum has been found to possess antioxidant, cytotoxic and anti-inflammatory activities, its active principle is not known and majority of studies are limited to the crude extract of the plant. Several cytotoxic compounds have been identified from the chloroform extract of A. indicum leaves like methyl trans-p-coumarate, methyl caffeate, syringic acid and pinellic acid.13 Among these, methyl caffeate is found to be more cytotoxic compared to others. Another compound phytol has been found from the ethanolic extract of A. indicum leaves, but its activity is not studied.14 S. pombe can be used as a model organism to study the cytotoxic and apoptotic effects of different compounds, and their cellular metabolism.15–17 In the present work, our aim was to find whether cytotoxicity caused on S. pombe by A. indicum is due to apoptosis or some other mechanism and also to find out the active principle responsible for the cytotoxicity.
Results
Effect of crude plant extracts (AP, AL) on the growth of S. pombe
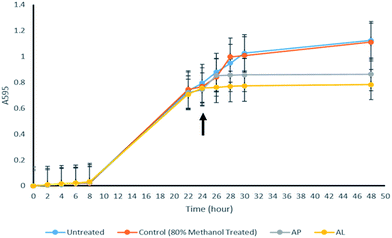
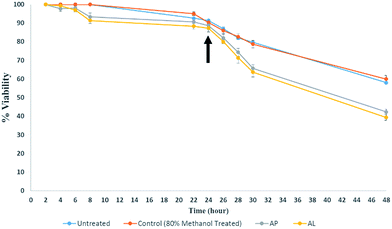
In order to check the cytotoxicity of AP and AL, growth of S. pombe cells in their presence was monitored. Lag phase was around 8 hours in untreated as well as in AP and AL treated S. pombe cells. Log period was about 24 hours in untreated cells, but reduced to 16 hours in AP and AL treated cells (Fig. 1). In AP and AL treated cells, stationary phase reached earlier as compared to untreated cells (Fig. 1). There was no statistically significant difference between untreated and 80% methanol treated samples while there was a significant difference between untreated and AL treated samples.That the growth of S. pombe was reduced due to AP and AL treatment and not due to any other reason, was further verified by analysing the viability of AP and AL treated and untreated S. pombe cells. Fig. 2 shows the decrease in viability due to treatment of AP and AL compared to untreated S. pombe cells. The number of viable cells decreased gradually after the treatment. After four hours of treatment, the percentage viability decreased from 82% to 74% due to AP and to 71% due to AL treatments. After six hours of treatment, viability was reduced by another 13% due to AP and another 15% due to AL treatments. The percentage viability reached to 42% (AP) and 39% (AL) after twenty four hours of treatments. The decrease in the percentage viability was very significant between the untreated and AP treated samples, while there was no significant difference between the untreated and 80% methanol treated samples.
Deviation of growth curve in S. pombe cells treated with AP and AL highlights that both the crude extracts of A. indicum have cytotoxicity to the fission yeast. As AP and AL showed similar cytotoxicity on S. pombe cells (Fig. 2), and as handling and storage of AP was easier compared to AL, AP was qualitatively and quantitatively analysed by chemical extraction and HPTLC.
Phytochemical investigation of plant extract
The powder extract (AP) was subjected to preliminary phytochemical analysis, which revealed the presence of numerous secondary metabolites like tannins, alkaloids, saponins, terpenoids, cardiac glycosides and lignins (active compounds) and absence of starch, flavonoids, anthraquinones, anthocyanides, carbohydrates, amino acids, steroids, phenolic flavonoids and phlobatannins (Table 1).| Sr. No. | Test name | Coloration | AP |
|---|---|---|---|
| a +, ++, +++ represent the degree of intensity of coloration while − represents no change of color i.e. absence of phytochemical groups. | |||
| 1 | Starch: weak iodine solution | Blue black | − |
| 2 | Flavonoids: 10% NaOH | Yellowish brown | − |
| 3 | Tannins: 1 ml of filtrate with 2 ml of ferric chloride | Dark green | +++ |
| 4 | Phenolic flavonoids: 1 ml of filtrate with 2 ml of 10% lead acetate | Brown precipitate | − |
| 5 | Alkaloids: 1 ml of the filtrate with 2 ml of Dragendroff's reagent | Turbid orange colour | +++ |
| 6 | Saponins: 1 ml of filtrate with 2 ml distilled water shaken vigorously and allowed to stand for 10 minutes | Development of foam on the surface of the mixture, lasting for 10 minutes | + |
| 7 | Anthraquinones: to 1 ml of the filtrate; 10 ml of benzene was added, filtered and 5 ml of 10% (v/v) ammonia was added to the filtrate and shaken well | Pinkish coloured solution | − |
| 8 | Anthocyanides: 1 ml of filtrate with 5 ml of dilute HCl | Appearance of pale pink colour | − |
| 9 | Carbohydrates: to 2 ml of Fehling's (A + B) + few drops of sample heated on burner | Brick red or brown precipitates | − |
| 10 | Amino acids: 0.2% ninhydrin reagent | Lemon yellow | − |
| 11 | Steroids: to 1 ml of the filtrate; 10 ml of chloroform was added followed by addition of 10 ml of H2SO4 by the sides of the test tube | Upper layer turned red and sulphuric acid layer showed yellow with green fluorescence | − |
| 12 | Terpenoids: 1 ml of the filtrate with 2 ml CHCl3 and carefully added a few drops of conc. H2SO4 | An interface with a reddish brown coloration | ++ |
| 13 | Cardiac glycosides: to 1 ml of the filtrate; 1 ml of FeCl3 reagent (mixture of 1 vol of 5% FeCl3 solution + 99 vol of glacial acetic acid) was added and followed by the addition of a few drops of conc. H2SO4 | Greenish blue colour appeared within few minutes | + |
| 14 | Phlobatannins: to 1 ml of the filtrate, a few drops of 1% aqueous HCl was added | Red precipitate | − |
| 15 | Lignins: phloroglucinol reagent | Yellowish orange | + |
The crude extracts (AP and AL) were analysed further by TLC. AP showed 12 bands while AL showed 11 bands (data not shown). The phytochemicals present in AP were studied for their effect on S. pombe cells to establish the most active principle responsible for the cytotoxicity of plant extracts.
Screening of cytotoxic compound from crude extract (AP)
For the screening of cytotoxic compounds from crude extract, each of the separated compound in TLC was eluted in 80% methanol and further tested by viability test with appropriate controls. A compound with Rf value 0.68 from AP and another compound from AL with almost same Rf value showed highest cytotoxicity. The effect of both of these compounds on viability of S. pombe cells were found much closer, which means that AP1 and AL1 may be similar compounds isolated by two different methods. Moreover, the crude and purified cytotoxic compound possessed almost similar cytotoxic activity. As the same cytotoxic compound (having a similar Rf value and similar cytotoxicity), was found in AP and AL extracts, for the purification of major cytotoxic compound only AP was used. Since the percent viability showed by AP1 was closely similar to percent viability showed by crude extract (AP), AP1 was considered as the major cytotoxic compound and studied further (Table 2).| Sample names | Untreated | Negative control (80% methanol) | Phytol (2.7 μM) (Sigma) (+ve control) | AL (crude) | AL1 | AP (crude-0.35 μM) | AP1 (0.37 μM purified) | L-Ascorbic acid (10 μM) | AP1 (0.37 μM) + L-ascorbic acid (10 μM) |
|---|---|---|---|---|---|---|---|---|---|
| % viability | 79 | 75 | 37 | 40 | 41 | 42 | 42 | 77 | 58 |
Major cytotoxic compound (AP1) from crude extract (AP)
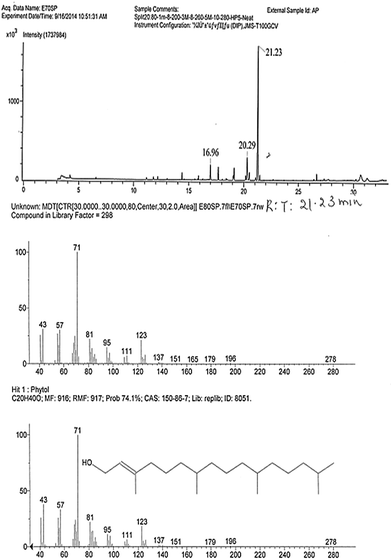
The major cytotoxic compound (AP1, with 0.68 Rf value) was separated and its purity was confirmed by 1D and 2D TLC (data not shown). The single band of purified compound was visualized by spraying vanillin–H2SO4 reagent, which confirmed its purity as well as terpenoid nature. The isolated compound was further subjected to spectral analysis between 190 and 1100 nm with UV visible spectrophotometer (specord200plus, analytical jena). It showed maximum absorbance at a wavelength of 203 nm. The compound was further subjected to gas chromatography/high resolution mass spectrometry (GC-HRMS) analysis and structure prediction. GC-HRMS data clearly showed that the compound has retention time 21.23 min with 68.20% peak area. The split injection mode specified using split ratio 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80. The sharp peak potently showed the involvement of compound with molecular weight 297. The compound showed different fragments of MS at 43, 57, 71, 81, 95, 111, 123, 137, 151, 165, 179, 196, 278 m/z values. The comparison with the National Institute of Standards and Technology (NIST) database showed a similar m/z pattern at 43, 57, 71, 81, 95, 111, 123, 137, 151, 165, 179, 196, 278 values which is a characteristic MS pattern of the compound phytol (Fig. 3).
80. The sharp peak potently showed the involvement of compound with molecular weight 297. The compound showed different fragments of MS at 43, 57, 71, 81, 95, 111, 123, 137, 151, 165, 179, 196, 278 m/z values. The comparison with the National Institute of Standards and Technology (NIST) database showed a similar m/z pattern at 43, 57, 71, 81, 95, 111, 123, 137, 151, 165, 179, 196, 278 values which is a characteristic MS pattern of the compound phytol (Fig. 3).
Phytol was the purified compound from A. indicum leaf extract responsible for cytotoxicity. Phytol isolated from A. indicum and commercial phytol (procured from Sigma) showed almost similar cytotoxicity.
Measurement of concentration of active compound (AP1–phytol) in crude extract (AP)
Phytol present in the AP was found to be 111 μg gram−1 of A. indicum leaves. The quantification of the phytol was carried out in comparison to the standard phytol (Sigma) by HPTLC. The Rf value and peak of a standard phytol matched with AP1 (Fig. 4).Effect of AP1 on ROS levels in S. pombe cells
A. indicum has been reported to possess antioxidant activity.18 We studied the effect of AP and AP1 (0.37 μM) on ferric reducing antioxidant power (FRAP) values (increased Reactive Oxygen Species [ROS]) in S. pombe cells. Untreated S. pombe cells showed 1.27 times higher FRAP value than AP treated cells and 1.87 times higher than AP1 treated S. pombe cells (Fig. 5A). The reduced FRAP value due to AP and AP1 treatment shows reduced anti-oxidation power in S. pombe cells (increased ROS levels).An increased ROS level in treated S. pombe cells was also supported by increased activities of anti-oxidative enzymes, superoxide dismutase [SOD] and catalase. The cells treated with AP as well as AP1 showed 36 and 32 higher units of catalase (U mg−1) than the untreated cells (Fig. 5B). The AP1 treated cells showed 9% higher SOD activity compared with the untreated cells, while crude extract of AP showed 16% higher SOD activity (Fig. 5C). AP1 and AP also increased the lipoxygenase (LOX) activity as compared to untreated cells (Fig. 5D).
Measurement of ROS generation was also carried out by the fluorescence probe. In untreated S. pombe cells, there were very few cells showing green fluorescence (Fig. 6A), while upon exposure to 0.37 μM of the major cytotoxic compound (AP1) in association with the DCFDA probe there were more fluorescent cells (Fig. 6B and C). In presence of antioxidant N-acetyl cysteine (2 mM) the amount of fluorescence decreased (Fig. 6D).
Effect of AP1 on integrity of DNA of S. pombe cells
As increased ROS is known to affect the integrity of macromolecules, therefore the integrity of DNA isolated from AP and AP1 treated S. pombe cells was studied, which showed a smear when separated in 1% agarose gel electrophoresis (Fig. 7). DNA of untreated cells was found to be intact. To confirm whether increased ROS is responsible for affecting the integrity of DNA or not, viability and DNA integrity of AP and AP1 treated S. pombe cells were studied in the presence of antioxidant L-ascorbic acid. The % viability of S. pombe cells was found to increase from 42% to 58% in the presence of L-ascorbic acid (Table 2), this proves that the cytotoxicity of AP and AP1 is due to ROS. The presence of L-ascorbic reduced the cytotoxicity as well as the DNA damage caused by AP and AP1 (Fig. 7).Mechanism of cytotoxicity of AP1
The smear pattern of DNA isolated from AP1 treated (0.37 μM) S. pombe cells led us to study whether AP1 is inducing apoptosis in S. pombe cells or not. Therefore AP1 treated cells were stained with AO–EtBr. AP1 was indeed found to induce apoptosis (Fig. 8B).Early apoptotic cells showed chromatin condensation seen as bright green patches as compared to late apoptotic cells showing orange or red nuclei with fragmented chromatin (Fig. 8C). AO stained live cells green (Fig. 8A), which were found to be 40%, whereas EtBr stained dead cells orange, which were 48% (Table 3). There were 15% of AP1 treated cells found necrotic, with bright uniform orange nuclei and condensed chromatin.
| S. pombe cells | Different phases of programmed cell death | |||
|---|---|---|---|---|
| Early apoptotic | Late apoptotic | Necrotic | Viable cells | |
| Untreated cells | 13% | 16% | 15% | 58% |
| AP1 treated cells (0.37 μM) | 20% | 28% | 15% | 40% |
Moreover, the effect of AP1 was also studied by 4,6-diamidino-2-phenylindole (DAPI) staining. The DAPI bound DNA region showed blue fluorescence. Untreated cells showed densely stained intact DNA (Fig. 9A) while AP1 treated (0.37 μM) cells showed degraded DNA as a smear. Degraded DNA is indicated by arrows (Fig. 9B and C).
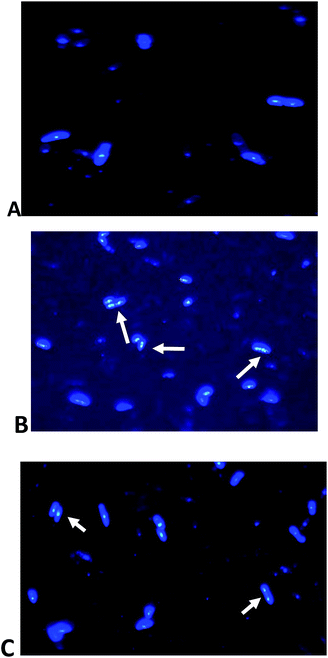 | ||
| Fig. 9 (A) Untreated S. pombe cells stained with DAPI (B) AP1 treated (0.37 μM) S. pombe cells and (C) phytol treated cells (2.7 μM) white arrows indicates the degraded DNA. | ||
Although A. indicum crude extract is reported for their cytotoxic properties,13,19 to the best of the authors' knowledge, this is the first report of apoptosis inducing property of A. indicum leaf extract, and the principle bioactive compound found to be phytol.
Discussion
Medicinally important bioactive compounds are normally accumulated as secondary metabolites in all plant cells, but their concentration varies according to the plant parts, season, climate, and growth phase. A. indicum has been known to contain useful secondary metabolites and a number of researchers have tried to find out the major cytotoxic compound from it.20–22 Leaf is one of the important plant parts preferred for the therapeutic use.3,23 Therefore, in the present study, A. indicum leaves were used for isolation and identification of a potential cytotoxic substance.Generally, shade dried plant powder extract is studied, but we also prepared extract from fresh leaves to negate loss of any compound during drying. Capecka et al. observed the reduction in antioxidant activity in lemon balm and peppermint due to drying, however, the amount of total phenolics remained unchanged. Moreover, they also reported significant loss of ascorbic acid and carotenoids in dried plant material.24 Cold percolation method used in the present study has advantages over the Soxhlet extraction that negates the chances of loss of heat labile components during the extraction.
Ethanolic, chloroform, methanolic, hexane, petroleum ether and water extracts from A. indicum leaves were reported for their antimicrobial activity against Gram positive and Gram negative pathogens like Sarcina luteae, Bacillus cereus, Bacillus megaterium, Staphylococcus aureus, Salmonella typhi, Escherichia coli, Shigella dysenteriae, Vibrio parahemolyticus, Pseudomonas aeruginosa, Klebsiella pneumoniae and Agrobacterium tumefaciens.12,20,25 Ahmad and Khan have reported the antioxidant activity of leaf extract of A. indicum, but the constituent responsible for the antioxidant activity was not found.18 Cytotoxicity of crude extract of A. indicum was reported by Abdul et al. on brine shrimp, however, the principal compound responsible for cytotoxicity was not found.22 Phytol has been reported to reduce the motor activity of worms and cause their death. Phytol has been used to control worm infection in mice.26 In the present work, we used S. pombe cells to study cytotoxicity of leaf extract of A. indicum. S. pombe is an exceptional model system to study genetic and biochemical analyses which helps in the understanding of basic cellular functions. Cell cycle control and many of the cellular mechanisms in fission yeast are similar to those of humans. Therefore information obtained from yeast studies can be extrapolated to human system. This is the main reason for choosing this organism as a model system to study the cytotoxicity of A. indicum extract. Karad et al. also studied the cytotoxicity of polyhydroquinoline and morpholinoquinoline based chemically synthesized compounds on S. pombe cells.27,28
Due to its cytotoxicity and antioxidant activities, A. indicum has been screened for bioactive secondary metabolites by a number of researchers. Alkaloids, flavonoids, steroids, terpenoids and saponins have been isolated and characterized previously from genus Abutilon.7 Similar phytoconstituents were found in the ethanolic and aqueous leaf extracts of A. indicum as reported elsewhere.8–10,20 The major antioxidant compounds reported from A. indicum were phenolics and flavonoids. However, our isolation and screening results showed that phytol, a terpenoid is also the major antioxidant and cytotoxic compound.14,20,21 The presence of phytol was confirmed by comparison of MS pattern of the isolated phytol with the NIST database as well as by comparison with the standard phytol obtained from Sigma. This is the first report confirming phytol as a major cytotoxic compound isolated from A. indicum. Phytol purified from A. indicum at 0.37 μM concentration gave considerable cytotoxic effect (with 42% viability) on S. pombe cells (Table 2).
Phytol is already known as non-mutagenic, common food additive with potential medicinal application. Phytol is widespread in nature. It occurs ubiquitously as a component of chlorophyll. It is well absorbed through the gut of mice (30–66% of the administered dose) and has the acute oral LD50 in rats greater than 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mg kg−1.26 Moreover, its simple structure, easy availability, well-characterised toxicity, cost effectiveness and high tolerance by mammals epitomizes it as a promising candidate for the development of novel and biologically safe drug.29
000 mg kg−1.26 Moreover, its simple structure, easy availability, well-characterised toxicity, cost effectiveness and high tolerance by mammals epitomizes it as a promising candidate for the development of novel and biologically safe drug.29
Phytol has also been extracted from moss and edible marine algae by microwave assisted extraction and high speed counter current chromatography.30,31 The amount of phytol obtained from 15 grams of Undaria pinnatifida was 10.7 mg.31 In the present work, phytol was separated from A. indicum leaf extract and quantified by HPTLC (Fig. 4). We obtained 111 μg phytol from 1 gram of leaves of A. indicum.
AP (crude extract of A. indicum) and AP1 (isolated bioactive compound-phytol) treated S. pombe cells showed reduced ferric reducing antioxidant power (FRAP) values, suggesting an increase in reactive oxygen species (ROS) levels. Increased levels of anti-oxidative enzymes in AP and AP1 treated S. pombe cells (Fig. 5) also provided evidence for increased ROS levels. The increased level of ROS was also confirmed by fluorescence probe DCFDA. Similar observations were made in the study of curcumin on S. pombe cells.32 Various genes are known to encode proteins that generate ROS in cells undergoing apoptosis.33 Patel and Thakkar assessed the ROS generation in S. pombe cells treated with citral and 5-FU using fluorescence inducing probe. S. pombe cells treated with 5-FU showed significantly higher percentage of ROS compared to untreated cells. Increased percentage of ROS values were found with citral treatment.34 ROS are known to be involved in various physiological and pathological processes like inflammation and pain.35 ROS, due to their strong chemical reactivity with biomolecules such as proteins, lipids and DNA can cause harmful alterations such as the destruction of cell membranes by lipid peroxidation and DNA denaturation, leading to changes in protein synthesis and cell duplication.35
In the present study, we found that the increased amount of ROS caused DNA degradation (Fig. 7), suggesting induction of apoptosis. The nuclei fragmentation of apoptotic cells was observed by DAPI staining (Fig. 9B and C). The fragmented chromatin observed by AO/EtBr staining confirmed that cell death due to AP1 treatment was by apoptosis. The reduction in the percentage viability of S. pombe cells treated with AP1 was due to increase in apoptosis (Table 3). The reduction in ROS by antioxidant treatment (Fig. 6D), reduced the cytotoxicity as seen by improvement in viability (Table 2) as well as prevented DNA degradation (Fig. 7). These results clearly confirmed that the cytotoxicity induced by phytol was ROS mediated apoptosis.
Experimental
Plant material
Leaves of Abutilon indicum L. were collected from Vallabh Vidyanagar, Gujarat, India, in July 2014, and identified by Dr A. S. Reddy, Sardar Patel University. A voucher specimen (AIVRT-01) was deposited in the Herbarium of the Department of Botany, Sardar Patel University, Gujarat, India.Extraction
A. indicum leaves were collected, washed under running tap water and dried by blotting. For preparing dried powder leaf extract (AP), washed leaves were dried at 40 °C in hot air oven for about 4–5 days and ground to make fine powder. This powder was passed through a sieve to obtain a homogenous mixture. Hydromethanol (80% methanol + 20% water) was used as a solvent suitable for the extraction of great number of bioactive compounds. 1 gram of powder was taken with 10 ml of 80% methanol and incubated at 20 °C for the cold percolation extraction. Then the hydro-methanolic extract was concentrated (ten times) by evaporating to dryness and resuspended in 80% methanol (1/10th volume of the total filtrate). The concentrated extract was centrifuged at 6000 rpm for 10 minutes to remove sediments and the supernatant collected was labelled as AP and stored at 4 °C. For preparing an extract from fresh leaves (AL) same method was used. Briefly, 1 gram of fresh leaves were extracted in 80% methanol and concentrated 10 times and centrifuged to collect the supernatant, which was labelled as AL and also stored at 4 °C. The percentage yield was measured by the taking weight of powder/leaves before extraction and after extraction.36Percentage yield = (B − A) × 100; B = weight of dried powder/leaves after extraction, A = weight of dried powder/leaves before extraction. Hydromethanolic extract of dried leaves of A. indicum yielded 0.516 gram% (Abutilon Powder – AP), whereas extraction from fresh leaves yielded 0.396 gram% (Abutilon Leaves (fresh) – AL).
Effect of plant crude extracts (AP and AL) on the growth of S. pombe
For preparing inoculum, a single colony of wild type strain of S. pombe, obtained from the Microbial Type Culture Collection (MTCC 191), Chandigarh, India, was grown in 10 ml of YES medium (3% glucose + 0.5% yeast extract + 22.5 mg% of uracil, lysine, leucine, histidine, adenine) and incubated at 30 °C, 130 rpm for 24 hours. One ml of culture was transferred aseptically to four flasks containing 100 ml of YEG media and grown for 24 hours at 30 °C, 130 rpm. After 24 hours, three previously labelled flasks were inoculated with 2 ml of AP, AL and 80% methanol (control) respectively. One flask was not given any treatment to study normal growth. Samples were withdrawn from each flask at every two hours; for growth curve its absorbance was measured at 595 nm and then it was also used for viability studies. Viability was studied from three independent experiments by trypan blue staining and percentage viability was calculated for each sample.37 In this experiment the treatment period was of 24 hours.Phytochemical analysis
AP was analyzed for its qualitative phytochemical constituents using standard procedures.38–40Thin layer chromatography
A TLC plate (EM silica gel 60 F254) of 10 × 10 cm size, pre-ran with methanol, was loaded with 50 μl of AP and AL separately and developed in toluene–ethyl acetate (7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) solvent system. The TLC plate was air dried and visualized under visible, short UV (254 nm) and long UV (366 nm) lights. Rf values of each spot was calculated (Rf = distance travelled by solute/distance travelled by solvent).
3) solvent system. The TLC plate was air dried and visualized under visible, short UV (254 nm) and long UV (366 nm) lights. Rf values of each spot was calculated (Rf = distance travelled by solute/distance travelled by solvent).
Purification and characterization of major cytotoxic compound (AP1)
The cytotoxic components studied were eluted and dissolved in 80% methanol and its purity was checked by 1D and 2D TLC using toluene![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethyl acetate (7
ethyl acetate (7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) as mobile phase in the first dimension and chloroform
3) as mobile phase in the first dimension and chloroform![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) methanol (9
methanol (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) in 2nd dimension. Vanillin–H2SO4 reagent was used as a spray reagent. Major purified cytotoxic compound (AP1) was subjected to spectral analysis at 190–1100 nm in a UV-visible spectrophotometer (Specord 200plus) for its absorbance maxima. GC-HRMS analysis was carried out at SAIF, IIT-Bombay (India). Agilent GC-MS system was used with the split injection mode with ratio 20
1) in 2nd dimension. Vanillin–H2SO4 reagent was used as a spray reagent. Major purified cytotoxic compound (AP1) was subjected to spectral analysis at 190–1100 nm in a UV-visible spectrophotometer (Specord 200plus) for its absorbance maxima. GC-HRMS analysis was carried out at SAIF, IIT-Bombay (India). Agilent GC-MS system was used with the split injection mode with ratio 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80. For better separation, the split injection mode was used. To run the sample HP5 capillary column was used as per the protocol 1m-8-200-3m-8-260-5M-10-280 (1 °C per minute for 8 minutes until the column temperature reached to 200 °C, then 3 °C per minute until the column achieved temperature 260 °C and 5 °C per minute until the column temperature reached to 280 °C).
80. For better separation, the split injection mode was used. To run the sample HP5 capillary column was used as per the protocol 1m-8-200-3m-8-260-5M-10-280 (1 °C per minute for 8 minutes until the column temperature reached to 200 °C, then 3 °C per minute until the column achieved temperature 260 °C and 5 °C per minute until the column temperature reached to 280 °C).
Quantitative determination of AP1 by high performance thin layer chromatography (HPTLC)
Preconditioning of TLC plates was carried out for aluminum-backed silica gel 60 F254 TLC foils (10 × 10 cm) of 0.25 mm thickness (Merck, Darmstadt, Germany) was run with methanol as the mobile phase and the plates were dried in an oven at 120 °C for 20 min. Sample loading onto TLC foils was performed with a Linomat 5 applicator (CAMAG, Muttenz, Germany) using a 100 μl syringe. The operational settings were: band length, 6 mm; application rate, 150 nL s−1; table speed, 10 mm s−1; distance between bands was set automatically, distance from edge of plate to origin on the X-axis was 15 mm and on the Y-axis 8 mm. Toluene![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethylacetate (7
ethylacetate (7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) mobile phase was used for the effective separation. 15 μl of sample (AP1) and 30 μl of sample from crude extract (AP) were loaded onto the plate along with a standard phytol (Sigma, Bangalore, India). After developing in the mobile phase, TLC plates were dried. The purified compound from crude extract was detected in comparison to standard phytol and subjected to spectral scanning for quantification. The peak area of the AP1 band and standard phytol was quantitated by linear scanning at 203 nm using the Camag TLC Scanner 3 with deuterium source. A calibration plot of standard phytol was produced using the winCATS software version 1.2.2. The concentration of AP1 in samples was automatically calculated from the calibration curve by comparing the peak area of standard phytol.
3) mobile phase was used for the effective separation. 15 μl of sample (AP1) and 30 μl of sample from crude extract (AP) were loaded onto the plate along with a standard phytol (Sigma, Bangalore, India). After developing in the mobile phase, TLC plates were dried. The purified compound from crude extract was detected in comparison to standard phytol and subjected to spectral scanning for quantification. The peak area of the AP1 band and standard phytol was quantitated by linear scanning at 203 nm using the Camag TLC Scanner 3 with deuterium source. A calibration plot of standard phytol was produced using the winCATS software version 1.2.2. The concentration of AP1 in samples was automatically calculated from the calibration curve by comparing the peak area of standard phytol.
ROS scavenging activity of AP and AP1
ROS scavenging activity of AP1 was measured by FRAP assay in S. pombe cells.41 For this, 0.37 μM of AP1 was added to the S. pombe cells in exponential phase of growth. Treatment time for the experiment was of 17–18 hour. Catalase, SOD and LOX activities were also measured from the same sample.42–44 All experiments were repeated thrice. The results of multiple experiments were expressed as mean ± standard deviation (SD). Measurement of ROS generation was also carried out using fluorescence probe named 2,7-dichlorofluorescein diacetate (Sigma). Briefly, exponentially grown treated and untreated cells were harvested followed by washing thrice with 1X PBS. 5 μg of fluorescence probe was added in 100 μl suspended cells and incubated for 30 minutes followed by washing with 1X PBS. Cells were placed on a glass slide and covered with 22 mm coverslip and observed under a fluorescence microscope (excitation at 485 nm, emission at 535 nm).45 These experiments were conducted with the 0.37 μM of the purified compound (AP1), standard phytol (2.7 μM) in the presence or absence of the antioxidant N-acetyl cysteine (NAC). Treatment time was of 3 hour.Effect of AP and AP1 on integrity of DNA of S. pombe
DNA isolation of treated and untreated S. pombe cells was carried out by the alkali lysis method along with the mechanical shearing with glass beads described by Patel and Thakkar32 and electrophoresed on 1% agarose gel. Images of DNA in gel were captured in BioRad gel doc system. Treatment time for the experiment was of 17–18 hour.Study of apoptotic traits of major cytotoxic compound (AP1)
Treated and untreated S. pombe cells in the exponential phase of growth were harvested and washed with PBS. Apoptotic traits induced by AP1 were measured by acridine orange/ethidium bromide (AO/EtBr) staining. Briefly, 20 μl of dye mixture (10 μl AO and 10 μl EtBr) was mixed with 100 μl of cell suspension in PBS. After incubation for 10 min at room temperature, 10 μl of the cell–dye mixture was placed on a clean microscope slide and covered with 22 mm coverslip and observed under the fluorescence microscope.34 DAPI staining was performed. Briefly, treated and untreated S. pombe cells in the exponential phase of growth were harvested; washed three times with 1X PBS. Cells were suspended in 100 μl of PBS. 1 μl of cell suspension and 1 μl of DAPI (50 μg ml−1) were mixed; placed on a glass slide and covered with 22 mm coverslip, observed under a fluorescence microscope (excitation at 360 nm, emission at 420 nm).46 These experiments were conducted with the 0.37 μM of the purified compound (AP1), standard phytol (2.7 μM). Treatment time for the experiments was of 17–18 hour.Statistical analyses
Analysis of variance (ANOVA) was carried out using GraphPad Prism 6 software followed by Tukey's multiple comparison test (p < 0.05).Conclusions
AP1 was found to induce chromatin condensation and apoptosis. This is the first report of an apoptosis inducing compound identified from the hydromethanolic extract of A. indicum leaves. Hence the phytol isolated from A. indicum leaves not only decreased the viability (42%), but also showed DNA degradation and different phases of programmed cell death (ROS mediated) in AO–EtBr stained S. pombe cells as well as in DAPI staining. Moreover, based on the study, researchers can translate this compound to in vitro cancer study with the different cell lines. Subject area of the cell lines will spread out the doorways for the researchers to find out the exact mechanism of action of phytol as a one of the potential candidates for the malignant neoplastic disease treatment.Acknowledgements
Authors are thankful to BRD School of Biosciences, Sardar Patel University to provide infrastructure facilities. Authors are thankful to Mr Dilip B. Raval for providing help during the fluorescence microscopy experiments. Mr Parth Thakor is thankful to DST INSPIRE program for the fellowship (DST INSPIRE FELLOWSHIP/2013/44).References
- H. Wagner, Pure Appl. Chem., 1990, 62, 1217 CrossRef CAS.
- M. Gupta, U. Mazumdher and R. Sambath, Acta Pharmacol. Sin., 2004, 25, 1070 CAS.
- M. Razia, B. Rajalakshmi, K. Lavanya, V. Karthiga, W. Bernala and P. Deboral, Int. J. Biol. Pharmaceut. Res., 2013, 4, 256 Search PubMed.
- R. Saraswathi, L. Upadhyay, R. Venkatakrishnan, R. Meera and P. Devi, Int. J. Pharm. Pharm. Sci., 2011, 3, 154 Search PubMed.
- C. Khare, Encyclopedia of Indian medicinal plants, Springer, Berlin Heidelberg New York, 2004 Search PubMed.
- Y. N. Seetharam, G. Chalageri, S. R. Setty and Bheemachar, Fitoterapia, 2002, 73, 156 CrossRef CAS PubMed.
- D. Singh and R. Gupta, Pharmacologyonline, 2008, 1, 253 Search PubMed.
- G. Chakraborthy, Int. J. Pharm. Sci. Drug Res., 2009, 1, 28 Search PubMed.
- S. Sripathi and U. Sankari, Ethnobotanical Leaflets, 2010, 14, 173 Search PubMed.
- R. Ramasubramaniaraja, Asian Journal of Pharmaceutical Analysis, 2011, 1, 88 Search PubMed.
- N. Bhajipale, International Journal Of Ayurvedic And Herbal Medicine, 2012, 2, 598 Search PubMed.
- P. S. Devi, V. Santhi, A. Kannagi and J. J. Shobana, Int. J. Curr. Microbiol. Appl. Sci., 2014, 3, 1069 Search PubMed.
- R. S. Khan, M. Senthi, P. Chandra Rao, A. Basha, M. Alvala, D. Tummuri, H. Masubuti, Y. Fujimoto and S. A. Begum, Nat. Prod. Res., 2015, 29, 1069 CrossRef CAS PubMed.
- K. Shanthi and P. Gowri, Int. J. Curr. Sci., 2013, 7E, 16 Search PubMed.
- H. Willem, W. H. Mager and J. Winderickx, Trends Pharmacol. Sci., 2005, 26, 265 CrossRef PubMed.
- F. Christophe, P. Mathieu, T. Agathe and M. Bernard, Biosci. Rep., 2002, 22, 59 CrossRef.
- M. Shigemi, N. Shahrzad and C. R. John, Curr. Opin. Microbiol., 1999, 2, 618 CrossRef.
- J. Ahmad and I. Khan, J. Plant Pathol. Microbiol., 2012, 3, 3 Search PubMed.
- B. Pejin, V. Kojic and G. Bogdanovic, Nat. Prod. Res., 2014, 28, 2053 CrossRef CAS PubMed.
- R. Sarkar, A. Haque, S. Ranjan and M. Sarker, Pharmacologyonline, 2015, 1, 94 Search PubMed.
- H. Singh, A. Dixit, R. A. Sharma and A. Sharma, Int. J. Pharm. Sci. Rev. Res., 2015, 32, 100 CAS.
- M. M. Abdul, A. A. Sarker, I. M. Saiful and A. Muniruddin, Int. J. Pharm. Phytopharm. Res., 2010, 2, 1 Search PubMed.
- S. Khadabadi and N. Bhajipale, Res. J. Pharm., Biol. Chem. Sci., 2010, 1, 718 Search PubMed.
- E. Capecka, A. Mareczek and M. Leja, Food Chem., 2005, 93, 223 CrossRef CAS.
- A. Sharma, R. A. Sharma and H. Singh, Int. J. Pharm. Sci. Rev. Res., 2013, 20, 120 CAS.
- J. de Moraes, R. N. de Oliveira, J. P. Costa, A. L. G. Junior, D. P. de Sousa, R. M. Freitas, S. M. Allegretti and P. L. S. Pinto, PLoS Neglected Trop. Dis., 2014, 8, e2617 Search PubMed.
- S. C. Karad, V. B. Purohit, D. K. Raval, P. N. Kalaria, J. R. Avalani, P. Thakor and V. R. Thakkar, RSC Adv., 2015, 5, 16000 RSC.
- S. C. Karad, V. B. Purohit, P. Thakor, V. R. Thakkar and D. K. Raval, Eur. J. Med. Chem., 2016, 112, 270 CrossRef CAS PubMed.
- B. Pejin, A. Ciric, J. Glamoclija, M. Nikolic and M. Sokovic, Nat. Prod. Res., 2015, 29, 374 CrossRef CAS PubMed.
- B. Pejin, A. Savic, M. Sokovic, J. Glamoclija, A. Ciric, M. Nikolic, K. Radotic and M. Mojovic, Nat. Prod. Res., 2014, 28, 372 CrossRef CAS PubMed.
- X. Hua Xiao, Z. Quan Yuan and G. Ke Li, Sep. Purif. Technol., 2013, 104, 284 CrossRef.
- P. B. Patel and V. R. Thakkar, Asian J. Cell Biol., 2013, 8, 22 CrossRef CAS.
- K. Polyak, Y. Xia, J. L. Zweier, K. W. Kinzler and B. Vogelstein, Nature, 1997, 389, 300 CrossRef CAS PubMed.
- P. B. Patel and V. R. Thakkar, Indian J. Exp. Biol., 2015, 53, 152 Search PubMed.
- C. C. M. P. Santos, M. S. Salvadori, V. G. Mota, L. M. Costa, A. A. C. de Almeida, G. A. L. de Oliveira, J. P. Costa, D. P. de Sousa, R. M. de Freitas and R. N. de Almeida, Neurosci. J., 2013, 1 CrossRef CAS PubMed.
- S. Mandal, A. Patra, A. Samanta, S. Roy, A. Mandal, T. Mahapatra, S. Pradhan, K. Das and D. Nandi, Asian Pac. J. Trop. Biomed., 2013, 3, 960 CrossRef CAS PubMed.
- J. V. Mehta, S. B. Gajera, P. Thakor, V. R. Thakkar and M. N. Patel, RSC Adv., 2015, 5, 85350 RSC.
- H. Edeoga, D. Okwu and B. Mbabie, Afr. J. Biotechnol., 2005, 4, 685 CrossRef CAS.
- U. Veerachari and A. Bopaiah, Int. J. Pharm. Biol. Sci., 2012, 3, 260 Search PubMed.
- S. Nag, A. Paul and R. Dutta, International Journal of Scientific and Research Publications, 2013, 3, 1 Search PubMed.
- F. Benzie and J. Strain, Methods Enzymol., 1999, 299, 15 Search PubMed.
- K. Shah, R. G. Kumar, S. Verma and R. S. Dubey, Plant Sci., 2001, 161, 1135 CrossRef CAS.
- S. Gao, C. Ouyang, S. Wang, Y. Xu, L. Tang and F. Chen, Plant, Soil Environ., 2008, 54, 374 CAS.
- D. D. Patel, R. R. Patel and V. R. Thakkar, Appl. Biochem. Biotechnol., 2015, 175, 513 CrossRef CAS PubMed.
- M. A. Marchetti, M. Weinberger, Y. Murakami, W. C. Burhans and J. A. Huberman, J. Cell Sci., 2005, 119, 124 CrossRef PubMed.
- The Noguchi Lab Protocols, http://www.eishinoguchi.com.
| This journal is © The Royal Society of Chemistry 2016 |