Application of Fe78Si9B13 amorphous particles in magnetorheological fluids
Abstract
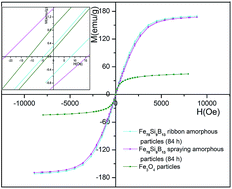
Fe78Si9B13 amorphous particles are applied in the preparation of silicon oil-based magnetorheological (MR) fluids. The structure and morphology of the Fe78Si9B13 amorphous particles were characterized by X-ray diffraction and scanning electron microscopy, respectively. The magnetic properties of the Fe78Si9B13 amorphous particles and the MR fluids prepared with Fe78Si9B13 amorphous particles were characterized by vibrating sample magnetometry. The MR effect of the MR fluids was investigated with a rotational rheometer. The results of VSM show that the saturation magnetization of Fe78Si9B13 amorphous particles is 169.48 emu g−1, accompanying low remanence and low coercivity. The results of rotational rheometry manifest that the MR fluids with Fe78Si9B13 amorphous particles have an improved magnetorheological response compared with magnetic fluids with Fe3O4 nanoparticles under an applied magnetic field.

 Please wait while we load your content...
Please wait while we load your content...