Controllable synthesis of CdS/WO3 heterostructures with enhanced photoelectrochemical performance
Yi Liu*a,
Yingde Cuib,
Feiyan Huanga and
Xinzhe Yanga
aSchool of Chemistry and Chemical Engineering, Guangdong Pharmaceutical University, Guangzhou 510006, China. E-mail: liuyi915@126.com
bGuangzhou Vocational College of Science and Technology, Guangzhou, Guangdong 510550, China
First published on 25th January 2016
Abstract
With the increasing energy and environmental problems, photoelectrochemical (PEC) water splitting has recently attracted a great deal of attention. Herein, we report the facile synthesis of CdS/WO3 heterostructures as high-performance photoanodes for PEC water splitting. CdS/WO3 heterostructures are successfully prepared on F doped SnO2 glass substrates by a two-step process, which involves a hydrothermal growth and electrochemical deposition process. In comparision to pristine WO3 nanostructures, CdS/WO3 heterostructures exhibit substantially high harvesting ability. The CdS/WO3 heterostructures yield a significant photocurrent density of 1.8 mA cm−2 at 0 V vs. Ag/AgCl, which is substantially higher than that of bare WO3 and CdS electrodes. These findings will open up new opportunities to develop other efficient solar-light-driven photocatalytic cells.
Introduction
As the energy crisis and environmental pollution are getting more and more serious, hydrogen has attracted an enormous amount of research attention as a clean energy source because its product of combustion is just water and it does not bring about any greenhouse gases.1–3 Among the various methods to produce hydrogen, one of the most promising and environmentally friendly ways is photoelectrochemical (PEC) water splitting over semiconductors.4–9 Tungsten trioxide (WO3), with a band gap of around 2.6 eV, is of great interest due to its excellent intrinsic stability, environmental compatibility and excellent solar-to-chemical performance under visible light irradiation.10–15 However, some key drawbacks of WO3 photoanodes, such as relatively high recombination of the photo-generated electron–hole pairs and low absorption coefficient of visible light, have severely limited their practical applications.10,16,17 Several strategies have been explored to reduce electron–hole recombination in WO3, including nanostructuring and doping with transition elements.11,12,16,18,19 However, the efficiency is still not satisfactory and it is urgently required to find suitable approaches to enhance the PEC performance of WO3 photoanodes.Sulfides are emerging as attractive visible-light-driven photocatalysts since they have narrow band gaps with suitable conduction bands at relatively high potentials.20–24 Among them, CdS is one of the most attractive II–VI group semiconductors with a direct band gap of 2.4 eV, which has been extensively studied in photocatalytic hydrogen production and the photo-degradation due to its strong ability for harvesting visible light.23,25–29 Considerable efforts have been devoted to enhance the photo–catalytic activity of WO3 by combining it with the CdS, and some achievements have been successfully made.30–33 However, there are few reports about the PEC performance of the CdS/WO3 heterostructures. Thus, it is highly desirable to investigate the properties of CdS/WO3 heterostructures for their PEC water splitting. Herein, we demonstrated the design and synthesis of CdS/WO3 heterostructures on F doped SnO2 (FTO) glass substrates, following by studying their PEC water splitting. The CdS/WO3 heterostructures exhibited a significant photocurrent density of 1.8 mA cm−2 at 0 V vs. Ag/AgCl with good stability. These findings will open up new opportunities to develop other efficient solar-light-driven photocatalytic cells.
Results and discussion
X-ray powder diffraction (XRD) analysis was conducted to identify the crystalline phase of the products. Fig. 1a shows the typical XRD spectra of the WO3 and CdS/WO3 samples. All of the reflections of the XRD pattern for WO3 sample could be indexed to monoclinic WO3 with a lattice constant a = 7.297 Å, b = 7.539 Å and c = 7.688 Å, compatible with JCPDS Card no. 43-1035. The diffraction peaks rose at diffraction angle of 23.1°, 23.6°, 24.4°, 33.3°, 34.2°, 55.6° and 56.0° corresponding to (002), (020), (200), (022), (202), (140) and (420) planes were observed, indicating good crystallinity of the as-prepared WO3 sample. The strong diffraction peaks of SnO2 at 26.7°, 33.9°, 37.9° and 51.7° come from FTO substrate. No other obvious peaks were obtained, demonstrating that high purity of the WO3. The SEM image of a typical WO3 nanostructure is presented in Fig. 1b. Many nanosheets constituted with some small nanoparticles were observed. The width of the nanosheets was around 500 nm. After the electro-deposition at the potential of −1.0 V vs. Ag/AgCl for 300 s at room temperature, the CdS/WO3 heterostructures were prepared. No characteristic peaks corresponding to CdS were observed, which may be due to the amorphous structures and/or the low intensity of the CdS. It is noteworthy that the width of the CdS/WO3 heterostructures is also closed to 500 nm, suggesting that a thin CdS layer was constructed on the surface of WO3 (Fig. 1c and d). | ||
| Fig. 1 (a) XRD patterns of pure WO3 and CdS/WO3 heterostructures. SEM images of: (b) pure WO3, (c and d) CdS/WO3 heterostructures. | ||
In order to better understand the microstructure of CdS/WO3 heterostructures, transmission electron microscopy (TEM) was conducted and displayed in Fig. 2. It shows that the WO3 was covered by a shell of CdS. SAED analysis clearly demonstrate that both the CdS and WO3 have high degree of crystallinity (Fig. 2b). Fig. 2c is the high-resolution TEM (HR-TEM) image taken from the rectangle part of the CdS/WO3 (Fig. 2a). The lattice spacing of 0.37 nm and 0.38 nm match well with the (020) and (002) lattice spacing of monoclinic structure WO3 (JCPDS Card no. 43-1035). In addition, the well-resolved lattice space of 0.24 nm corresponds to the interplanar spacing of (102) plane of hexagonal CdS (JCPDS Card no. 65-3414). Therefore, CdS/WO3 heterostructures are successfully synthesized.
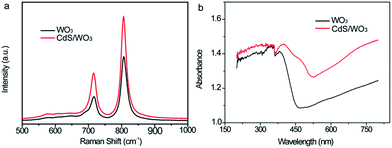
As presented in Fig. 3a, Raman spectra of WO3 sample showed well defined bands at 715 and 808 cm−1 corresponding to the monoclinic phase of tungsten oxide. Both these two characteristic peaks originate from the O–W–O bending modes and stretching modes.34 No obvious peaks can be assigned to CdS. The UV-vis absorption spectra of bare WO3 and CdS/WO3 heterostructures were measured in order to reveal the influence of the CdS. As shown in Fig. 3b, the WO3 have a shape absorption edge around 450 nm, which is consistent with its band gap of 2.6 eV.16 On the other hand, it has a visible light absorption from 450 to 560 nm due to the strong absorption of CdS in the visible region. The absorption edge of the CdS/WO3 heterostructures shifts to 580 nm due to the strong absorption of the CdS in the visible region. Hence, the surface of WO3 nanostructures was successfully coated with a visible-light absorption material of CdS.
X-ray photoelectron spectroscopy (XPS) analysis was also conducted to study the composition of pure WO3 and CdS/WO3 heterostructures, which is presented in Fig. 4. Fig. 4a compares the XPS survey spectra of the pure WO3 and CdS/WO3 heterostructures. In comparison to pure WO3, the XPS survey spectrum of CdS/WO3 heterostructure shows additional peaks of S and Cd, which demonstrates the formation of CdS. Additionally, the atom ratio of Cd, S, W and O in these CdS/WO3 heterostructures is about 4.51%, 3.74%, 13.13% and 37.94%, respectively. The ratio of W to O is closed to the chemical formula of WO3, while the ratio of Cd to S is higher than 1, suggesting that Cd is slightly rich than S in our sample. Fig. 4b displays the W 3d core level XPS spectra. After coating with CdS, the intensity of the W was decreased, indicating the CdS is generated at the surface of the WO3 nanostructures. The Cd 3d core level XPS spectrum of CdS/WO3 sample is showed in Fig. 4c. Two obvious peaks located at 404.9 and 411.7 eV are observed, which are consistent with the reported values for Cd2+.35 As shown in Fig. 4d, the S 2p core level XPS spectrum can be deconvolved to two peaks. The peaks centered at 161.3 and 162.4 eV are attributed to the S 2p3/2 and S 2p2/1 of S2−.36 All these results clearly confirm that the successful preparation of the CdS/WO3 heterostructures, which consists with the XRD and TEM analyses.
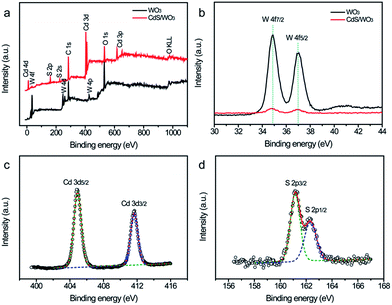 | ||
| Fig. 4 (a) XPS survey spectrum, (b) W 4f, (c) Cd 3d, (c) O 1s, and (d) S 2p core level XPS spectrum of the pure WO3 and CdS/WO3 heterostructures. | ||
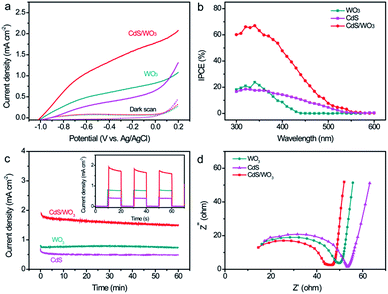
Because the nanostructure can provide high pathway for the electron transport, well-aligned nanostructure with narrow band gap is very promising and efficient for the conversion of solar energy.8 Therefore, the as-prepared CdS/WO3 heterostructures are used as photoanodes in a PEC cell. For better comparison, pristine CdS electrode was prepared by directly using FTO as substrate at the same synthetic condition with CdS/WO3 electrode. The PEC activities of bare WO3, CdS and CdS/WO3 photoelectrodes were firstly characterized by measuring the linear sweep voltammogram (LSW) curves with or without light irradiation. As presented in Fig. 5a, the scans for all the samples collected in the potential range from −1.0 V to 0.2 V vs. Ag/AgCl showed a negligible background current without light irradiation. In contrast, under the light illumination, all the samples showed pronounced photocurrent. It is noteworthy that the CdS/WO3 electrode achieved a remarkable photocurrent density of 1.8 mA cm−2 at the potential of 0 V vs. Ag/AgCl, which is substantially higher than that of bare WO3 (0.8 mA cm−2) and CdS (0.6 mA cm−2) electrodes at the same potential. Such the enhancement of photocurrent for the CdS/WO3 heterostructures can be attributed to the improvement of light-harvesting (Fig. 3b).
The incident photon-to-current conversion efficiency (IPCE) of the samples was measured in the solution of 0.1 M Na2S and 0.1 M Na2SO3 at −0.2 V vs. Ag/AgCl in order to quantitatively investigate the relation between the light absorption and the PEC performance. As shown in Fig. 5b, the maximum IPCE value of the bare WO3 electrode is approximately 23% at the wavelength of 340 nm and then it gradually decrease ranging from 350 to 450 nm and to almost zero at λ > 500 nm last, indicating that the band gap of WO3 is ca. 2.6 eV. As for the bare CdS electrode, its maximum IPCE is about 19% and it showed a higher IPCE value than that of the WO3 electrode at the wavelength of 380–550 nm, revealing the superior photoactivity of CdS under visible light. Significantly, the IPCE value of the CdS/WO3 heterostructures was up to ca. 67%, much higher than the WO3 and CdS samples. It also shows remarkable photo response of 23% at 450 nm and gradually decreases to zero at 550 nm. All these results above fully confirm that the synergistic effect of CdS layer and WO3 nanostructures can significantly enhance the photoelectrochemical activity of the CdS/WO3 heterostructures.
Beside photocurrent, stability is another one of important parameters to evaluate the performance of the photoanode. The current–time (i–t) chronoamperometric response of the WO3, CdS and CdS/WO3 photoanodes at a potential of 0 V vs. Ag/AgCl were collected in Fig. 5c. Obviously, the CdS/WO3 electrode exhibited remarkably higher photocurrent than that of the bare WO3 and CdS electrodes during the long-term test, again showing the outstanding photoelectrochemical performance of CdS/WO3 heterostructures. The bare CdS electrode remained only 69.8% of its initial photocurrent after 60 min under light irradiation. In contrast, the CdS/WO3 electrode showed enhanced stability that can retain more than 80.1% of its initial photocurrent density. This indicates that the construction of heterostructures can improve the stability of CdS. On the other hand, we also collected the temporal photocurrent response of all photoanodes at the potential of 0 V vs. Ag/AgCl. It can be seem from the inset in Fig. 5c that the current of the CdS/WO3 electrode suffered from a relatively sharp decrease at the initial short time and then retained stable. This temporal decrease might be due to the recombination of photo-generated charge carries by the surface (or defect) states of the CdS/WO3 heterostructures.37
Electrochemical impedance spectroscopy (EIS) spectra of the WO3, CdS and CdS/WO3 electrodes under light irradiation were collected to further understand the charge-transfer and separation properties of the hetero-structures, as shown in Fig. 5d. The radius of the arc is correlated with the charge-transfer ability at the corresponding electrode/electrolyte interface. A smaller radius correlated with a lower charge-transfer resistance and an effective separation of photo-generated electron–hole pairs.39 The arc radius of CdS/WO3 heterostructures is obviously reduced compared to those of both the CdS and WO3 under light irradiation, which means an effective interfacial charge transfer and charge separation.
Conclusions
In summary, we have designed and synthesized the CdS/WO3 heterostructures for the PEC water splitting. The images of SEM and TEM indicated that the CdS/WO3 heterostructures were successfully prepared, and the UV-vis absorption spectra showed an obvious red-shift of the absorption edge. Under the light illumination, the CdS/WO3 heterostructures achieved a significant photocurrent density of 1.8 mA cm−2 at the potential 0 V vs. Ag/AgCl, which is much larger than that of the bare WO3 and CdS electrodes at the same potential. The IPCE spectra also show the enhanced photo-response in the visible region. These results suggest that the heterostructures can greatly enhance the photoactivity of the WO3 nanostructures. This achievement makes it possible to design composite photoelectrodes to more effectively utilize the solar spectrum.Experimental
Preparation of WO3 nanostructures
All the reagents used in this study were of analytical grade and were used directly without any purification. The FTO glass was cleaned ultrasonically in ethanol, distilled water, and acetone and finally rinsed in ethanol. The preparation of the WO3 nanostructures can be found in our previous work.38 In general, 1.8 g H2WO4 was dissolved into 35 mL H2O and 5 mL H2O2 (30%) under strong stirring for 4 h. Then, the clear solution was heated at 95 °C with stirring, and further diluted into 40 mL with deionized water. Then, around 4 mL of the precursor was mixed with 16 mL of ethanol and NH4Cl, and transferred to Teflon-lined stainless steel autoclave. A piece of FTO-glass substrate was sealed into the solution and the autoclave maintained at 180 °C for 10 h. After reaction, the autoclave cooled down naturally to room temperature, and the FTO were washed with deionized water and ethanol for 3 times to remove ions that possibly remained at the final products, following by dried at 60 °C for 12 h.Synthesis of the CdS quantum dots (CdS QDs)
Cadmium acetate and thioacetamide were dissolved separately at a concentration of 10 mM in deionized water at room temperature as precursor solutions. CdS QDs was obtained by mixing precursors with the same volume and aging 30 min before electrochemical deposition.Synthesis of the CdS/WO3 heterostructures
The CdS/WO3 heterostructures were prepared by a facile electrochemical deposition. In a typical synthesis route, chronopotentiometry was carried out in a conventional three-electrode cell with a Pt wire as the counter electrode and a saturated calomel electrode (SCE) serving as the reference electrode. The working electrode was the WO3 nanostructures on the FTO substrate. The deposition was carried out by applying the potential of −1.0 V vs. Ag/AgCl for 300 s at room temperature. Pristine CdS/FTO electrode was also prepared by using FTO as substrate at the same synthetic condition.Materials characterizations
The surface morphology, microstructure and the composition of the samples were analyzed by scanning electron microscope (SEM, Quanta 400), transmission electron microscopy (TEM, JEM2010-HR), X-ray diffraction (XRD, D8 ADVANCE) with Cu Kα radiation (λ = 1.5418 Å), and X-ray Photoelectron Spectroscopy (XPS, ESCALab250) and the Raman spectroscopy (Raman, RenishawinVia). The optical properties of the samples were measured with a UV-vis-NIR Spectrophotometer (UV, Shimadzu UV-3150).Photoelectrochemical characterizations
PEC measurements were carried out in a home-made three-electrode cell with a quartz window to facilitate illumination in a mixed solution of 0.1 M Na2S and 0.1 Na2SO3. The working electrode is the as-prepared samples, while a Pt wire and a saturated Ag/AgCl electrode was used as the counter electrode and reference electrode, respectively. All photoelectrochemical experiments were measured by a CHI 760D electrochemical station. The illumination source was a 150 W xenon lamp (Newport 69907) with intensity of about 100 mW cm−2. The incident-photon-to-current-conversion efficiencies (IPCE) were measured with a CHI 760D electrochemical station with a solar simulator (Newport 69911, 300 W xenon lamp) coupled with an aligned monochromator (Oriel Cornerstone 260 1/4 m).Acknowledgements
This work was supported by the Science and technology project of Guangdong Province (2015A090905013), Guangzhou Yuexiu District Science and Technology Projects Funds (2014-WS-029), Science and technology project of self financing in Guangdong Province.Notes and references
- X. Lu, S. Xie, H. Yang, Y. Tong and H. Ji, Chem. Soc. Rev., 2014, 43, 7581–7593 RSC.
- M. Li, X. He, Y. Zeng, M. Chen, Z. Zhang, H. Yang, P. Fang, X. Lu and Y. Tong, Chem. Sci., 2015, 6, 6799–6805 RSC.
- C. Yao, B. Wei, H. Ma, L. Meng, X. Zhang and Q. Gong, J. Power Sources, 2013, 237, 295–299 CrossRef CAS.
- G. Wang, Y. Ling, H. Wang, L. Xihong and Y. Li, J. Photochem. Photobiol., C, 2014, 19, 35–51 CrossRef CAS.
- S. Xie, T. Zhai, W. Li, M. Yu, C. Liang, J. Gan, X. Lu and Y. Tong, Green Chem., 2013, 15, 2434–2440 RSC.
- J.-J. Park, D.-Y. Kim, J.-G. Lee, Y.-H. Cha, M. T. Swihart and S. S. Yoon, RSC Adv., 2014, 4, 8661–8670 RSC.
- G. Wang, H. Wang, Y. Ling, Y. Tang, X. Yang, R. C. Fitzmorris, C. Wang, J. Z. Zhang and Y. Li, Nano Lett., 2011, 11, 3026–3033 CrossRef CAS PubMed.
- S. Xie, M. Li, W. Wei, T. Zhai, P. Fang, R. Qiu, X. Lu and Y. Tong, Nano Energy, 2014, 10, 313–321 CrossRef CAS.
- H. Li, J. Yang, J. Zhang and M. Zhou, RSC Adv., 2012, 2, 6258–6261 RSC.
- S. S. Kalanur, Y. J. Hwang, S. Y. Chae and O. S. Joo, J. Mater. Chem. A, 2013, 1, 3479–3488 CAS.
- C. Santato, M. Odziemkowski, M. Ulmann and J. Augustynski, J. Am. Chem. Soc., 2001, 123, 10639–10649 CrossRef CAS PubMed.
- A. Tanaka, K. Hashimoto and H. Kominami, J. Am. Chem. Soc., 2013, 136, 586–589 CrossRef PubMed.
- G. Hodes, D. Cahen and J. Manassen, Nature, 1976, 260, 312–313 CrossRef CAS.
- J. Su, L. Guo, N. Bao and C. A. Grimes, Nano Lett., 2011, 11, 1928–1933 CrossRef CAS PubMed.
- J. Lin, P. Hu, Y. Zhang, M. Fan, Z. He, C. K. Ngaw, J. S. C. Loo, D. Liao and T. T. Y. Tan, RSC Adv., 2013, 3, 9330–9336 RSC.
- G. Wang, Y. Ling, H. Wang, X. Yang, C. Wang, J. Z. Zhang and Y. Li, Energy Environ. Sci., 2012, 5, 6180–6187 CAS.
- W. Li, J. Li, X. Wang, J. Ma and Q. Chen, Int. J. Hydrogen Energy, 2010, 35, 13137–13145 CrossRef CAS.
- F. Zhan, R. Xie, W. Li, J. Li, Y. Yang, Y. Li and Q. Chen, RSC Adv., 2015, 5, 69753–69760 RSC.
- B. Cole, B. Marsen, E. Miller, Y. Yan, B. To, K. Jones and M. Al-Jassim, J. Phys. Chem. C, 2008, 112, 5213–5220 CAS.
- W. Li, S. Xie, M. Li, X. Ouyang, G. Cui, X. Lu and Y. Tong, J. Mater. Chem. A, 2013, 1, 4190–4193 CAS.
- G. Wang, X. Yang, F. Qian, J. Z. Zhang and Y. Li, Nano Lett., 2010, 10, 1088–1092 CrossRef CAS PubMed.
- S. Yang, X. Wen, W. Zhang and S. Yang, J. Electrochem. Soc., 2005, 152, G220–G226 CrossRef CAS.
- S. Xie, X. Lu, T. Zhai, J. Gan, W. Li, M. Xu, M. Yu, Y.-M. Zhang and Y. Tong, Langmuir, 2012, 28, 10558–10564 CrossRef CAS PubMed.
- H. Li, C. Chen, X. Huang, Y. Leng, M. Hou, X. Xiao, J. Bao, J. You, W. Zhang, Y. Wang, J. Song, Y. Wang, Q. Liu and G. A. Hope, J. Power Sources, 2014, 247, 915–919 CrossRef CAS.
- W. Li, M. Li, S. Xie, T. Zhai, M. Yu, C. Liang, X. Ouyang, X. Lu, H. Li and Y. Tong, CrystEngComm, 2013, 15, 4212–4216 RSC.
- W. Kim, M. Seol, H. Kim, J. B. Miller, A. J. Gellman and K. Yong, J. Mater. Chem. A, 2013, 1, 9587–9589 CAS.
- W.-T. Sun, Y. Yu, H.-Y. Pan, X.-F. Gao, Q. Chen and L.-M. Peng, J. Am. Chem. Soc., 2008, 130, 1124–1125 CrossRef CAS PubMed.
- H. Tada, T. Mitsui, T. Kiyonaga, T. Akita and K. Tanaka, Nat. Mater., 2006, 5, 782–786 CrossRef CAS PubMed.
- H. Li, C. Yao, L. Meng, H. Sun, J. Huang and Q. Gong, Electrochim. Acta, 2013, 108, 45–50 CrossRef CAS.
- Z.-G. Zhao, Z.-F. Liu and M. Miyauchi, Adv. Funct. Mater., 2010, 20, 4162–4167 CrossRef CAS.
- H.-I. Kim, J. Kim, W. Kim and W. Choi, J. Phys. Chem. C, 2011, 115, 9797–9805 CAS.
- H. Kim, Y. Tak, K. Senthil, J. Joo, S. Jeon and K. Yong, J. Vac. Sci. Technol., B: Microelectron. Nanometer Struct.–Process., Meas., Phenom., 2009, 27, 2182–2186 CrossRef CAS.
- H. Li, Y. Zhou, L. Chen, W. Luo, Q. Xu, X. Wang, M. Xiao and Z. Zou, Nanoscale, 2013, 5, 11933–11939 RSC.
- M. Boulova, N. Rosman, P. Bouvier and G. Lucazeau, J. Phys.: Condens. Matter, 2002, 14, 5849 CrossRef CAS.
- C. Wang, L. Sun, H. Yun, J. Li, Y. Lai and C. Lin, Nanotechnology, 2009, 20, 295601 CrossRef PubMed.
- C. Z. Yao, B. H. Wei, H. X. Ma, H. Li, L. X. Meng, X. S. Zhang and Q. J. Gong, J. Power Sources, 2013, 237, 295–299 CrossRef CAS.
- Y. Mao, J. He, X. Sun, W. Li, X. Lu, J. Gan, Z. Liu, L. Gong, J. Chen, P. Liu and Y. Tong, Electrochim. Acta, 2012, 62, 1–7 CrossRef CAS.
- Y. Liu, S. Xie, C. Liu, J. Li, X. Lu and Y. Tong, J. Power Sources, 2014, 269, 98–103 CrossRef CAS.
- H. Li, Y. Zhou, L. Chen, W. Luo, Q. Xu, X. Wang, M. Xiao and Z. Zou, Nanoscale, 2013, 5, 11933–11939 RSC.
| This journal is © The Royal Society of Chemistry 2016 |