Synthesis of CuS nanoplate-containing PDMS film with excellent near-infrared shielding properties†
Wenjun Zhonga,
Nuo Yua,
Lisha Zhang*b,
Zixiao Liua,
Zhaojie Wanga,
Junqing Hua and
Zhigang Chen *a
*a
aState Key Laboratory for Modification of Chemical Fibers and Polymer Materials, College of Materials Science and Engineering, Donghua University, Shanghai 201620, China. E-mail: zgchen@dhu.edu.cn
bCollege of Environmental Science and Engineering, Donghua University, Shanghai 201620, China. E-mail: lszhang@dhu.edu.cn
First published on 5th February 2016
Abstract
The selective shielding of solar light has drawn much attention for the application of innovative energy-saving windows, and a prerequisite for development is obtaining cost-efficient optical materials and coatings which can transmit visible light but cut off near-infrared (NIR) light. In the present work, we have developed low-cost CuS nanoplates as a novel kind of NIR shielding material. CuS nanoplates with a size of 150–250 nm and a thickness of about 30 nm are synthesized by a simple hydrothermal route, and they exhibit weak absorption in the visible region but strong absorption in the NIR region due to their localized surface plasmon resonances. Subsequently, CuS nanoplates are further mixed with polydimethylsiloxane (PDMS) to fabricate CuS/PDMS films. These flexible films retain good transparency in the visible region and strong absorption in the NIR region. For example, a 0.8 mm-thick CuS/PDMS film with 0.10 wt% CuS can transmit 63.0% visible light (400–780 nm) but shield 78.1% NIR light (780–2500 nm). With this film-coated glass as a window of the sealed black box, the interior air temperature of the box goes up from room temperature of 23.0 °C to ∼27.7 °C in 1500 s under the irradiation of strong solar light with an intensity of 0.5 W cm−2, and the temperature elevation (ΔT = 4.7 °C) is much lower compared with that observed with a glass slide (ΔT = 13.7 °C) or ITO glass (ΔT = 9.3 °C) as the window under identical conditions. These facts confirm that the CuS/PDMS film can efficiently prevent the elevation of room temperature, due to its excellent NIR shielding properties. Therefore, CuS nanoplates have great potential as novel NIR shielding materials for the design and development of cost-efficient optical coatings as innovative energy-saving windows in modern buildings and vehicles.
1 Introduction
The selective shielding of solar light has drawn much attention for the objectives of meeting the growing demands of thermo-hygrometric and environmental comfort and of improving the energy efficiency of buildings (or automobiles).1,2 A prerequisite for the development of selective photo-shielding is to obtain optical materials and coatings whose photo-absorption can be well controlled in a wide wavelength range. Two types of optical materials and coatings have been chiefly developed. One is smart glass or switchable glass which changes from translucent (blocking some/all wavelengths of light) to transparent (letting light pass through) when a voltage, light or heat is applied. Typical smart glass includes electrochromic glass,3–5 photochromic glass,1,6,7 and thermochromic glass.2,8,9 The use of smart glass should be the most ideal technology for the selective shielding of solar light but it has a relatively high cost for large-scale practical applications in buildings and automobiles. The other one is non-smart semi-transparent heat-insulation coatings which usually absorb or reflect visible and infrared (IR) light.10–23 The simple and direct use of semi-transparent heat-insulation coatings should be an affordable and inexhaustible technology for the shielding of solar light, which has attracted increasing interest in practical applications. Currently, the widely used semi-transparent heat-insulation coatings should be metal (such as Ag and Ni10–12) films. Metal films on glass can reflect some visible/IR light and thereby reduce the penetration of solar light. However, the preparation of these metal films usually involves a vacuum process (such as vacuum thermal evaporation and magnetron sputtering) which is too costly and would hinder large-scale production of metal films with a large area. Therefore, it is necessary to develop new optical materials and a new preparation technology for semi-transparent heat-insulation coatings.Recently, some nanomaterials have been found to exhibit tuned photo-shielding ability, and the solution-phase process of nanomaterial–polymer films has been demonstrated to be a cost-efficient alternative to conventional metal films. For example, tin-doped indium oxide (ITO)13,14 and antimony-doped tin oxide (ATO)15,16 have been prepared as novel near-infrared (NIR) shielding agents. It should be noted that ITO and ATO can only shield NIR light with a wavelength greater than 1500 nm and indium is an expensive and rare metal (content in the earth’s crust: ∼0.16 ppm). To broaden the photo-shielding range, tungsten (W)-based nanomaterials including W18O49![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 17,18 and MxWO3 (Mx+ = Na+, K+, Rb+, Cs+ and NH4+)19–23 have been developed, and they exhibit part transmittance in the visible region and excellent NIR shielding properties (range: 1000–2700 nm). However, tungsten is also expensive and its content in the earth’s crust is very low (∼1.1 ppm). Therefore, it is still desired to develop low-cost efficient NIR shielding nanomaterials.
17,18 and MxWO3 (Mx+ = Na+, K+, Rb+, Cs+ and NH4+)19–23 have been developed, and they exhibit part transmittance in the visible region and excellent NIR shielding properties (range: 1000–2700 nm). However, tungsten is also expensive and its content in the earth’s crust is very low (∼1.1 ppm). Therefore, it is still desired to develop low-cost efficient NIR shielding nanomaterials.
Previously, we have developed W-based (W18O49![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 24,25 and CsxWO3
24,25 and CsxWO3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 26) and Cu-based (CuS,27 Cu9S5,28 Fe3O4@Cu2−xS,29 and CuS@polymer30) nanomaterials as efficient NIR photothermal agents, all of which exhibit very strong photo-absorption in the NIR region. Furthermore, we find that compared with W-based materials, Cu-based nanomaterials have excellent photostability and the Cu element is very cheap and abundant (content in the earth’s crust: ∼68 ppm). These features trigger our interest in the novel concept of developing Cu-based nanomaterials as a new kind of cost-efficient NIR shielding agent. In the present work, we have designed and prepared CuS nanoplates by a simple hydrothermal method, and CuS nanoplates exhibit high transparency in the visible region (400–780 nm) and strong photo-absorption in the NIR range. With CuS nanoplates and polydimethylsiloxane (PDMS) as the model, flexible CuS–PDMS composite films have been prepared using coating/drying technology. These films can transmit visible light (400–780 nm) but shield efficiently NIR light (780–2500 nm). Importantly, with CuS/PDMS (0.8 mm-thick and 0.10 wt% CuS content) film-coated glass as the window of the sealed box, the elevation of the interior air temperature (ΔT = 4.7 °C) is much lower than that observed with a glass slide (ΔT = 13.7 °C) or ITO glass (ΔT = 9.3 °C) as the window, under the irradiation of simulated solar light with a high intensity of 0.5 W cm−2.
26) and Cu-based (CuS,27 Cu9S5,28 Fe3O4@Cu2−xS,29 and CuS@polymer30) nanomaterials as efficient NIR photothermal agents, all of which exhibit very strong photo-absorption in the NIR region. Furthermore, we find that compared with W-based materials, Cu-based nanomaterials have excellent photostability and the Cu element is very cheap and abundant (content in the earth’s crust: ∼68 ppm). These features trigger our interest in the novel concept of developing Cu-based nanomaterials as a new kind of cost-efficient NIR shielding agent. In the present work, we have designed and prepared CuS nanoplates by a simple hydrothermal method, and CuS nanoplates exhibit high transparency in the visible region (400–780 nm) and strong photo-absorption in the NIR range. With CuS nanoplates and polydimethylsiloxane (PDMS) as the model, flexible CuS–PDMS composite films have been prepared using coating/drying technology. These films can transmit visible light (400–780 nm) but shield efficiently NIR light (780–2500 nm). Importantly, with CuS/PDMS (0.8 mm-thick and 0.10 wt% CuS content) film-coated glass as the window of the sealed box, the elevation of the interior air temperature (ΔT = 4.7 °C) is much lower than that observed with a glass slide (ΔT = 13.7 °C) or ITO glass (ΔT = 9.3 °C) as the window, under the irradiation of simulated solar light with a high intensity of 0.5 W cm−2.
2 Experimental
2.1 Materials
Copper(II) chloride dihydrate (CuCl2·2H2O), sulphide ammonia solution ((NH4)2S, ≥17 wt%), polyvinylpyrrolidone (PVP, K30), hexadecyl trimethyl ammonium bromide (CTAB) and chloroform were purchased from Sinopharm Chemical Reagent Co., Ltd (China) and used without further purification. Polydimethylsiloxane and a curing agent (PDMS, Sylgard 184) were obtained from Dow Corning Corporation.2.2 Synthesis of CuS nanoplates
CuS nanoplates were synthesized by a modified hydrothermal method according to our previous study:27 CuCl2·2H2O (0.085 g, 0.5 mmol) and PVP (2 g) were dissolved in deionized water (30 mL) under magnetic stirring, forming a transparent blue solution. Subsequently, (NH4)2S solution (0.6 mL, ≥17 wt%) was injected rapidly into the above solution, and the resulting solution became black immediately. After being magnetically stirred for 30 min, the resulting suspension was transferred to a 45 mL autoclave, sealed, and hydrothermally treated at 180 °C for 12 h. The system was cooled to room temperature naturally, and the solid sample was separated via centrifugation and washed three times by re-suspending in ethanol and centrifugation, respectively. Finally, part of the precipitate was dried under vacuum at 50 °C for 24 h, and part of the precipitate was re-dispersed in ethanol for further use.2.3 Preparation of CuS/PDMS films
To prepare uniform CuS/PDMS films, CuS nanoplates capped with PVP were transferred to hydrophobic media by CTAB through the ligand wrapped reaction. In a typical process, chloroform solution containing CTAB (10 mL, 36.4 mg mL−1) was added to an ethanol suspension (10 mL) containing CuS nanoplates capped with PVP (4 mg mL−1). The resulting suspension was magnetically stirred at 60 °C and slowly evaporated to dryness. CuS powder was dispersed in chloroform and then was separated via centrifugation to remove the redundant CTAB. At last, the CuS powder was re-dispersed in chloroform to obtain a chloroform suspension with CuS nanoplates (2 mg mL−1).In a typical preparation process of CuS/PDMS films (CuS: 0.1 wt%), PDMS (10 g, 10 mL) and the curing agent (1 g, 1.5 mL) were mixed in a beaker to form a transparent solution (11 g, ∼11.5 mL). Then, a chloroform suspension (5.5 mL) containing CuS nanoplates (2 mg mL−1) was added to the above PDMS-curing agent solution under magnetic stirring, forming a homogeneous dark green colloidal dispersion (slurry). Subsequently, the above slurry with different volumes (2, 3, and 4 mL) was coated on the glass slides (76 × 26 mm2), respectively. Then the glass slides were respectively placed on a hotplate at 80 °C to remove solvent, until the CuS/PDMS films were solidified. The as-prepared CuS/PDMS films with different thicknesses (about 0.6, 0.8, and 1.0 mm) can be easily peeled off from the glass slides. For comparison, 0.8 mm-thick CuS/PDMS films with a different CuS content (0.05 wt% and 0.2 wt%) were also prepared by changing the volume (2.75 or 11 mL) of chloroform suspension containing the CuS nanoplates under identical conditions.
2.4 Characterization and NIR shielding measurements
The morphologies of the CuS sample and CuS/PDMS composite films were investigated using field emission scanning electron microscopy (FESEM, Hitachi S-4800) and high-resolution transmission electron microscopy (HRTEM, JEOL, JEM-2010F). The X-ray diffraction (XRD) pattern of the CuS sample was recorded on a Bruker D4 X-ray diffractometer using Cu Kα radiation (λ = 0.15418 nm). Energy-dispersive X-ray spectroscopy (EDS) of the CuS sample was obtained on a Bruker Quantax 400 EDS system attached to a Hitachi S-4800 field emission scanning electron microscope. The Fourier transform infrared (FTIR) spectrum was measured from the CuS sample in KBr pellets using an IRPrestige-21 spectrometer (Shimadzu). The UV-visible-NIR absorption spectrum of the CuS suspension was measured on a Shimadzu UV-3600 ultraviolet-visible-near-infrared spectrophotometer using quartz cuvettes with an optical path of 1 cm. The thickness of the CuS/PDMS composite films was measured using a vernier caliper.To measure its photothermal effect, CuS powder (∼1 mg) was put on white paper. Under the irradiation of a xenon lamp with the intensity of 0.5 W cm−2, the temperature of the CuS powder was recorded in real time by an infrared thermal imaging camera (FLIR A300). To evaluate the NIR shielding performances, the transmittance and reflectance spectra of the CuS/PDMS films (CuS/PDMS film-coated glass) as well as ITO glass were measured by a UV-visible-NIR spectrophotometer operating from 300 to 2500 nm (Shimadzu UV-3600). To accelerate the aging process, the CuS/PDMS films were irradiated by simulated solar light with a high intensity of 0.5 W cm−2 for 24 h, or were heated in an oven at 100 °C for 48 h, or were immersed in water for 7 days. Their transmittance spectra were also measured by a UV-visible-NIR spectrophotometer.
To further measure the heat-insulation performance, we constructed two sealed boxes, where a facet was covered by a glass slide, commercial ITO glass or CuS/PDMS film-coated glass as the window. An electronic thermometer with an accuracy of ±0.1 °C was inserted into the sealed boxes. A xenon lamp was used as the light source, and its output intensity was independently calibrated using a hand-held optical power meter (Newport model 1918-C, CA, USA). Under the irradiation of the xenon lamp with a light intensity of 0.5 W cm−2, the interior room temperature of the sealed box was recorded in real time by the thermometer. It should be noted that the thermometer should not be directly illuminated by the xenon lamp for keeping accuracy.
3 Results and discussion
3.1 Preparation and characterization of CuS nanoplates
The CuS sample was synthesized by a modified PVP-assisted hydrothermal route at 180 °C for 12 h. The morphology and size of the CuS sample were studied using SEM images (Fig. 1a and b). Obviously, the CuS sample consists of nanoplates with a hexagonal shape, and these nanoplates have a size of 150–250 nm and a thickness of about 30 nm (Fig. 1a and b). In addition, some nanoplates are assembled, interlaced and perpendicular to one another. The microstructure of these hexagonal nanoplates was further investigated using TEM images (Fig. 1c and d). TEM analysis confirms the formation of nanoplates with a size of ∼200 nm (Fig. 1c), which agrees well with the result from the SEM images. The HRTEM image exhibits the well-resolved 2D lattice fringes (Fig. 1d). Two adjacent lattice spacings are determined to be 0.19 nm and 0.33 nm, which can be attributed to the (11![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 0) and (10
0) and (10![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 0) plane spacings with an interfacial angle of 30°. Its corresponding fast Fourier transform (FFT) pattern is also captured (Fig. 1d inset). As visibly illustrated in the FFT pattern, one can find the ordered hexagonal-like spot arrays, again confirming the formation of CuS with a hexagonal shape. All these results strongly signify the single crystallinity nature of the CuS hexagonal nanoplates.
0) plane spacings with an interfacial angle of 30°. Its corresponding fast Fourier transform (FFT) pattern is also captured (Fig. 1d inset). As visibly illustrated in the FFT pattern, one can find the ordered hexagonal-like spot arrays, again confirming the formation of CuS with a hexagonal shape. All these results strongly signify the single crystallinity nature of the CuS hexagonal nanoplates.
 | ||
| Fig. 1 SEM (a and b) and TEM (c and d) images of the CuS nanoplates. The inset in (d) is the corresponding FFT pattern. | ||
The phase and composition of the CuS nanoplates were also investigated using XRD and EDS patterns. The XRD pattern (Fig. 2a) suggests that the CuS nanoplates have high crystallinity with strong and well-defined diffraction peaks. The main diffraction peaks at 27.68°, 29.30°, 31.80°, 32.87°, 47.98°, 52.72° and 59.36° are respectively assigned to the (101), (102), (103), (006), (110), (108) and (116) planes, which are in good consistency with the standard pattern of single hexagonal phase covellite (JCPDS 06-0464). In addition, the EDS pattern confirms that there are Cu and S elements in the CuS sample (Fig. 2b), besides the presence of the Al peak from the aluminum foil substrate during SEM measurement.
Subsequently, a PVP ligand on the surface of the CuS nanoplates was identified using the FTIR spectrum (Fig. 3). Obviously, the CuS sample exhibits a broad band at around 3433 cm−1, which results from the O–H stretching vibration from water molecules. Two bands at 2920 and 2852 cm−1 can be found, which are respectively assigned to the asymmetric (νas) and symmetric (νs) stretching vibrations of methylene (CH2) units inside PVP. The band at 1630 cm−1 corresponds to a strong C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O absorption from the lactam group in the side chains of PVP.27 The bands at 1265 and 1434 cm−1 are attributed to the N → H–O complex and the pyrrolidone ring vibrations, respectively.31 In addition, the broad band near 1065 cm−1 corresponds to a C–O stretching vibration coordinating to metal cations.32 Based on the above results, one can conclude that there are PVP ligands on the surface of the CuS nanoplates.
O absorption from the lactam group in the side chains of PVP.27 The bands at 1265 and 1434 cm−1 are attributed to the N → H–O complex and the pyrrolidone ring vibrations, respectively.31 In addition, the broad band near 1065 cm−1 corresponds to a C–O stretching vibration coordinating to metal cations.32 Based on the above results, one can conclude that there are PVP ligands on the surface of the CuS nanoplates.
Due to the presence of PVP ligands, the CuS nanoplates are hydrophilic and can be easily dispersed in water. The aqueous dispersion containing CuS nanoplates (0.25 mg mL−1) exhibits a strong green-black color (Fig. 4 inset), and has high stability, even remaining unchanged for several weeks. The optical properties of the aqueous dispersion were studied using UV-vis-NIR spectroscopy (Fig. 4). The spectrum is similar to that in our previous study on flower-like CuS superstructures,27 and it exhibits the short wavelength absorption edged at approximately 550 nm, corresponding to the bandgap absorption of covellite. One can find a relatively low absorbance in the visible region (400–780 nm), indicating that part of the visible region can be transmitted through the CuS nanoplate solution. Importantly, the spectrum also shows an increased absorption with the increase of wavelength from 550 nm to 1137 nm, for example, the extinction coefficient is determined to be 8.55 × 103 cm2 g−1 at 1137 nm. The strong NIR photo-absorption is characteristic of CuS nanomaterials, resulting from the localized surface plasmon resonances in vacancy-doped semiconductors.33,34
 | ||
| Fig. 4 UV-vis-NIR absorption spectrum of the aqueous dispersion containing CuS nanoplates (0.25 mg mL−1). Inset: photo of the aqueous dispersion. | ||
As a result of their photo-absorption in the NIR range, the CuS nanoplates can absorb some visible light while they cut off almost all UV and NIR light, probably resulting in the photothermal conversion. To investigate their photothermal effect, CuS nanoplate powder (1 mg) was directly put on white paper (Fig. 5a inset). Under the irradiation of simulated solar light with an intensity of 0.5 W cm−2, the temperature distribution was recorded by a thermal imaging camera (Fig. 5a). Before the light irradiation, both the CuS powder and the white paper have the same temperature of 30.4 °C (Fig. 5a and b), which is the room temperature. During the irradiation process, the maximum temperature of the CuS powder goes up dramatically with increasing the irradiation time to 30 s, and then exhibits a relatively slow increase to 73.6 °C at 180 s (Fig. 5a), as vividly demonstrated in the thermographic image (Fig. 5c). The heating rate becomes slow with the increase of irradiation time, due to the faster heat loss at higher temperatures.27,35 In addition, although part of the white paper was also irradiated simultaneously, the temperature of the paper away from the CuS sample remains the same as the ambient temperature at about 30.4 °C (Fig. 5c). These facts reveal that the CuS nanoplates can rapidly and efficiently absorb NIR light and convert it to heat. To further quantitatively determine the photothermal conversion efficiency, we used a classic solution measurement method developed by Roper et al. in 2007.35 The CuS nanoplates exhibited a photothermal efficiency of 26.4% under the irradiation of a 980 nm laser (Fig. S1 and S2 in the ESI†).
3.2 Synthesis and NIR shielding properties of the CuS/PDMS composite film
It is well known that solar light is chiefly concentrated in the wavelength range between 0.2 and 2.5 μm, including UV light (0.2–0.4 μm, ∼5% of the total energy), visible light (0.4–0.78 μm, ∼45% of the total energy), and NIR light (0.78–4.5 μm, ∼50% of the total energy). To simultaneously satisfy visual effects and reduce the heating effects, it is a good choice to develop a NIR shielding film which can only transmit visible light but cuts off NIR light. The strong NIR photo-absorption of the CuS nanoplates motivated us to investigate their potential in NIR shielding films for buildings or automobiles. For outdoor applications, flexible and hydrophobic polymer films should be better compared with hard and hydrophilic films. In the present study, we used PDMS as the polymer matrix due to its advantages such as non-toxicity, good hydrophobicity and optical transparency. CuS/PDMS composite films were prepared using coating/drying technology, where the CuS/PDMS-curing agent slurries (weight ratio: 5.5–22 mg/11 g) with different volumes (2–4 mL) were respectively coated on the glass slides (76 × 26 mm2), and then the slides were dried at 80 °C to solidify the films (Fig. 6a). These films could be easily peeled off from the glass slides. We obtained three CuS–PDMS composite films with a CuS content of 0.1 wt% and of different thicknesses (about 0.6, 0.8, and 1.0 mm) as well as two 0.8 mm-thick films with a different CuS content (0.05 wt% and 0.2 wt%). All these films are light black in color with good flexibility. | ||
| Fig. 6 (a) Schematic illustration of the preparation process of the flexible CuS/PDMS films. (b and c) Typical photographs of the 0.8 mm-thick CuS/PDMS film with 0.1 wt% CuS. | ||
Since the 0.8 mm-thick CuS/PDMS film with 0.1 wt% CuS has the middle thickness and CuS content, it was used as a model for the subsequent morphology characterization. Obviously, this CuS/PDMS film is light black, and it is also highly transparent (Fig. 6b). In addition, this film is free-standing and can be easily bent at large angles (0–360°) reversibly due to the excellent flexibility of the PDMS matrix (Fig. 6c). Its surface and cross-section morphologies were also investigated using SEM images (Fig. 7). Obviously, the film surface is uniform and smooth, and no cracks can be found (Fig. 7a). In addition, it is clear that the CuS nanoplates are embedded in the PDMS film with good dispersibility (Fig. 7a inset). The cross-sectional SEM image further reveals that the composite film has a typical thickness of ∼0.8 mm (Fig. 7b), which corresponds to the data measured by the vernier caliper. In the middle of the film, a cross-sectional SEM image with higher magnification further confirms that there are many CuS nanoplates embedded in the PDMS film (Fig. 7b inset), which is consistent with the surface morphology.
 | ||
| Fig. 7 Typical surface (a) and cross-section (b) morphologies of the 0.8 mm-thick CuS/PDMS film with 0.1 wt% CuS. | ||
Subsequently, we investigated the optical properties of these CuS/PDMS films using a UV-visible-NIR spectrophotometer. Fig. 8a presents the transmittance spectra of the 0.8 mm-thick CuS/PDMS films with a different CuS content (0.05, 0.10, and 0.20 wt%). For comparison, we also measured the transmittance spectrum of a 0.8 mm-thick pure PDMS film, and this film exhibits high transmittance (≥85%) in the entire UV-vis-NIR region (300–2500 nm), indicating the negligible photo-absorption. Interestingly, when the CuS content is low (0.05 wt%), the 0.05 wt% CuS/PDMS film transmits the majority of visible light in the range of 400–780 nm; for example, the maximum transmittance (Tmax) is about 95.6% at 580 nm. Simultaneously, the 0.05 wt% CuS/PDMS film can shield NIR light in the range of 780–2500 nm (T < 60%) and the minimum transmittance (Tmin) is 29.8% at 1370 nm, while the transmittance goes up with a further increase of the wavelength (1370–2500 nm). With the increase of the CuS content from 0.05 to 0.10 and 0.20 wt%, both films still can transmit the majority of visible light in the range of 400–780 nm, but Tmax at 580 nm reduces to 72.1% and 54.2%, respectively. In addition, the transmittance of both films in the range of 780–2500 nm is very low, where T in the wavelength of 1000–2500 nm is ∼13% for the 0.10 wt% CuS/PDMS film and ∼8% for the 0.20 wt% CuS/PDMS film, indicating the efficient NIR shielding effect. Furthermore, the effects of the film thickness on its transmittance were further investigated using the CuS/PDMS film with 0.10 wt% CuS and different thicknesses (0.6, 0.8, and 1.0 mm) as the model (Fig. 8b). Obviously, with the increase of film thickness from 0.6 to 0.8 and 1.0 mm, the entire transmittance in the visible region (400–780 nm) goes down, where Tmax at 580 nm decreases from 77.7% to 72.1% and 44.1%, respectively. Simultaneously, the entire transmittance in the NIR region (780–2500 nm) also decreases with the increase of the film thickness, where T in the wavelength of 1000–2500 nm declines from ∼24% for the 0.6 mm-thick film to ∼13% for the 0.8 mm-thick film and ∼2.0% for the 1.0 mm-thick film. Strong NIR photo-absorption of these CuS/PDMS films should be attributed to the presence of CuS nanoplates, since CuS nanoplates can absorb efficiently NIR light and then convert it to heat (Fig. 4 and 5), while the PDMS matrix is very transparent in a broad wavelength range (300–2500 nm).36 All these facts indicate that the transmittance and shielding of visible/NIR light can be well adjusted by the CuS content and/or film thickness, for example, a higher CuS content and/or thicker film results in lower transmittance in the NIR region and a higher shielding effect.
It is well known that the ideal shielding film should transmit visible light significantly but cut off NIR light efficiently. We further evaluated the transmitted efficiency (TE) and shielding efficiency (SE) of different light by the CuS/PDMS films, assuming the fact that the CuS/PDMS film is irradiated by standard solar light (AM 1.5, 100 mW cm−2). The transmitted intensity (IT) of different light can be calculated according to the transmittance spectra and standard solar spectrum (Fig. 8) using the following equation:
| IT = ∫F(λ)T(λ)dλ | (1) |
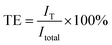 | (2) |
| Type of CuS/PDMS film | Transmitted intensity (mW cm−2) | Transmitted efficiencya (TE, %) | Shielding efficiency (SE, %) | SETS | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| UV | Vis | NIR | UV | Vis | NIR | UV | Vis | NIR | ||
| a The total light intensity from standard solar light: UV (305–400 nm) = 4.11 mW cm−2, vis (400–780 nm) = 46.2 mW cm−2, and NIR (780–2500 nm) = 40.3 mW cm−2. | ||||||||||
| 0.8 mm + 0.05 wt% | 2.42 | 41.6 | 20.8 | 58.9 | 90.1 | 51.6 | 41.1 | 9.87 | 48.4 | 1.39 |
| 0.8 mm + 0.10 wt% | 1.21 | 29.1 | 8.82 | 29.4 | 63.0 | 21.9 | 70.6 | 37.0 | 78.1 | 1.41 |
| 0.8 mm + 0.20 wt% | 0.632 | 20.6 | 4.98 | 15.4 | 44.6 | 12.4 | 84.6 | 55.4 | 87.6 | 1.32 |
| 0.6 mm + 0.10 wt% | 1.63 | 33.0 | 13.9 | 39.6 | 71.4 | 34.5 | 60.4 | 28.6 | 65.5 | 1.37 |
| 1.0 mm + 0.10 wt% | 0.259 | 16.0 | 2.11 | 6.31 | 34.6 | 5.23 | 93.7 | 65.4 | 94.8 | 1.29 |
With the 0.8 mm-thick CuS (0.10 wt%)/PDMS film as a model, we coated the CuS/PDMS film on a glass slide, and then further investigated its transmittance and reflectance properties (Fig. 9). For comparison, the transmittance and reflectance properties from commercial ITO glass were also studied, since ITO has been demonstrated to be a NIR shielding material.13,14 ITO glass exhibits high transmittance in the range of 400–1300 nm (Tmax = 74.1% at 580 nm) and a strong NIR shielding performance in the range of 1300–2500 nm (T < 20%). In addition, ITO glass shows strong light reflectance in the NIR region, indicating that its NIR shielding property results from light reflectance rather than absorption.17 Interestingly, the CuS/PDMS film-coated glass slide can transmit the majority of visible light (Tmax = 71.2% at 580 nm) and shield most of the NIR light (T in the wavelength of 1000–2500 nm is ∼12%), which is similar to the 0.8 mm-thick CuS (0.10 wt%)/PDMS film. Furthermore, it exhibits very low reflectance (∼10%) in the range of 700–2500 nm. These facts indicate that the strong NIR light shielding ability of the CuS/PDMS film should be attributed to large photo-absorption instead of the reflectance in the NIR region.
 | ||
| Fig. 9 Transmittance spectra and reflectance spectra of commercial ITO glass (10 Ω □−1) and the 0.8 mm-thick CuS (0.10 wt%)/PDMS film-coated glass. | ||
The long-term stability of the CuS/PDMS films is very important for their future application in NIR shielding heat-insulation materials. For evaluating their long-term stability under light irradiation, the 0.8 mm-thick CuS (0.10 wt%)/PDMS film was continuously irradiated by simulated solar light with a high intensity of 0.5 W cm−2 for 24 h. Its transmittance spectrum before and after the light irradiation was investigated (Fig. 10a). No significant change can be observed in the transmittance spectrum after the light irradiation compared with that before the treatment, indicating that the light irradiation process has no adverse effect on the optical properties of CuS. Furthermore, we accelerated the aging process by heating the 0.8 mm-thick CuS (0.10 wt%)/PDMS films in an oven at 100 °C for 48 h or immersing them in water for 7 days. Their transmittance spectra before and after the aging process were also investigated, as demonstrated in Fig. 10b. Obviously, both heat and immersion treatments result in a slight decrease in the transmittance spectra. For example, Tmax at 580 nm decreases from 72.1% before the aging process to ∼68% after the aging process, and the transmittance in the NIR region (1300–2500 nm) also decreases from ∼13% to 8–9%. In fact, this slight decrease in the transmittance facilitates a better NIR shielding effect. Therefore, all these facts suggest that the CuS/PDMS films have high long-term stability and water tolerance.
 | ||
| Fig. 10 The transmittance spectra of the CuS/PDMS films: (a) before and after the irradiation of simulated solar light; and (b) before and after water immersion/heat treatment. | ||
As a transparent coating with efficient NIR shielding and high visible transmittance, the CuS/PDMS film should be a promising candidate as a solar filter in the application of energy-saving windows, which may efficiently reduce the heating effect of solar light. To evaluate the practical application of CuS/PDMS films as solar filters, we constructed a simulated experiment which is similar to previous reports,17,18,20 where the sealed black box was covered by a blank glass slide, commercial ITO glass or CuS/PDMS film-coated glass (76 × 26 cm2) as the window, as demonstrated in Fig. 11a. Herein, the 0.8 mm-thick CuS/PDMS film with 0.10 wt% CuS was used as a model, and a xenon lamp was used as the simulated solar light source. Solar light should pass only through the glass window to reach the interior of the sealed black boxes. The room temperature is determined to be 23.0 °C, and the interior air temperature of the box was determined by an electronic thermometer that cannot be directly illuminated by light.
To enlarge the heating effect of solar light, herein we used simulated solar light with a high intensity (0.5 W cm−2) to illuminate the box through the glass window (Fig. 11b). When the glass slide is used as the window of the sealed box, its interior air temperature goes up sharply with the increase of the irradiation time to 300 s, then exhibits a relatively flat section and reaches a maximum of about 36.7 °C at 1500 s, indicating a very high temperature elevation (ΔT1 = 13.7 °C). Subsequently, ITO glass is used as the window, the interior air temperature rises to 30.7 °C in 300 s and achieves a maximum of about 32.3 °C at 1500 s, showing a moderate temperature elevation (ΔT2 = 9.3 °C). Importantly, when CuS/PDMS film-coated glass is used as the window, the interior air temperature goes up slowly and reaches a maximum of about 27.7 °C at 1500 s, suggesting a very low temperature elevation (ΔT3 = 4.7 °C) which is just 34.3% or 50.5% of that with the glass slide (ΔT1 = 13.7 °C) or ITO glass (ΔT2 = 9.3 °C) as the window (Fig. 11b).
The reason for the large difference is analyzed as follows. The high temperature elevation (ΔT1 = 13.7 °C) from the glass slide window should be attributed to the fact that the glass slide has a poor photo-shielding effect, resulting in the high transmittance of solar light and then the strong heating effect. The moderate temperature elevation (ΔT2 = 9.3 °C) from the ITO glass window should be associated with the fact that ITO can only shield NIR light with a wavelength greater than 1300 nm. These strong heating effects will inevitably make humans uncomfortable and then increase the energy consumption due to the use of air-conditioning. On the contrary, the use of CuS/PDMS film-coated glass as the window results in an obviously low temperature elevation (ΔT3 = 4.7 °C), since the CuS/PDMS film can shield efficiently NIR light (Fig. 8 and Table 1). It should be noted that the temperature elevation is inevitable although the CuS/PDMS film-coated glass is used as the window, because the CuS/PDMS film should transmit visible light for satisfying visual effects. But it also can be expected that the temperature elevation can be controlled by adjusting the NIR shielding and visible transmittance effects, for example, by changing the film thickness and CuS content as well as using the combination of different films.
4 Conclusions
In summary, CuS nanoplates have been prepared by a simple hydrothermal route, and they exhibit good visible transmittance and efficient NIR shielding. Then CuS/PDMS composite films have been synthesized, and they retain excellent transparency in the visible region, strong absorption in the NIR region and excellent long-term stability. With the CuS/PDMS film-coated glass as the window of the sealed box, the elevation (ΔT = 4.7 °C) of the interior air temperature is much lower than that observed with a glass slide (ΔT = 13.7 °C) or ITO glass (ΔT = 9.3 °C) as the window, under the irradiation of simulated solar light with a high intensity (0.5 W cm−2). Therefore, the CuS/PDMS films have great potential as semi-transparent heat-insulation coatings in the application of energy-saving windows for modern building and vehicles. More importantly, this work provides some insights into the design of efficient and low-cost semiconductor-based NIR shielding materials for energy-saving windows.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (grant no. 21477019, 51272299, and 51473033), project of the Shanghai Committee of Science and Technology (13JC1400300), the Fundamental Research Funds for the Central Universities, and DHU Distinguished Young Professor Program.Notes and references
- R. Pardo, M. Zayat and D. Levy, Chem. Soc. Rev., 2011, 40, 672–687 RSC.
- A. Seeboth, D. Lötzsch, R. Ruhmann and O. Muehling, Chem. Rev., 2014, 114, 3037–3068 CrossRef CAS PubMed.
- D. R. Rosseinsky and R. J. Mortimer, Adv. Mater., 2001, 13, 783–793 CrossRef CAS.
- S. Cong, Y. Tian, Q. Li, Z. Zhao and F. Geng, Adv. Mater., 2014, 26, 4260–4267 CrossRef CAS PubMed.
- Y. Tian, W. Zhang, S. Cong, Y. Zheng, F. Geng and Z. Zhao, Adv. Funct. Mater., 2015, 25, 5833–5839 CrossRef CAS.
- C. Bechinger, S. Ferrere, A. Zaban, J. Sprague and B. A. Gregg, Nature, 1996, 383, 608–610 CrossRef CAS.
- W. Shi-Chao, P. Mei, L. Kang, W. Sujuan, Z. Jianyong and S. Cheng-Yong, Adv. Mater., 2014, 26, 2072–2077 CrossRef PubMed.
- H. Kim, Y. Kim, K. S. Kim, H. Y. Jeong, A. R. Jang, S. H. Han, D. H. Yoon, K. S. Suh, H. S. Shin, T. Kim and W. S. Yang, ACS Nano, 2013, 7, 5769–5776 CrossRef CAS PubMed.
- P.-C. Guo, T.-Y. Chen, X.-M. Ren, Z. Chu and W. Jin, J. Mater. Chem. A, 2014, 2, 13698–13704 CAS.
- K. Yoshimura, Y. Yamada and M. Okada, Appl. Phys. Lett., 2002, 81, 4709–4711 CrossRef CAS.
- C. L. Tan, S. J. Jang and Y. T. Lee, Opt. Express, 2012, 20, 17448–17455 CrossRef CAS PubMed.
- F. I. Lizama-Tzec, J. D. Macias, M. A. Estrella-Gutierez, A. C. Cahue-Lopez, O. Ares, R. de Coss, J. J. Alvarado-Gil and G. Oskam, J. Mater. Sci.: Mater. Electron., 2015, 26, 5553–5561 CrossRef CAS.
- M. Kanehara, H. Koike, T. Yoshinaga and T. Teranishi, J. Am. Chem. Soc., 2009, 131, 17736–17737 CrossRef CAS PubMed.
- A. Llordes, G. Garcia, J. Gazquez and D. J. Milliron, Nature, 2013, 500, 323–326 CrossRef CAS PubMed.
- J. Qu, J. R. Song, J. Qin, Z. N. Song, W. D. Zhang, Y. X. Shi, T. Zhang, H. Q. Zhang, R. P. Zhang, Z. Y. He and X. Xue, Energy Build., 2014, 77, 1–10 CrossRef.
- Q. Tan, G. Yu, Y. Liao, B. Hu and X. Zhang, Colloid Polym. Sci., 2014, 292, 3233–3241 CAS.
- C. Guo, S. Yin, Y. Huang, Q. Dong and T. Sato, Langmuir, 2011, 27, 12172–12178 CrossRef CAS PubMed.
- C. Guo, S. Yin, M. Yan, M. Kobayashi, M. Kakihana and T. Sato, Inorg. Chem., 2012, 51, 4763–4771 CrossRef CAS PubMed.
- H. Takeda and K. Adachi, J. Am. Ceram. Soc., 2007, 90, 4059–4061 CAS.
- C. Guo, S. Yin, L. Huang and T. Sato, ACS Appl. Mater. Interfaces, 2011, 3, 2794–2799 CAS.
- C. Guo, S. Yin, Q. Dong and T. Sato, CrystEngComm, 2012, 14, 7727–7732 RSC.
- C. Guo, S. Yin, Q. Dong and T. Sato, Nanoscale, 2012, 4, 3394–3398 RSC.
- X. Z. Zeng, Y. J. Zhou, S. D. Ji, H. J. Luo, H. L. Yao, X. Huang and P. Jin, J. Mater. Chem. C, 2015, 3, 8050–8060 RSC.
- Z. Chen, Q. Wang, H. Wang, L. Zhang, G. Song, L. Song, J. Hu, H. Wang, J. Liu and M. Zhu, Adv. Mater., 2013, 25, 2095–2100 CrossRef CAS PubMed.
- W. J. Xu, Q. Y. Tian, Z. G. Chen, M. G. Xia, D. K. Macharia, B. Sun, L. Tian, Y. F. Wang and M. F. Zhu, J. Mater. Chem. B, 2014, 2, 5594–5601 RSC.
- W. J. Xu, Z. Q. Meng, N. Yu, Z. G. Chen, B. Sun, X. Z. Jiang and M. F. Zhu, RSC Adv., 2015, 5, 7074–7082 RSC.
- Q. W. Tian, M. H. Tang, Y. G. Sun, R. J. Zou, Z. G. Chen, M. F. Zhu, S. P. Yang, J. L. Wang, J. H. Wang and J. Q. Hu, Adv. Mater., 2011, 23, 3542–3547 CrossRef CAS PubMed.
- Q. W. Tian, F. R. Jiang, R. J. Zou, Q. Liu, Z. G. Chen, M. F. Zhu, S. P. Yang, J. L. Wang, J. H. Wang and J. Q. Hu, ACS Nano, 2011, 5, 9761–9771 CrossRef CAS PubMed.
- Q. Tian, J. Hu, Y. Zhu, R. Zou, Z. Chen, S. Yang, R. Li, Q. Su, Y. Han and X. Liu, J. Am. Chem. Soc., 2013, 135, 8571–8577 CrossRef CAS PubMed.
- Z. Q. Meng, F. Wei, R. Wang, M. Xia, Z. G. Chen, H. P. Wang and M. F. Zhu, Adv. Mater., 2015, 11, 245–253 Search PubMed.
- J. Y. Chen, D. L. Wang, J. F. Xi, L. Au, A. Siekkinen, A. Warsen, Z. Y. Li, H. Zhang, Y. N. Xia and X. D. Li, Nano Lett., 2007, 7, 1318–1322 CrossRef CAS PubMed.
- M. P. Zheng, M. Y. Gu, Y. P. Jin and G. L. Jin, Mater. Sci. Eng., B, 2000, 77, 55–59 CrossRef.
- Y. Zhao, H. Pan, Y. Lou, X. Qiu, J. Zhu and C. Burda, J. Am. Chem. Soc., 2009, 131, 4253–4261 CrossRef CAS PubMed.
- J. M. Luther, P. K. Jain, T. Ewers and A. P. Alivisatos, Nat. Mater., 2011, 10, 361–366 CrossRef CAS PubMed.
- D. K. Roper, W. Ahn and M. Hoepfner, J. Phys. Chem. C, 2007, 111, 3636–3641 CAS.
- M. Ramuz, B. C. K. Tee, J. B. H. Tok and Z. Bao, Adv. Mater., 2012, 24, 3223–3227 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: The determination of the photothermal conversion efficiency of the CuS nanoplates. See DOI: 10.1039/c5ra22611b |
| This journal is © The Royal Society of Chemistry 2016 |





