Thermally sensitive conductive hydrogel using amphiphilic crosslinker self-assembled carbon nanotube to enhance neurite outgrowth and promote spinal cord regeneration
Abstract
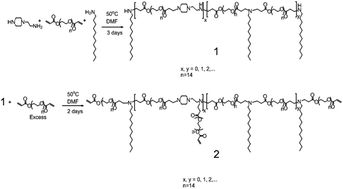
Spinal cord injury leads to severe sensory or motor damage in the human body. Efforts have been made to activate the nerve function by trying physical and biochemical strategies. Carbon nanotubes as conductive materials have been used to transmit electrical signals to improve cell–cell communication and cross-talk, besides providing an extracellular scaffold for neurons. We reported a thermally sensitive hydrogel using copolymerization of n-isopropylacrylamide, the oligomeric amphiphilic crosslinker of polyethylene glycol diacrylate–dodecylamine–1-(2-aminoethyl)piperazine (PEGDA–DD–AEP), and single-walled carbon nanotubes. We hypothesized that carbon nanotubes in the gel could improve neurite outgrowth and nerve regeneration. In order to overcome the aggregation issue of carbon nanotubes, the hydrophobic chains of the amphiphilic crosslinker were used to stabilize the nanotubes. The carbon nanotube–poly(n-isopropylacrylamide) (PNIPAAM) hydrogel was injectable and improved the electrical conductivity. We found that the hydrogel may have potential to promote the growth of SH-SY5Y cells, with significant neurite outgrowth while electrical stimulation was given. In a spinal cord injury model, creating a 1 mm × 1 mm × 1 mm cavity at C7, we found that the hydrogel promoted nerve tissue regeneration and reduced the formation of scar tissue. Therefore, the hydrogel may be a potential repairing biomaterial for neuron network reconstruction and spinal cord regeneration.

 Please wait while we load your content...
Please wait while we load your content...