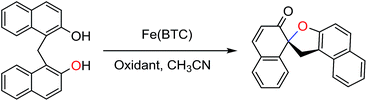
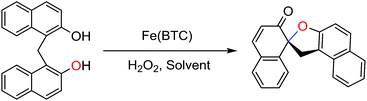
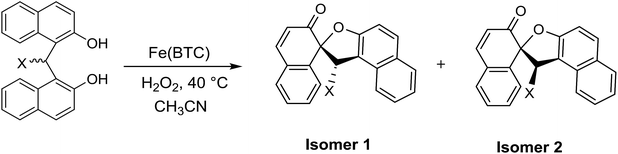
Iron-based metal–organic framework, Fe(BTC): an effective dual-functional catalyst for oxidative cyclization of bisnaphthols and tandem synthesis of quinazolin-4(3H)-ones†
Ali Reza Oveisi,
Ahmad Khorramabadi-zad* and
Saba Daliran
Faculty of Chemistry, Bu-Ali Sina University, P.O. Box: 6517838683, Hamedan, Iran. E-mail: khoram@gmail.com; Fax: +98 (81) 38257407; Tel: +98 9188130018
First published on 16th December 2015
Abstract
An iron-based metal–organic framework [Fe(BTC) (BTC: 1,3,5-benzenetricarboxylate)] has been shown to be an active and heterogeneous catalyst for both oxidative cyclization of methylenebisnaphthols and a modern tandem process (an in situ oxidation-aminal formation-oxidation sequence). Such a potential catalytic utility of Fe(BTC) makes it quite attractive for sustainable industrial chemistry.
Introduction
Calixarenes are synthetic macrocyclic compounds consisting of methylenebisnaphthol or bisphenol skeletons interconnected by methylene bridges.1 Oxidation of calixarenes can afford bis(spirodienone) derivatives, which are attractive intermediates in forming functionalized calixarenes.2 Although several attempts for the oxidation of methylenebisnaphthols to the corresponding spirodienones have been performed in the presence of different reagents such as DDQ,3a phenyltrimethylammonium tribromide (PTMATB) or Br2,3b (diacetoxyiodo)benzene in benzene,3c Ph3Bi,3d chloramine-T or N-chlorosuccinimide,3e N-hydroxyimides3f and magnetic core–shell nanoparticle-supported TEMPO (2,2,6,6-tetramethylpiperidine-oxyl),3g the use of environmentally benign, inexpensive reagents, milder reaction conditions, and decrease of byproducts are highly demanded.Quinazolinones and their derivatives have been investigated as biologically active molecules exhibiting a wide range of pharmacological and biological activities such as antitumor, anticonvulsant, antidiabetic and other anticipated activities.4 Recently, several synthetic methodologies employing different types of catalysts such as I2,5a iridium,5b ruthenium,5c and α-MnO2 (ref. 5d)‡ have been reported for the preparation of quinazolin-4(3H)-ones. These methods have some drawbacks such as the use of toxic and/or expensive catalysts or reaction media, not easily available reagents, additives, tedious procedure, and time-consuming.
In recent years, a new class of porous materials such as metal–organic frameworks (MOFs) and porous organic polymers (POPs) have been deeply attracted due to their potential in applications such as chemical catalysis,6 gas adsorption,7 sensing,8 conductivity9 and light harvesting sensitizer in solar cells.10 Metal–organic frameworks are porous crystalline hybrid materials, which are constructed from organic struts and inorganic vertices (metal ions or clusters).6c,11 Iron-based metal–organic framework Fe(BTC) also known as Basolite F300, produced by BASF, has a semi-amorphous structure, a Brunauer–Emmett–Teller (BET) surface area of 1300–1600 m2 g−1 with the same chemical composition of MIL–100(Fe).12 The latter is comprised of BTC ligands coordinated to iron octahedra [(Fe3-μ3O) sharing a common vertex μ3O]; these iron octahedra contain coordinatively unsaturated metal sites (CUSs) participating in catalytic activities. However, it is reasonable to expect that both Fe(BTC) and MIL(100)–Fe frameworks, have probably the same building units, which can serve to activate substrates for chemical catalysis.12a Iron-based MOF particularly Fe(BTC) have been used for some reactions such as Friedel–Crafts type reactions,12b ring-opening of styrene oxide with methanol and aniline,13a Knoevenagel condensation,13b hydroxyalkylation reaction,13b aerobic oxidation of thiols to disulfides,13c and synthesis of nitriles.13d
Although, many studies have been conducted on catalytic activity of metal–organic frameworks,12a,13,14 only a small number of reports have explored their application in oxidative cyclization and tandem reactions.6d,15
Our interest in oxidative cyclization of bisnaphthols,3d–g quinazolinones6a,16 synthesis and porous materials,6a motivated us to assay further the catalytic potential of metal–organic framework Fe(BTC) for the oxidative cyclization of methylenebisnaphthols to the corresponding spirodienones and a tandem process (an in situ oxidation-aminal formation-oxidation sequence) for the conversion of benzyl alcohols to quinazolin-4(3H)-ones.
Results and discussion
Initial studies were carried out using Fe(BTC) as catalyst for the oxidative cyclization of bis(2-hydroxy-1-naphthyl)methane, as a model reaction, in different oxidation conditions (Table 1). It should be noted that Fe(BTC) was activated at 150 °C for 3 h prior to performing these reactions. The oxidation cyclization reaction did not proceed in the presence and absence of Fe(BTC) and 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO, a stable free nitroxyl radical)17 (Table 1, Entries 1–3). As indicated in Table 1, the best oxidant was proven to be hydrogen peroxide (Entries 4–8). The optimum amount of catalyst was found to be 15 mg (3.75 mg of iron from inductively coupled plasma (ICP) analysis. 0.067 mmol of Fe) (Table 1, Entry 6). Carrying out the reaction at room temperature yielded the desired product in 30% after 12 h (Table 1, Entry 7). We have also investigated the effect of solvent (ethanol, acetonitrile, dichloromethane and ethanol/water) and hence, acetonitrile was chosen as the best one (Table 2), affording the highest yield (82%) of the product with less time-consuming than those of ethanol and ethanol/water ones, probably because it is relatively inert toward oxidation in the course of oxidation reactions. Also it should be noted that acetonitrile is relatively a green solvent as compared with dichloromethane.| Entry | Fe(BTC)b (mg) | Fe (mol%) | Oxidant | Time (h) | Yieldc (%) |
|---|---|---|---|---|---|
| a Reaction conditions: C21H16O2 (0.5 mmol), Fe(BTC) as catalyst, oxidant (1.6 mmol, 3.2 eq.), and acetonitrile (5 ml) at 40 °C.b Fe(BTC) is contain of 25% of Fe.c Total yield (purified by column chromatography).d Reaction was carried out at room temperature.e 1.5 equiv. of TEMPO was used. | |||||
| 1 | 15 | 13 | — | 12 | — |
| 2 | 15 | 13 | TEMPO | 12 | — |
| 3 | — | — | H2O2 | 12 | Trace |
| 4 | 8 | 7 | H2O2 | 8 | 55 |
| 5 | 25 | 22 | H2O2 | 7 | 78 |
| 6 | 15 | 13 | H2O2 | 8 | 82 |
| 7d | 15 | 13 | H2O2 | 12 | 30 |
| 8e | 15 | 13 | H2O2 | 12 | 30 |
| Entry | Solvent | Time (h) | Yield (%) |
|---|---|---|---|
| a Reaction conditions: C21H16O2 (0.5 mmol), Fe(BTC) (15 mg, 0.067 mmol Fe), H2O2 (1.6 mmol), and solvent (5 ml) at 40 °C. | |||
| 1 | EtOH | 10 | 22 |
| 2 | EtOH/H2O (30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 70) 70) |
10 | 31 |
| 3 | CH2Cl2 | 8 | 76 |
| 4 | CH3CN | 8 | 82 |
Having completed the optimization conditions, we were encouraged to examine the oxidative cyclization reaction on a range of bis(2-hydroxy-1-naphthyl)methane derivatives (Table 3). As shown in Table 3, spirodienone derivatives bearing electron-withdrawing and electron-donating substituents were obtained in reasonable yields. Each product was comprised of two sets of diastereomers in nearly equal amounts, assigned by their 1H NMR spectra because of the different anisotropic shielding effect of the μ-phenyl ring on vinylic H-3′ of two diastereomers; 1 and 2.3d,e,g
| Entry | X | Time (h) | Yieldb (%) | M.p. (°C) | Diastereomeric ratioc (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Found | Lit.10–13 | |||||||||
| 1 | 2 | 1 | 2 | 1 | 2 | |||||
| a Reaction conditions: bisnaphthol (0.5 mmol), Fe(BTC) (15 mg, 0.067 mmol Fe), H2O2 (1.6 mmol, 3.2 eq.), and acetonitrile (5 ml) at 40 °C.b Yield refer to the isolated pure products (isomer 1 + isomer 2).c The ratio of the two diastereomers (isomer 1 and isomer 2) is distinguished by 1H NMR. | ||||||||||
| 1 | H | 8 | 82 | 170–171 | 170–171 | — | — | |||
| 2 | C6H5 | 8 | 80 | 209–211 | 264–266 | 210–211 | 263–264 | 75 | 25 | |
| 3 | 4-CH3C6H4 | 8 | 76 | 199–201 | 228–230 | 198–200 | 227–229 | 50 | 50 | |
| 4 | 3-CH3C6H4 | 10 | 81 | 187–189 | 225–227 | 187–188 | 225–227 | 50 | 50 | |
| 5 | 2-CH3OC6H4 | 7 | 78 | 228–231 | 194–196 | 229–230 | 194–196 | 55 | 45 | |
| 6 | 2,4-Cl2C6H3 | 9 | 85 | 208–209 | 195–197 | 204–206 | 195–197 | 55 | 45 | |
| 7 | 4-FC6H4 | 10 | 80 | 214–216 | 145–146 | 214–215 | 145–146 | 50 | 50 | |
| 8 | 2-BrC6H4 | 8 | 78 | 219–221 | 147–149 | 218–223 | 146–148 | 55 | 45 | |
A test for reusability of Fe(BTC) catalyst was then performed using oxidative cyclization of bis(2-hydroxy-1-naphthyl)methane, as a model reaction. After each run, the catalyst was removed by centrifugation and washed with acetonitrile, ethyl acetate, and then acetone followed by drying at 150 °C. Fe(BTC) was reused for three cycles with a little loss in its efficiency (a yield of 76% and 68% of product was obtained after the second and third cycles, respectively), due to some blocked sites by chemicals or partially damage of Fe(BTC) network (Table S1†). Atomic absorption spectroscopy (AAS) analysis of supernatant, at the end of reaction, showed 0.3 ppm Fe leaching out which is less than 0.01% of the initial Fe present in the catalyst (0.13 and 0.17 ppm for the second and the third cycles, respectively). Hence, hot-filtration based leaching test was carried out to understand the nature of active species of catalyst in the reaction. Therefore, Fe(BTC) catalyst was removed from the reaction mixture after 4 h, while the supernatant ran further under the same conditions. There was almost no further yield of spirodienone, even after 8 h in the absence of catalyst (see Table S1 in (ESI)†). Thus, the results confirmed heterogeneity of the participated catalyst.
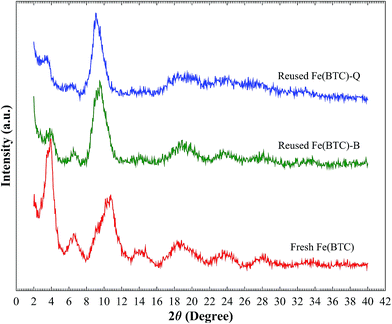
Powder X-ray diffraction (PXRD) patterns of the Fe(BTC) exhibit semi-amorphous nature of the matter, resulting in relatively broad or overlapping peaks because of the small size of crystals and its disordered structure (Fig. 1); its characterization by XRD being therefore limited, and no crystal structural data of Fe(BTC) have yet been reported.
To get further information, we compare the PXRD patterns of fresh Fe(BTC) with the simulated crystalline MIL–100(Fe) one (Fig. S1 & S2†). As illustrated in Fig. 1 and reported in literature,12a,b,18 Fe(BTC) shows a broad diffraction peak at low angle, 2θ ∼ 3.6° with d spacing of 21.9 Å. The value is in agreement with the 220, 311 and 222 reflections observed in MIL–100(Fe), suggesting the presence of some mesoporous cages in the Fe(BTC) [strong peaks at low angle (2θ = 2–4°)19 XRD patterns manifest the presence of mesoporous structure], consistent with pore diameter 21 Å obtained from nitrogen adsorption measurements.12a,13 Moreover, no diffraction peak appeared in PXRD pattern of Fe(BTC) with d spacing of 42.3 Å, 2θ ∼2.0°, corresponding to the 111 reflection, characteristic of the largest mesoporous cages (the largest d spacing of the structure, pore diameter of 29 Å) in MIL–100(Fe). However, the XRD patterns indicate that Fe(BTC) contain just the smallest mesoporous cages established in the MIL–100(Fe). Also, a broad peak (2θ ∼10.6°) shown in the PXRD patterns of Fe(BTC) correlates with the 822, 840 and 911 reflections of observed in MIL–100(Fe).
Comparison of the PXRD of the fresh and the reused catalyst indicates some decrease in the peak intensity of the reused Fe(BTC), suggesting an increase of disordering in the structure or a change in the mesopores content (pore-filling effect) (as shown in Fig. 1, fresh and reused Fe(BTC)-B). Also Fig. 1 shows a slight shift of the main peak positions of the XRD, after reusing, to lower 2θ values (larger d-spacing), suggesting that there is an expansion of the framework in response to the chemicals. It might be attributed to the strain effect of the framework upon strong coordination of reactants or products within the MOF during the reaction. This is consistent with breathing effect in MOFs.20
As scanning electron microscopy (SEM) images show (Fig. 2), the morphology of the Fe(BTC) was retained under the reaction conditions. SEM images of Fe(BTC) showed agglomerates of spheroid particles, with particle-size ranging from 45 to 105 nm.
A comparison of the FT-IR (see Fig. S3 & S4†) of the fresh and reused (the first and the last cycles) catalyst indicated a slightly higher relative intensity of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O band about 1710 cm−1 for the latter, which may be attributed to small amount of spirodienones or impurity along with the free carboxylic groups, in agreement with the observed changes in powder XRD patterns.
O band about 1710 cm−1 for the latter, which may be attributed to small amount of spirodienones or impurity along with the free carboxylic groups, in agreement with the observed changes in powder XRD patterns.
When TEMPO was used as a radical scavenger combined with Fe(BTC) and H2O2, significant decrease was observed in yield of spirodienone (Table 1, Entry 8). This reveals that the reaction proceeds through the formation of radical species.12c,21 Based on literature survey3g,12a,c and this observation, we propose a mechanism for the oxidative cyclization of methylenebisnaphthols as shown in Scheme 1. At first, hydrogen peroxide molecules coordinate to those Fe(III) sites which are not saturated with the framework carboxylate groups, to give Fe(III)–OOH complexes (I). Then the homolytic cleavage of the peroxidic O–O bond in intermediate (I) produces the active species (HO–O˙, II) followed by abstracting a hydrogen-atom from methylenbisnaphthol to afford radical intermediate (III). Abstraction of another hydrogen atom from (III) results in the formation of a diradical intermediate (IV), which proceeds to give the product. Subsequently, Fe(II) is re-oxidized to Fe(III) active sites via an electron transfer to hydrogen peroxide.
 | ||
| Scheme 1 Proposed mechanism for the oxidative cyclization of bisnaphthols in the presence of Fe(BTC). | ||
We next examined the potential catalytic activity of this metal–organic framework for a modern tandem process: quinazolin-4(3H)-ones synthesis by the reaction of benzyl alcohols and o-aminobenzamide in the presence of oxidant (Tables 4 and 5). Owing to optimization conditions, at the outset, benzyl alcohol reacted with o-aminobenzamide as a model reaction, which afforded 2-phenylquinazolin-4(3H)-one (Table 4). As indicated in Table 4, the use of TBHP (tert-butyl hydroperoxide), as an oxidant, gave a higher yield with respect to hydrogen peroxide (Table 4, Entries 1 and 2). It might be due to instability of H2O2 compared with that of TBHP in catalytic reaction conditions. The best results were obtained with DMSO (dimethyl sulfoxide) as solvent (Table 4, Entries 5–8), affording the highest yield (81%) of the product as compared with the other solvents. It should be noted that DMSO could act as a mild oxidant, in addition its solvent role. The amount of Fe(BTC) was optimized to be 15 mg (0.067 mmol Fe) in order to reduce the consumed catalyst and to increase the product yield (Table 4, Entry 5). The effect of temperature on this reaction was also investigated, which showed that the reaction should be performed at 60 °C to get a high yield (Table 4, Entry 5). As indicated in Table 4, the reaction did not proceed by DMSO without TBHP (Table 4, Entry 9).
| Entry | Fe(BTC)b (mg) | Fe (mol%) | Oxidant | T (°C) | Solvent | Time (h) | Yieldc (%) |
|---|---|---|---|---|---|---|---|
| a Reaction conditions: benzyl alcohol (1.5 mmol), o-aminobenzamide (0.5 mmol), oxidant (2 mmol), solvent (3–5 ml), Fe(BTC) (10–22 mg).b 15 mg Fe(BTC) is equal to 3.75 mg Fe (0.067 mmol Fe).c Yield refer to the isolated pure products. | |||||||
| 1 | 15 | 13 | H2O2 | 40 | CH3CN | 14 | 21 |
| 2 | 15 | 13 | TBHP | 40 | CH3CN | 14 | 39 |
| 3 | 15 | 13 | TBHP | 60 | CH3CN | 14 | 47 |
| 4 | 15 | 13 | TBHP | 60 | Xylene | 14 | 30 |
| 5 | 15 | 13 | TBHP | 60 | DMSO | 14 | 81 |
| 6 | 15 | 13 | TBHP | 85 | DMSO | 14 | 78 |
| 7 | 10 | 9 | TBHP | 60 | DMSO | 14 | 75 |
| 8 | 22 | 20 | TBHP | 60 | DMSO | 14 | 82 |
| 9 | 15 | 13 | — | 60 | DMSO | 14 | — |
In the next step, the optimal condition for the preparation of quinazolin-4(3H)-one derivatives was evaluated. The results showed good versatility of this method for neutral, electron rich and electron deficient groups such as H, Me, OMe, Cl, and NO2 (Table 5). An additional test was also carried out to examine the reusability of the catalyst, using a model reaction of benzyl alcohol with o-aminobenzamide. The Fe(BTC) was found to be an efficient and reusable catalyst, with a slight decrease in its activity (Table 5, Entry 1). AAS analysis of supernatant, at the end of reaction, showed 0.7 ppm Fe leaching out for the reaction, attributing to 0.02% of the initial Fe present in the catalyst. Some decrease in the peak intensity of PXRD pattern of the reused Fe(BTC) was observed as compared with that of fresh Fe(BTC) one (see Fig. 1), suggesting an increase of disordering in the structure or a change in the mesopores content (pore-filling effect).
According to literature survey, the catalyst sites are the weak Brønsted acid sites (demonstrated by IR pyridine adsorption,12a,22 which may be attributed to unknown impurities or uncoordinated BTC carboxylic groups), the redox-active12c and Lewis acid13a iron sites. So, this catalyst can act usefully both as acid and redox active sites platform.
Based on literature survey and the above argument, we introduce a mechanism for the tandem process as follows (Scheme 2). At first, the coordination of t-BuOOH to the Fe(III) sites afford t-Bu-OO-Fe(III) species (A) which subsequently produces the active species (t-Bu-O-O˙, II). Then, abstraction of a hydrogen-atom from benzyl alcohol gives a radical intermediate (C). As the proposed mechanism shows, three the pathways: (1), (2) and (3) give the aldehyde product (E). The pathway (1) passes through the carbocation formation with the concomitant reduction of Fe(III) to Fe(II). The pathway (2) proceeds via a gem-diol-like structure formation, and dehydration. The other pathway involves a hydrogen-atom abstraction from (C) by the t-BuOO˙ and t-BuO˙ radicals. After that, activated aldehyde reacts with o-aminobenzamide to give (G), which in turn, afford aminal intermediate. Finally, oxidation of aminal intermediate results in quinazolin-4(3H)-one formation followed by oxidation of Fe(II) to Fe(III) accomplishes with an electron transfer to t-BuOOH.
 | ||
| Scheme 2 Proposed mechanism for the quinazoline-4(3H)-ones from benzyl alcohols in the presence of Fe(BTC). | ||
Conclusion
We have further developed the application of porous iron-based MOF as a heterogeneous and catalytically active species for the oxidative cyclization of methylenebisnaphthols, leading to the corresponding two diastereomers in nearly equal amounts (isomers 1 and 2), as well as a modern tandem process (an in situ oxidation-aminal formation-oxidation sequence) for the conversion of benzyl alcohols to quinazolin-4(3H)-ones. The catalyst can usefully act as both acid and redox active sites platform. This work consistently has the advantages such as availability of MOF, inexpensive catalyst, mild reaction conditions, reasonable yields, and simple experimental procedures.Acknowledgements
The authors gratefully acknowledge the financial support for this work from the Bu-Ali Sina University, Hamedan, Iran.Notes and references
- (a) P. E. Georghiou, M. Ashram, H. J. Clase and J. N. Bridson, J. Org. Chem., 1998, 63, 1819–1826 CrossRef CAS; (b) S. Chowdhury and P. E. Georghiou, J. Org. Chem., 2002, 67, 6808–6811 CrossRef CAS PubMed; (c) F. Davis and S. Higson, in Macrocycles: Construction, Chemistry and Nanotechnology Applications, John Wiley & Sons, Ltd, Chichester, UK, 2011, ch. 5, pp. 126–189 Search PubMed.
- (a) S. E. Biali, Synlett, 2003, 0001–0011 CrossRef CAS; (b) R. L. Varma, V. B. Ganga and E. Suresh, J. Org. Chem., 2007, 72, 1017–1019 CrossRef CAS PubMed; (c) S. B. Nimse and T. Kim, Chem. Soc. Rev., 2013, 42, 366–386 RSC.
- (a) T. R. Kasturi, B. Rajasekhar, G. J. Raju, G. M. Reddy, R. Sivaramakrishnan, N. Ramasubbu and K. Venkatesan, J. Chem. Soc., Perkin Trans. 1, 1984, 2375–2382 RSC; (b) P. E. Georghiou, M. Ashram, H. J. Clase and J. N. Bridson, J. Org. Chem., 1998, 63, 1819–1826 CrossRef CAS; (c) D. J. Bennett, F. M. Dean, G. A. Herbin, D. A. Matkin, A. W. Price and M. L. Robinson, J. Chem. Soc., Perkin Trans. 1, 1980, 1978–1985 RSC; (d) A. Khoramabadi-zad, I. Yavari, A. Shiri and A. Bani, J. Heterocycl. Chem., 2008, 45, 1351–1358 CrossRef CAS; (e) A. Khoramabadi-zad, A. Shiri, F. Derakhshan-Panah and Z. Salimi, Mol. Diversity, 2010, 14, 829–832 CrossRef CAS PubMed; (f) A. Khoramabadi-zad, S. Mohammadi and M. Azadmanesh, Z. Naturforsch., B: J. Chem. Sci., 2014, 69, 444–450 Search PubMed; (g) A. Khorramabadi-zad, S. Daliran and A. R. Oveisi, C. R. Chim., 2013, 16, 972–976 CrossRef CAS.
- (a) S. Eguchi, in Bioactive heterocycles I, ed. S. Eguchi, Springer-Verlag, Berlin-Heidelberg, Germany, 2006, vol. 6, ch. 2, pp. 113–156 Search PubMed; (b) V. Sridharan, P. A. Suryavanshi and J. C. Menéndez, Chem. Rev., 2011, 111, 7157–7259 CrossRef CAS PubMed.
- (a) W. Ge, X. Zhu and Y. Wei, RSC Adv., 2013, 3, 10817–10822 RSC; (b) J. Zhou and J. Fang, J. Org. Chem., 2011, 76, 7730–7736 CrossRef CAS PubMed; (c) A. J. A. Watson, A. C. Maxwell and J. M. J. Williams, Org. Biomol. Chem., 2012, 10, 240–243 RSC; (d) Z. Zhang, M. Wang, C. Zhang, Z. Zhang, J. Lu and F. Wang, Chem. Commun., 2015, 51, 9205–9207 RSC.
- (a) A. R. Oveisi, K. Zhang, A. Khorramabadi-zad, O. K. Farha and J. T. Hupp, Sci. Rep., 2015, 5, 10621–10628 CrossRef CAS PubMed; (b) J. Lee, O. K. Farha, J. Roberts, K. A. Scheidt, S. T. Nguyen and J. T. Hupp, Chem. Soc. Rev., 2009, 38, 1450–1459 RSC; (c) A. Dhakshinamoorthy and H. Garcia, Chem. Soc. Rev., 2014, 43, 5750–5765 RSC; (d) M. H. Beyzavi, N. A. Vermeulen, A. J. Howarth, S. Tussupbayev, A. B. League, N. M. Schweitzer, J. R. Gallagher, A. E. Platero-Prats, N. Hafezi, A. A. Sarjeant, J. T. Miller, K. W. Chapman, J. F. Stoddart, C. J. Cramer, J. T. Hupp and O. K. Farha, J. Am. Chem. Soc., 2015, 137, 13624–13631 CrossRef CAS PubMed; (e) P. Kaur, J. T. Hupp and S. T. Nguyen, ACS Catal., 2011, 1, 819–835 CrossRef CAS.
- (a) A. M. Fracaroli, H. Furukawa, M. Suzuki, M. Dodd, S. Okajima, F. Gándara, J. A. Reimer and O. M. Yaghi, J. Am. Chem. Soc., 2014, 136, 8863–8866 CrossRef CAS PubMed; (b) X. Zhang, Y.-Z. Zhang, D.-S. Zhang, B. Zhu and J.-R. Li, Dalton Trans., 2015, 44, 15697–15702 RSC; (c) N. Nijem, H. Bluhm, M. L. Ng, M. Kunz, S. R. Leone and M. K. Gilles, Chem. Commun., 2014, 50, 10144–10147 RSC; (d) J. A. Mason, M. Veenstra and J. R. Long, Chem. Sci., 2014, 5, 32–51 RSC.
- (a) X. Zhu, H. Zheng, X. Wei, Z. Lin, L. Guo, B. Qiu and G. Chen, Chem. Commun., 2013, 49, 1276–1278 RSC; (b) Z. Hu, B. J. Deibert and J. Li, Chem. Soc. Rev., 2014, 43, 5815–5840 RSC.
- (a) Y. Shen, X.-F. Yang, H.-B. Zhu, Y. Zhao and W.-S. Li, Dalton Trans., 2015, 44, 14741–14746 RSC; (b) P. Ramaswamy, N. E. Wong and G. K. H. Shimizu, Chem. Soc. Rev., 2014, 43, 5913–5932 RSC.
- (a) D. Y. Lee, E.-K. Kim, C. Y. Shin, D. V. Shinde, W. Lee, N. K. Shrestha, J. K. Lee and S.-H. Han, RSC Adv., 2014, 4, 12037–12042 RSC; (b) D. Y. Lee, I. Lim, C. Y. Shin, S. A. Patil, W. Lee, N. K. Shrestha, J. K. Lee and S.-H. Han, J. Mater. Chem. A, 2015, 3, 22669–22676 RSC; (c) H.-J. Son, S. Jin, S. Patwardhan, S. J. Wezenberg, N. C. Jeong, M. So, C. E. Wilmer, A. A. Sarjeant, G. C. Schatz, R. Q. Snurr, O. K. Farha, G. P. Wiederrecht and J. T. Hupp, J. Am. Chem. Soc., 2013, 135, 862–869 CrossRef CAS PubMed.
- J. Liu, L. Chen, H. Cui, J. Zhang, L. Zhang and C.-Y. Su, Chem. Soc. Rev., 2014, 43, 6011–6061 RSC.
- (a) A. Dhakshinamoorthy, M. Alvaro, P. Horcajada, E. Gibson, M. Vishnuvarthan, A. Vimont, J.-M. Grenèche, C. Serre, M. Daturi and H. Garcia, ACS Catal., 2012, 2, 2060–2065 CrossRef CAS; (b) P. Horcajada, S. Surble, C. Serre, D.-Y. Hong, Y.-K. Seo, J.-S. Chang, J.-M. Greneche, I. Margiolaki and G. Ferey, Chem. Commun., 2007, 2820–2822 RSC; (c) A. Dhakshinamoorthy, M. Alvaro and H. Garcia, J. Catal., 2012, 289, 259–265 CrossRef CAS.
- (a) A. Dhakshinamoorthy, M. Alvaro and H. Garcia, Chem.–Eur. J., 2010, 16, 8530–8536 CrossRef CAS PubMed; (b) M. Opanasenko, A. Dhakshinamoorthy, J. Čejka and H. Garcia, ChemCatChem, 2013, 5, 1553–1561 CrossRef CAS; (c) A. Dhakshinamoorthy, M. Alvaro and H. Garcia, Chem. Commun., 2010, 46, 6476–6478 RSC; (d) A. Rapeyko, M. J. Climent, A. Corma, P. Concepción and S. Iborra, ChemSusChem, 2015, 8, 3270–3282 CrossRef CAS PubMed.
- (a) M. H. Beyzavi, R. C. Klet, S. Tussupbayev, J. Borycz, N. A. Vermeulen, C. J. Cramer, J. F. Stoddart, J. T. Hupp and O. K. Farha, J. Am. Chem. Soc., 2014, 136, 15861–15864 CrossRef CAS PubMed; (b) J. E. Mondloch, M. J. Katz, W. C. Isley Iii, P. Ghosh, P. Liao, W. Bury, G. W. Wagner, M. G. Hall, J. B. DeCoste, G. W. Peterson, R. Q. Snurr, C. J. Cramer, J. T. Hupp and O. K. Farha, Nat. Mater., 2015, 14, 512–516 CrossRef CAS PubMed.
- (a) A. Dhakshinamoorthy and H. Garcia, ChemSusChem, 2014, 7, 2392–2410 CrossRef CAS PubMed; (b) Y. Zhang, B. Li and S. Ma, Chem. Commun., 2014, 50, 8507–8510 RSC; (c) G. H. Dang, Y. T. H. Vu, Q. A. Dong, D. T. Le, T. Truong and N. T. S. Phan, Appl. Catal., A, 2015, 491, 189–195 CrossRef CAS.
- (a) H. R. Shaterian and A. R. Oveisi, Chin. J. Chem., 2009, 27, 2418–2422 CrossRef CAS; (b) H. R. Shaterian, A. R. Oveisi and M. Honarmand, Synth. Commun., 2010, 40, 1231–1242 CrossRef CAS.
- Q. Cao, L. M. Dornan, L. Rogan, N. L. Hughes and M. J. Muldoon, Chem. Commun., 2014, 50, 4524–4543 RSC.
- G. Majano, O. Ingold, M. Yulikov, G. Jeschke and J. Perez-Ramirez, CrystEngComm, 2013, 15, 9885–9892 RSC.
- (a) G. Férey, C. Mellot-Draznieks, C. Serre, F. Millange, J. Dutour, S. Surblé and I. Margiolaki, Science, 2005, 309, 2040–2042 CrossRef PubMed; (b) H.-j. Zhang, S.-d. Qi, X.-y. Niu, J. Hu, C.-l. Ren, H.-l. Chen and X.-g. Chen, Catal. Sci. Technol., 2014, 4, 3013–3024 RSC.
- (a) F. Millange, C. Serre, N. Guillou, G. Férey and R. I. Walton, Angew. Chem., Int. Ed., 2008, 47, 4100–4105 CrossRef CAS PubMed; (b) M. H. Alkordi, Y. Liu, R. W. Larsen, J. F. Eubank and M. Eddaoudi, J. Am. Chem. Soc., 2008, 130, 12639–12641 CrossRef CAS PubMed; (c) G. Ferey and C. Serre, Chem. Soc. Rev., 2009, 38, 1380–1399 RSC.
- M. O. Ratnikov and M. P. Doyle, J. Am. Chem. Soc., 2013, 135, 1549–1557 CrossRef CAS PubMed.
- C. Volkringer, H. Leclerc, J.-C. Lavalley, T. Loiseau, G. Férey, M. Daturi and A. Vimont, J. Phys. Chem. C, 2012, 116, 5710–5719 CAS.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra19013d |
| ‡ During the preparation of this work Wang and et al. published a paper related to quinazolinones synthesis using α-MnO2 as a catalyst.5d |
| This journal is © The Royal Society of Chemistry 2016 |