Iodine-catalyzed thiolation of electron-rich aromatics using sulfonyl hydrazides as sulfenylation reagents†
Abstract
Iodine-catalyzed thiolation of electron-rich aromatics, including substituted anisole, thioanisole, phenol, toluene, and naphthalene, using sulfonyl hydrazides as sulfenylation reagents was carried out. Sulfonothioates, the products of decomposition of sulfonyl hydrazides in the presence of iodine, are proposed as the major sulfenylation species in this transformation.
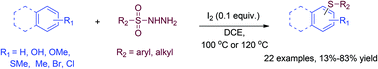
 Please wait while we load your content...
Please wait while we load your content...