Initial stage of the degradation of three common neonicotinoids: theoretical prediction of charge transfer sites†
Abstract
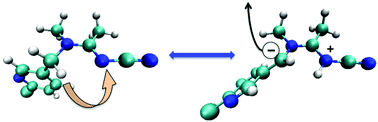
Three of the most important compounds in the insecticide industry, imidacloprid (IMI), thiacloprid (THIA) and acetamiprid (ACE), are theoretically analyzed. Wave-function and density functional theory (DFT) were used for the conformational analysis of the three compounds. In the context of conceptual DFT, by analyzing the local electrodonating power for the first stage of the degradation of these insecticides, we found that for THIA and ACE, solvated or free, there is more than one site where the electron is detached. Usually, just one nitrogen atom from the imidazolidine group is considered for the first step in the degradation process. However, in this work we propose at least one additional site for these two compounds, which involves another nitrogen atom. In addition, the theoretical approach presented in this article predicts one acetamiprid tautomer, which gives one additional site to detach one electron. Such a possibility has not been proposed before and to experimentally search new pathways for the degradation of these insecticides can give important information.


 Please wait while we load your content...
Please wait while we load your content...