Identification and optimisation of a series of N-(4-anilino-2-pyridyl)acetamide activin receptor-like kinase 1 (ALK1) inhibitors†‡
Abstract
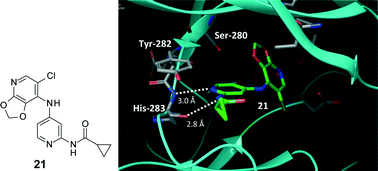
A novel class of N-(4-anilino-2-pyridyl)amide based activin receptor-like kinase (ALK1) inhibitors are disclosed, which were rapidly optimised to a ligand efficient probe compound 21 with good potency in enzyme (4 nM) and cell (45 nM) assays and favourable physical and pharmacokinetic properties (24 h free cover over cell IC50 after a single 50 mg kg−1 dose in nude mice). This was achieved by identifying a small, ligand efficient group in the solvent channel (C2) whilst optimising the selectivity pocket (C4) group for enzyme and cell potency, using related SAR that has been observed previously for Src inhibitors.

 Please wait while we load your content...
Please wait while we load your content...