Elementary and systemic views of the generation of toxic substances
Takayuki
Wakaki
a,
Kounosuke
Oisaki
a and
Motomu
Kanai
*ab
aGraduate School of Pharmaceutical Sciences, The University of Tokyo, 7-3-1 Hongo, Bunkyo-ku, Tokyo 113-0033, Japan. E-mail: kanai@mol.f.u-tokyo.ac.jp
bKanai Life Science Catalysis Project, JST-ERATO,
 | ||
| Fig. 1 The Twelve Principles of Green Chemistry.2 | ||
There is no denying the importance of this third principle. The time-dependent transition of the following three main alcohol oxidation methods represents the validity of this principle (Scheme 1). In the 1960s–1970s, chromium oxides such as pyridinium chlorochromate and pyridinium dichromate were frequently used for oxidation of alcohols to aldehydes, ketones, and carboxylic acids (Scheme 1.1). Those reagents generate stoichiometric amounts of heavy metal chromium-containing side products, which have significant toxicity to human health and the environment.3 The Swern oxidation, typically using dimethylsulfoxide (DMSO), oxalyl chloride, and triethylamine in methylene chloride solvent at −78 °C, does not produce heavy metal side products, and thus is a more suitable alternative for the relevant conversion than chromium oxide-mediated processes (Scheme 1.2). Indeed, the Swern oxidation is one of the most commonly used oxidation reactions on the laboratory scale. In industrial scale systems, however, the standard Swern oxidation is associated with several problems; (1) the toxicity and corrosive properties of oxalyl chloride, (2) a requirement for cryogenic reaction temperatures, (3) the use of carcinogenic, hepatopathic, and neuropathic methylene chloride solvent, and (4) the production of toxic carbon mono-oxide, fetid dimethyl sulfide, and triethylamine hydrochloride salt as stoichiometric side products. To address problems (1)–(3), trichloroacetic anhydride is a non-toxic and non-corrosive substituent of oxalyl chloride, allowing for alcohol oxidation reactions at 5 °C in toluene.4 The odor problem of dimethyl sulfide in (4) has been addressed by the subsequent oxidation of the produced dimethyl sulfide. With such modifications of the reaction conditions, Swern-type oxidations can be performed on an industrial scale.
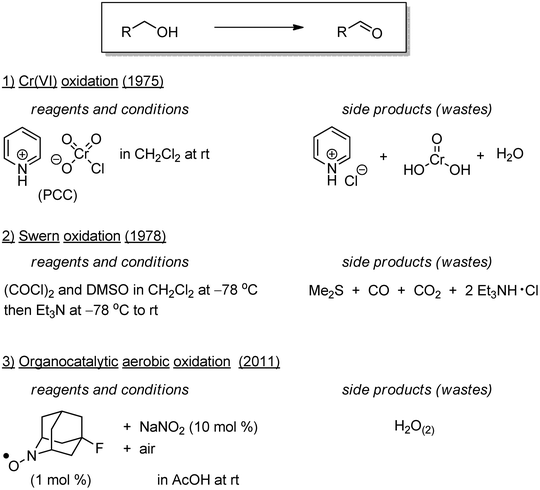 | ||
| Scheme 1 Progress of alcohol oxidation representing the 3rd principle of the Twelve Principles of Green Chemistry. | ||
Greener alcohol oxidation reactions using molecular oxygen as a terminal oxidant have been progressively developed over the last 25 years. The use of sustainable atmospheric oxygen in oxidative reactions is ideal in terms of availability and economic/environmental benefits. A terminal stoichiometric side product in these reactions is water. Groundbreaking research emerged from the field of homogeneous transition metal catalysis,5 especially that relying on palladium species.6 Based on pioneering works in the 1960s,7 Uemura reported a Pd(OAc)2/pyridine-catalyzed aerobic oxidation of alcohols in 1998.8 The reaction was extended to various applications, such as asymmetric catalysis,9 oxidations in water,10 and site-selective oxidation of polyols.11 With regard to the toxicity and rarity of palladium species, therefore, first-row transition metal catalysis is advantageous. Copper ion/N-oxyl radical catalyst conjugates, pioneered by Semmelhack in 1984,12 have been actively studied by Stahl, Iwabuchi, and our group since the 2010s.13 The reactions use inexpensive components and operationally simple procedures (e.g. in an open-flask at room temperature). The most remarkable advance beyond classical oxidation reactions is the high functional group-tolerance, allowing for late-stage modifications of complex molecules, such as natural products and proteins.13c
The recent progress of organocatalysis led to further developments of metal-free catalytic aerobic oxidation of alcohols.14 A milestone study was reported by Iwabuchi in 2011,15 using sterically-less demanding, chemically robust, and electronically tunable N-oxyl radicals (Scheme 1.3). In another direction, visible-light photoredox catalysis is paving the way for novel aerobic alcohol oxidation.16 Recent studies have also demonstrated the potential utility of organic dyes.17 The emerging concept of (organo)photoredox catalysis is providing a spectacular roadmap toward solar energy-based green synthetic chemistry.
Aerobic oxidation, however, is not very suitable for industrial-scale applications, at least so far. This is due both to difficulties in manipulating high-concentration oxygen gas on a large scale and the potential intermediary of explosive peroxide species. Conversely, development of industrial-scale aerobic oxidation processes is an important current challenge, as such processes could fulfill the requirements of green chemistry.
The transition of alcohol oxidation methods described above is in accord with the 3rd principle of the Twelve Principles of Green Chemistry. In addition to such an elementary viewpoint, however, the principle must be revisited today from a more systemic viewpoint. That is, even if a single chemical step generates substances that possess toxicity to human health and the environment, it may be acceptable provided that the overall reaction system reuses or recycles those toxic substances as intermediates in a whole chemical reaction chain. The concentration of toxic intermediates can be retained low enough as a whole system. Nature uses such a strategy for sustaining stationarity. The global carbon cycle is a typical example.18 Living activities, including respiration and industrial activities, generate carbon dioxide, a “toxic” substance to the environment. Plants, however, use carbon dioxide as a starting material in the photosynthesis of carbohydrates. By balancing the generation and assimilation of carbon dioxide in the global chemical system, the concentration was maintained at a constant level for many years. Today, however, excessive human activities have disrupted this balance. To re-adjust the balance, the acceleration of artificial carbon dioxide assimilation by developing sophisticated and robust chemical processes that can use carbon dioxide as a raw starting material for the synthesis of useful organic molecules will be important.19 The development of effective chemical methods for biomass conversion is also in accord with this direction.20 With re-adjustment of the global generation/assimilation balance from a systemic viewpoint, the carbon dioxide concentration could be maintained at an acceptable level. Such a systemic viewpoint is also valid to molecular synthesis; the overall chemical process will be green if generated toxic substances can be consumed in the overall synthetic scheme.
To summarize, the 3rd principle of the Twelve Principles of Green Chemistry today should be considered from a systemic viewpoint involving the reuse/recycling of generated toxic substances, as well as from an elementary viewpoint using unit reactions without the generation of toxic substances.
References
- P. Anastas, B. Han, W. Leitner and M. Poliakoff, Green Chem., 2016, 18, 12 RSC.
- P. T. Anastas and J. C. Warner, Green Chemistry: Theory and Practice, Oxford University Press, 1998 Search PubMed.
- K. E. Wetterhahn and D. Y. Cupo, Cancer Res., 1985, 45, 1146 Search PubMed.
- K. K.-C. Liu, S. M. Sakya, C. J. O'Donnell, A. C. Flick and H. X. Ding, Bioorg. Med. Chem., 2012, 20, 1155 CrossRef CAS PubMed.
- C. Parmeggiani and F. Cardona, Green Chem., 2012, 14, 547 RSC.
- (a) J. Muzart, Tetrahedron, 2003, 59, 5789 CrossRef CAS; (b) S. S. Stahl, Angew. Chem., Int. Ed., 2004, 43, 3400 CrossRef CAS PubMed; (c) M. J. Schultz and M. S. Sigman, Tetrahedron, 2006, 62, 8227 CrossRef CAS.
- (a) A. V. Nikiforova, I. I. Moiseev and Y. K. Syrkin, Zh. Obshch. Khim., 1964, 33, 3239 Search PubMed; (b) W. G. Lloyd, J. Org. Chem., 1967, 32, 2816 CrossRef CAS.
- (a) T. Nishimura, T. Onoue, K. Ohe and S. Uemura, Tetrahedron Lett., 1998, 39, 6011 CrossRef CAS; (b) T. Nishimura, T. Onoue, K. Ohe and S. Uemura, J. Org. Chem., 1999, 64, 6750 CrossRef CAS PubMed.
- (a) D. R. Jensen, J. S. Pugsley and M. S. Sigman, J. Am. Chem. Soc., 2001, 123, 7475 CrossRef CAS PubMed; (b) E. M. Ferreira and B. M. Stoltz, J. Am. Chem. Soc., 2001, 123, 7725 CrossRef CAS PubMed.
- G.-J. ten Brink, I. W. C. E. Arends and R. A. Sheldon, Science, 2000, 287, 1636 CrossRef CAS PubMed.
- K. Chung, S. M. Banik, A. G. De Crisci, D. M. Pearson, T. R. Blake, J. V. Olsson, A. J. Ingram, R. N. Zare and R. M. Waymouth, J. Am. Chem. Soc., 2013, 135, 7593 CrossRef CAS PubMed.
- M. F. Semmelhack, C. R. Schmid, D. A. Cortés and C. S. Chou, J. Am. Chem. Soc., 1984, 106, 3374 CrossRef CAS.
- (a) B. L. Ryland and S. S. Stahl, Angew. Chem., Int. Ed., 2014, 53, 8824 CrossRef CAS PubMed; (b) Y. Sasano, S. Nagasawa, M. Yamazaki, M. Shibuya, J. Park and Y. Iwabuchi, Angew. Chem., Int. Ed., 2014, 53, 3236 CrossRef CAS PubMed; (c) Y. Seki, K. Oisaki and M. Kanai, Angew. Chem., Int. Ed., 2014, 53, 6501 CrossRef CAS PubMed; (d) Y. Seki, K. Oisaki and M. Kanai, Tetrahedron Lett., 2014, 55, 3738 CrossRef CAS.
- S. Wertz and A. Studer, Green Chem., 2013, 15, 3116 RSC.
- M. Shibuya, Y. Osada, Y. Sasano, M. Tomizawa and Y. Iwabuchi, J. Am. Chem. Soc., 2011, 133, 6497 CrossRef CAS PubMed.
- M. Rueping, C. Vila, A. Szadkowska, R. M. Koenigs and J. Fronert, ACS Catal., 2012, 3, 2810 CrossRef.
- (a) D. A. Nicewicz and T. M. Nguyen, ACS Catal., 2014, 4, 355 CrossRef CAS; (b) S. Fukuzumi and K. Ohkubo, Org. Biomol. Chem., 2014, 12, 6059 RSC.
- R. A. Berner, Nature, 2003, 426, 323 CrossRef CAS PubMed.
- (a) Q. Liu, L. Wu, R. Jackstell and M. Beller, Nat. Commun., 2015, 6, 5933 CrossRef PubMed; (b) T. Sakakura, J. Choi and H. Yasuda, Chem. Rev., 2007, 107, 2365 CrossRef CAS PubMed.
- M. D. Kärkäs, B. S. Matsuura, T. M. Monos, G. Magallanes and C. R. J. Stephenson, Org. Biomol. Chem., 2016, 14, 1853 Search PubMed.
| This journal is © The Royal Society of Chemistry 2016 |
