Frustrated Lewis pair-mediated recrystallization of CH3NH3PbI3 for improved optoelectronic quality and high voltage planar perovskite solar cells†
Abstract
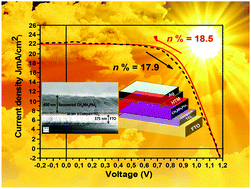
Films of the hybrid lead halide perovskite CH3NH3PbI3 were found to react with pyridine vapor at room temperature leading to complete bleaching of the film. In dry air or nitrogen atmosphere recrystallization takes place, leading to perovskite films with markedly improved optical and photovoltaic properties. The physical and chemical origin of the reversible bleaching and recrystallization mechanism was investigated using a variety of experimental techniques and quantum chemical calculations. The strong Lewis base pyridine attacks the CH3NH3PbI3. The mechanism can be understood from a frustrated Lewis pair formation with a partial electron donation of the lone-pair on nitrogen together with competitive bonding to other species as revealed by Raman spectroscopy and DFT calculations. The bleached phase consists of methylammonium iodide crystals and an amorphous phase of PbI2(pyridine)2. After spontaneous recrystallization the CH3NH3PbI3 thin films have remarkably improved photoluminescence, and solar cell performance increased from 9.5% for as-deposited films to more than 18% power conversion efficiency for recrystallized films in solar cells with planar geometry under AM1.5G illumination. Hysteresis was negligible and open-circuit potential was remarkably high, 1.15 V. The results show that complete recrystallization can be achieved with a simple room temperature pyridine vapor treatment of CH3NH3PbI3 films leading to high quality crystallinity films with drastically improved photovoltaic performance.


 Please wait while we load your content...
Please wait while we load your content...