Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Comparison of amorphous, pseudohexagonal and orthorhombic Nb2O5 for high-rate lithium ion insertion†
Shuang
Li
ab,
Qian
Xu
c,
Evan
Uchaker
b,
Xi
Cao
b and
Guozhong
Cao
*b
aSchool of Materials Science and Metallurgy, Northeastern University, Shenyang 110819, PR China
bDepartment of Materials Science and Engineering, University of Washington, Seattle, Washington 98195, USA. E-mail: gzcao@u.washington.edu
cState Key Laboratory of Advanced Special Steel, Shanghai University, Shanghai 200072, PR China
First published on 1st March 2016
Abstract
Amorphous, pseudohexagonal and orthorhombic Nb2O5 nanoparticles were synthesized using a facile and green sol–gel process followed by thermal treatment at 450 °C, 600 °C and 800 °C for 3 h in air, respectively. The resulting materials have been subjected to a detailed experimental study and comparison of their structural, electrical and electrochemical properties. The experiments have demonstrated that the pseudohexagonal Nb2O5 (TT-Nb2O5) exhibited higher storage capacity, largely due to its high specific surface area and small crystallites, and better cycling performance than both amorphous Nb2O5 (a-Nb2O5) and orthorhombic Nb2O5 (T-Nb2O5); such experimental findings were found to be associated with and thus ascribed to the lower charge transfer resistance and higher lithium ion diffusion coefficient of TT-Nb2O5 than those of a-Nb2O5 and T-Nb2O5. This research contributes to a better fundamental understanding of the relationship between the crystal structure and the crystallinity and electrochemical properties, particularly Li-ion storage properties, and leads to a possible new advancement in the research field of lithium ion batteries and pseudocapacitors.
Introduction
Electrochemical energy storage devices have attracted considerable attention in powering future advanced communication equipment, portable electronics, electric vehicles and smart grids.1–3 For electric vehicles (EVs), the current commercial lithium-ion batteries (LIBs) commonly using graphite as the anode cannot meet the stringent requirements of high power density, because of poor rate performance and especially safety concerns.4 Thus, developing advanced electrode materials, which possess better safety characteristics and high rates, becomes imperative. There are several promising candidates including silicon and tin-based compounds being actively studied as anodic materials for the next generation of batteries as they offer high lithium-ion storage capacities;5–8 however, they suffer from huge volume changes accompanied by lithium ion insertion and extraction.9 Lithium titanate (Li4Ti5O12) is another most noticeable anodic material under intensive study as it offers a high charge/discharge voltage of 1.55 V (vs. Li/Li+) and very stable cyclic stability since there is a very small volume change during lithium ion insertion and extraction; however, its lithium ion storage capacity is 170 mA h g−1 theoretically.10 Niobium pentoxide (Nb2O5) is another anodic material which attracts a lot of attention lately, due to its unique fast ion diffusion property during lithium ion intercalation and extraction, and most research are based on pseudocapacitors.11 It has been reported that the high rate is attributed to the fact that lithium ion insertion and extraction is rate-limited by the capacitive effect, not by lithium ion diffusion through the electrode.12 Compared to commercial graphite anodes, Nb2O5 exhibits a similar insertion/extraction voltage to Li4Ti5O12, which is higher than the 1 V voltage (vs. Li/Li+) of SEI formation and suppresses lithium dendrite deposition on the surface of the anode.13 In addition, Nb2O5 has a good theoretical lithium-ion storage capacity of 200 mA h g−1,14 which leads to a higher energy density and makes it a good candidate for niche applications.Due to its attractive physical properties, Nb2O5 has been extensively studied and applied in various technical applications such as electrochemical pseudocapacitors,11,12 gas sensors,15 catalysts,16 electrochromics17 and solar cells.17,18 The electrochemical properties of Nb2O5 were first investigated by Bard et al., who demonstrated that Nb2O5 exhibits lithium-ion intercalation properties, which led to further research of Nb2O5 as an anode for lithium ion batteries.19,20 In recent years, various nanostructures of Nb2O5 as electrodes for lithium ion batteries have been studied, such as nanotubes, nanowires, nanofibers and spherical nanoparticles.14,21–24 Some research studies revealed that Nb2O5–C composites and Ti- or Ta-substituted Nb2O5 possessed improved electrochemical properties.25–27 The Li intercalation properties of other Nb-based oxides are summarized in a recent review paper.28 Nb2O5 has many polymorphic forms, such as a-Nb2O5 (amorphous Nb2O5), TT-Nb2O5 (pseudohexagonal Nb2O5), T-Nb2O5 (orthorhombic Nb2O5) and M-Nb2O5 (monoclinic Nb2O5), which can be obtained mainly through controlled thermal treatment. The study on the transition and structures of TT-Nb2O5, T-Nb2O5 and M-Nb2O5 phases has been discussed in the literature.29 In Kodama et al.'s work, several crystal forms of Nb2O5 and their intercalation process were discussed mainly using in situ XAFS-XRD.30 A. Le Viet et al. reported that M-Nb2O5 exhibited the highest capacity compared with T-Nb2O5 and tetragonal Nb2O5; besides, heat-treated M-Nb2O5 (1100 °C) in argon shows a much enhanced discharge capacity of 192 mA h g−1 at a current density of 150 mA g−1.27 Recently, Kim et al. synthesized a-Nb2O5, TT-Nb2O5 and T-Nb2O5 using different methods and reported their rapid pseudocapacitive characteristics by drop casting Nb2O5 nanoscale powders onto steel foils as electrodes.11 These research studies have clearly indicated that Nb2O5 polymorphs are very promising lithium ion storage materials. What's more, fast transport kinetics is another important advantage along with high power density for lithium ion batteries besides large capacity. Thus, more studies based on a detailed comparison of the structural, electrical and electrochemical properties of a-Nb2O5, TT-Nb2O5 and T-Nb2O5 phase materials applied in lithium-ion batteries becomes necessary.
This paper reports the synthesis of nanostructured Nb2O5via a facile and green sol–gel process with forced hydrolysis and condensation and investigates the electrochemical properties of a-Nb2O5, TT-Nb2O5 and T-Nb2O5 nanoparticles, aimed at achieving a better fundamental understanding of the relationship between the electrochemical properties, in particular Li-ion storage properties, and the crystallinity and nanostructures. Specifically, we introduced NH4OH and H2O2 into the precursor solution to catalyze and promote rapid hydrolysis and condensation reactions. The resulting Nb2O5 nanoparticles were amorphous in nature and the subsequent annealing at elevated temperatures converted the amorphous niobium pentoxide to the pseudohexagonal phase and orthorhombic crystals. Upon evaluation as anode materials for lithium-ion batteries, the three Nb2O5 phases all showed very stable cycling performance, and the TT-Nb2O5 nanoparticles showed higher lithium ion storage capacity than both amorphous Nb2O5 and T-Nb2O5. Compared with the results for T-Nb2O5 and M-Nb2O5 reported in the literature, the pseudohexagonal crystal has the advantage of low processing temperature which can save energy in materials processing.
Experimental method
Materials synthesis
a-Nb2O5, TT-Nb2O5 and T-Nb2O5 nanoparticles were synthesized via a facile sol–gel process followed by annealing in air at 450 °C, 600 °C and 800 °C, respectively. In a typical reaction, 1 g of NbCl5 was dissolved in 2 ml of ethanol, and a clear yellow NbCl5 EtOH solution was formed. A white precipitate was subsequently obtained from this solution by dropwise addition of 5% dilute ammonia, and the pH value was tuned at 7.31 The precipitate was isolated from the solution and washed with deionized water four times by centrifugation at 6000 rpm. Then, 4 ml of 30 wt% aqueous hydrogen peroxide was added to the precipitate. The mixture was stirred for 5 minutes in an ice bath, and then a transparent yellow sol was obtained. This sol was dried in an oven at 70 °C for 24 hours and then a clear yellow xerogel was obtained. The xerogel was ground into white particles and then annealed under ambient atmosphere at 450 °C (a-Nb2O5), 600 °C (TT-Nb2O5) and 800 °C (T-Nb2O5) for 3 hours.Materials characterization
The crystalline structure of the particles obtained was characterized by X-ray diffraction (XRD, D8 Bruker X-ray diffractometer with Cu-Kα radiation (λ = 1.5418 Å)) within the range of 10–70° (2θ). X-ray photoelectron spectroscopy (XPS) analysis was performed using a VG Multilab 2000 with Al Kα as the X-ray source. The accelerating voltage and current were 40 kV and 40 mA, respectively. Scanning electron microscopy (SEM, JEOL, JSM-7000F) and transmission electron microscopy (TEM, FEI, Tecnai G2 F20) were used to examine the morphology and microstructure of the as-prepared samples. Thermogravimetric analysis (TGA) was performed on a TG instrument (NETZSCH STA 409C). Nitrogen adsorption–desorption isotherms were measured using a Quantachrome NOVA 4200e system. Samples were degassed at 250 °C under vacuum overnight prior to measurements. The specific surface areas and micropore volumes were determined by multipoint Brunauer–Emmett–Teller (BET), t-method and Barrett–Joyner–Halenda (BJH) desorption analyses, respectively.Electrochemical property measurement
The electrochemical performance of the Nb2O5 polymorphs was tested in coin-type cells, which were assembled using the as-prepared samples in an argon-filled glove-box (Innovative Technology, IL-2GB). The as-prepared active materials were mixed and ground with poly(vinylidene fluoride) (PVDF, Sigma-Aldrich) as a binder and Super P conductive carbon (TIMCAL Graphite & Carbon) as the conductive assistant materials at a respective weight ratio of 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20
20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 in an N-methyl-2-pyrrolidone (NMP, Alfa Aesar) solution. The as-prepared mixture was uniformly spread and pressed onto an aluminum foil and dried in a vacuum oven at 80 °C overnight for use as the working electrode. The mass of the active material is approximately 0.5 mg. A pure lithium foil was used as the counter and reference electrodes. A polypropylene membrane film (Celgard 2400) was used as the separator. 1 M LiPF6 in ethylene carbonate (EC)/dimethyl carbonate (DMC) was used as the electrolyte. Galvanostatic charge/discharge measurements were performed in the voltage range of 1.2 to 3.0 V (vs. Li/Li+) under different current densities. Cyclic voltammetry (CV) was conducted on an electrochemical analyzer (CH Instruments, model 605C) in the voltage range of 1.2 to 3.0 V (vs. Li/Li+) at a scan rate of 0.2 mV s−1. Electrochemical impedance spectroscopy (EIS) studies were performed on a Solartron 1287A in conjunction with a Solartron 1260FRA/impedance analyzer with an amplitude of 5.0 mV in the frequency range from 100 kHz to 0.1 Hz. The half-cells were tested at various current rates based on the weight of the active material.
10 in an N-methyl-2-pyrrolidone (NMP, Alfa Aesar) solution. The as-prepared mixture was uniformly spread and pressed onto an aluminum foil and dried in a vacuum oven at 80 °C overnight for use as the working electrode. The mass of the active material is approximately 0.5 mg. A pure lithium foil was used as the counter and reference electrodes. A polypropylene membrane film (Celgard 2400) was used as the separator. 1 M LiPF6 in ethylene carbonate (EC)/dimethyl carbonate (DMC) was used as the electrolyte. Galvanostatic charge/discharge measurements were performed in the voltage range of 1.2 to 3.0 V (vs. Li/Li+) under different current densities. Cyclic voltammetry (CV) was conducted on an electrochemical analyzer (CH Instruments, model 605C) in the voltage range of 1.2 to 3.0 V (vs. Li/Li+) at a scan rate of 0.2 mV s−1. Electrochemical impedance spectroscopy (EIS) studies were performed on a Solartron 1287A in conjunction with a Solartron 1260FRA/impedance analyzer with an amplitude of 5.0 mV in the frequency range from 100 kHz to 0.1 Hz. The half-cells were tested at various current rates based on the weight of the active material.
Results and discussion
When NbCl5 was dissolved in absolute ethanol, the following reaction would occur, leading to the formation of niobium chloride ethoxide:32| NbCl5 + xCH3CH2OH → NbCl5−x(CH3CH2O)x + xHCl | (1) |
Continuous stirring would allow the partial removal of HCl through evaporation, promoting reaction (1) further, and also the absorption of H2O from air, resulting in partial hydrolysis:
| NbCl5−x(CH3CH2O)x + yH2O → Nb(CH3CH2O, OH)5 + (5 − x)HCl + (y + x − 5)HOCH2CH3 | (2) |
When NH4OH was subsequently added, it reacted with HCl, increased the pH value, and promoted the hydrolysis and condensation reactions of niobium alkoxide as a catalyst, and the white precipitate of Nb(OH)5 was subsequently obtained:
| NH4OH + HCl → NH4Cl + H2O | (3) |
| Nb(CH3CH2O, OH)5 + 5H2O → Nb(OH)5 + HOCH2CH3 | (4) |
NH4Cl is a white precipitate in ethanol.32 However, in the present work, no NH4Cl precipitate was observed, probably due to the low percentage of ethanol.
Collectively, the purpose of adding NH4OH to the niobium oxide sol is to promote both the hydrolysis and condensation reactions, and it consequently resulted in a rapid nucleation process with a high density of nuclei of narrow size distribution. The addition of H2O2 dissolved the precipitate and turned the colour from ivory to yellow; upon drying in an oven at 70 °C, the release of the oxygen gas by-product benefits the nucleation process as well. The release and removal of oxygen gas bubbles from the sol helped homogenize the concentration and temperature of the sol so as to ensure a uniform size of the nanoparticles, and also prevented the possible formation of agglomerated nanoparticles. The SEM images shown in Fig. 4b and d have demonstrated the difference between the as-synthesized Nb2O5 nanoparticles synthesized with and without the addition of H2O2 through a sol–gel process under otherwise the same conditions. The Nb2O5 nanoparticles synthesized with the addition of H2O2 are smaller and are dispersed better, i.e., less aggregated.
Through a thermal treatment process, different Nb2O5 polymorphs were formed using the above-mentioned method. The crystalline structure and crystallinity of the as-prepared materials annealed at different temperatures were examined by XRD, and the results are displayed in Fig. 1. The intensity and shape of the diffraction peaks vary greatly as the annealing temperature changes. The as-prepared Nb2O5 xerogel was amorphous. At the annealing temperature of 450 °C, the diffraction peaks are mild, and the pattern has a shape close to that of an amorphous crystal. This might be attributed to the fact that the particles could not change into crystals very well at this annealing temperature. When the annealing temperature is 600 °C, pseudohexagonal crystals of Nb2O5 (TT-Nb2O5) are formed. All the diffraction peaks are clear and in good agreement with those of TT-Nb2O5 (space group: P6/mmm, JCPDS: 28-317), though the intensity of the peaks is weak, the peaks are broad and the signal-to-noise ratio is low, which are all indicative of small crystal size and/or poor crystallinity. The average crystallite size of TT-Nb2O5 was calculated to be approximately 22.5 nm using the Scherrer equation. Between 600 °C and 800 °C, there is a crystal transition. When the annealing temperature is raised to 800 °C, the diffraction peaks are consistent with the peaks of orthorhombic Nb2O5 (T-Nb2O5, space group: Pbam, JCPDS: 30-0873). The average crystallite size was estimated to be ∼113.3 nm using the Scherrer equation. The crystallites of the T-phase are significantly larger than those of TT-Nb2O5, as the annealing temperature is much higher. The high intensity and narrow width of the T-Nb2O5 peaks imply that it crystallized very well at the annealing temperature of 800 °C.
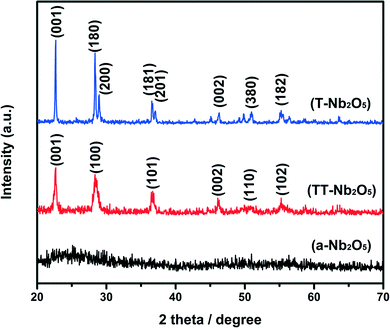 | ||
| Fig. 1 XRD patterns of Nb2O5 polymorphs annealed in air for 3 hours at different temperatures: (a-Nb2O5): 450 °C; (TT-Nb2O5): 600 °C; (T-Nb2O5): 800 °C. | ||
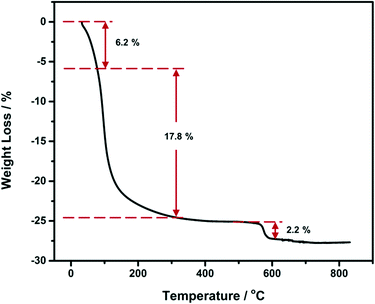
In order to study the crystalline water loss and the crystal transition during the annealing process, TGA was carried out in flowing air from 30 °C to 830 °C with a heating rate of 10 °C min−1 and the TGA curve of the pure Nb2O5 xerogel is shown in Fig. 2. From the TGA curve, we can see that the weight loss started with a mild curve from room temperature to around 90 °C which is supposed to be the physically adsorbed water molecules. A large mass loss of about 20% from 90 to 300 °C mainly involves fragments of organic molecules (CxHy) and HCl, and it is attributed to the build-up of the inorganic amorphous network.33 The lower mass loss of 2.2% at around 580 °C accompanying the crystallization process is due to a transformation from an uncharacterized niobium pentoxide hydrate, also known as niobic acid or columbic acid (Nb2O5·nH2O), gel to a Nb2O5 crystal (pseudohexagonal).31 From 600 °C to 800 °C, the weight loss is less than 0.5%, which means that although the morphology variation is significant and the crystal structure changes from pseudohexagonal to orthorhombic, the weight loss is inconspicuous. This result agrees well with the works published in the literature.17,24 In these works, no appreciable weight loss or crystallization peaks (from their differential thermal analysis results) were observed for the crystal structure change from pseudohexagonal to orthorhombic.
XPS analyses were carried out in the region of 0–800 eV to elucidate the chemical composition. Fig. 3 shows the typical XPS spectra taken from the niobium and oxygen regions of TT-Nb2O5. The binding energy scales of the spectra were calibrated by assigning the most intense C 1s peak a binding energy of 284.8 eV. Fig. 3a reveals that the sample contains the Nb, O and C elements. In Fig. 3b, the Nb 3d level binding energies of TT-Nb2O5 were 207.3 and 210.0 eV, which are attributed to the doublet Nb 3d5/2 and Nb 3d3/2 core levels of Nb5+, respectively. These binding energies of Nb5+ ions are in close agreement with the values reported.34,35Fig. 3c shows the binding energies of the O 1s peaks. Among these peaks, the sharp one located at 529.6 eV and the low one located at 531.9 eV are assigned to O2− in Nb-oxides and surface oxygen, respectively.24 Another O 1s peak located at 530.7 eV could be the peak of C–O from some possible residual groups.
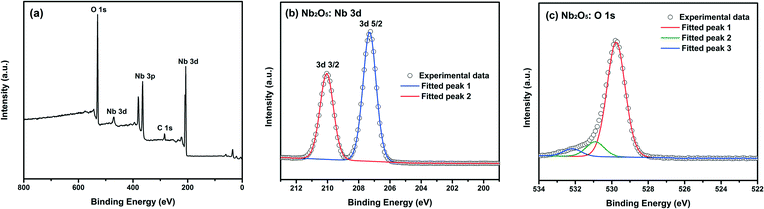 | ||
| Fig. 3 XPS spectrum of (a) wide survey of TT-Nb2O5 nanoparticles, (b) the zone-in Nb 3d spectrum and (c) the zone-in O 1s spectrum. | ||
SEM and TEM were carried out to investigate the microstructure and morphology of the as-synthesized samples. As shown in Fig. 4a, a-Nb2O5 annealed at 450 °C has smaller particle size, better dispersion and higher porosity. From Fig. 4b, it can be clearly seen that the particles are formed homogeneously after being annealed at 600 °C for 3 h in air, and the TT-Nb2O5 particles have a narrow size distribution at around 20 nm. When the powders were annealed at 800 °C (Fig. 4c), larger-sized T-Nb2O5 particles of around 100 nm size were obtained. This means that the rise in temperature not only modified the crystalline phase but also increased the size. Fig. 4d presents the SEM image of the Nb2O5 particles without addition of hydrogen peroxide and annealed at 600 °C for 3 h in air. In this image, connections between particles can be found. By comparing Fig. 4b and d, it can be clearly seen that the particles in Fig. 4b have much better uniform dispersion, which also proved that the addition of H2O2 can improve the dispersity of Nb2O5 nanoparticles. The high-resolution TEM images provide more detailed structural information as seen in Fig. 5. Compared to a-Nb2O5 shown in Fig. 5a, the lattice fringes of the TT-Nb2O5 and T-Nb2O5 particles can be clearly observed in Fig. 5b and c, suggesting the presence of a well-defined crystal structure. The periodic lattice fringe spacings in Fig. 5b were found to be 0.32 and 0.25 nm corresponding to the interplanar spacings of the (100) and (101) planes, respectively, for pseudohexagonal Nb2O5 (JCPDS: 28-317); in Fig. 5c, the periodic lattice fringe spacings were 0.38 and 0.31 nm which agreed well with the interplanar spacings of the (001) and (180) planes. These results further corroborate the findings from XRD.
The presence of pores and the porous distribution of the Nb2O5 polymorphs were confirmed by the corresponding nitrogen sorption isotherms and Barrett–Joyner–Halenda (BJH) desorption analyses which are shown in Fig. 6. The results in Fig. 6a indicated that the pore size of a-Nb2O5 centrally distributed at 3.5 nm which was the only mesopore distribution can be found, and the Brunauer–Emmett–Teller (BET)-derived surface area was determined to be 129.6 m2 g−1. Within the TT-Nb2O5 sample, the majority of the pores are around 15 nm (Fig. 6b) and exhibit a very narrow pore size distribution. The BET surface area of TT-Nb2O5 was 57.8 m2 g−1, which is a reasonable value providing excellent channels and cavities for complete and homogeneous Li+ diffusion and intercalation throughout the material.36 For the T-Nb2O5 sample (Fig. 6c), no obvious pore size distribution was observed in our study, and the peaks of pore size distributions at 3.5 and 5 nm are very weak. The surface area of T-Nb2O5 turns out to be only 43.0 m2 g−1, which is far lower than those of a-Nb2O5 and TT-Nb2O5.
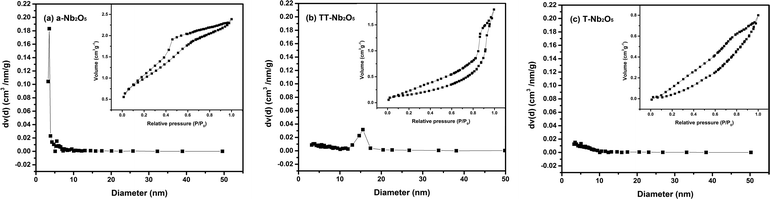 | ||
| Fig. 6 BJH desorption pore size distribution for the as-synthesized Nb2O5 polymorphs: (a) a-Nb2O5, (b) TT-Nb2O5 and (c) T-Nb2O5 (inset: N2 adsorption–desorption isotherm). | ||
In order to investigate the storage properties of the Nb2O5 polymorphs, CV, galvanostatic charge/discharge measurements and electrochemical impedance analysis have been performed. The CV curves of the first, second and fifth runs for the a-Nb2O5, TT-Nb2O5 and T-Nb2O5 samples collected at a scan rate of 0.2 mV s−1 in the voltage range of 1.2–3.0 V (vs. Li/Li+) are shown in Fig. 7. The three Nb2O5 polymorphs all showed good reusability. For T-Nb2O5 (Fig. 7c), the current response of the cathodic sweep increases greatly below 2.1 V (vs. Li/Li+), and two obvious reduction reactions located at 1.72 and 1.54 V correspond to the valence variation during the intercalation process. On the reverse sweep, an ambiguous broad peak at 1.82 V reveals that the continuous variation from Nb4+ back to Nb5+ takes place during the redox process.30 The cathodic and anodic peaks of TT-Nb2O5 (Fig. 7b) are broad and located at 1.5 V and 1.85 V, respectively. In contrast, the CV peaks of the a-Nb2O5 material (Fig. 7a) are very obscure except for the first cycle which exhibits a small reduction peak at 2.0 V and a wide shift of the CV curve to the other cycles.
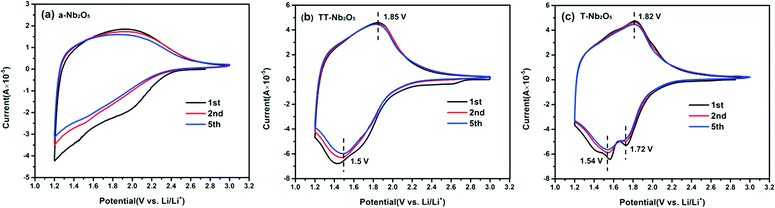 | ||
| Fig. 7 CV curves of Nb2O5 polymorph electrode materials: (a) a-Nb2O5, (b) TT-Nb2O5 and (c) T-Nb2O5 measured in the voltage range of 1.2–3.0 V (vs. Li/Li+) with a scan rate of 0.2 mV s−1. | ||
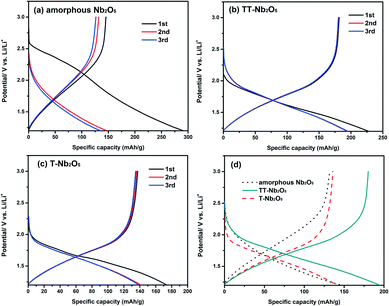
Fig. 8 displays the charge–discharge profiles of the Nb2O5 polymorphs for the first three cycles at a current rate of 10 mA g−1 in the voltage range of 1.2–3.0 V (vs. Li/Li+). As shown in Fig. 8a, a-Nb2O5 exhibited an initial discharge capacity of 291 mA h g−1, but it dropped to 150 mA h g−1, nearly half of the first discharge capacity, after the second cycle. Similarly, TT-Nb2O5 and T-Nb2O5 showed a decrease in capacity following the first cycle; however, the proportion of reversible capacity in the second cycle is much larger than that for a-Nb2O5, at around 80% for both TT-Nb2O5 and T-Nb2O5. a-Nb2O5 exhibited the highest initial discharge capacity and the largest decay in the second cycle. This agrees well with the CV curves in Fig. 7a, which possess a wide shift between the first and second reduction curves, suggesting a large capacity decrease. In contrast, the CV curves for crystalline Nb2O5 (Fig. 7b and c) are more reversible between the first two cycles. In addition, from Fig. 7a, a mild reduction peak located at 2.0 V revealed that an irreversible chemical reaction with Li could exist.37 On the other hand, from the TGA analysis in Fig. 2, a-Nb2O5 should involve the loss of 2.2% located at 580 °C in the form of hydroxyl groups (O–H), which could bring about an undesirable irreversible electrochemical reaction with Li in the initial cycle.38,39 The existence of O–H in amorphous materials has been proven in previous reports, and it is also not detected in a fully crystallized material.40 From the figures of TT-Nb2O5 and T-Nb2O5, curves of a similar shape can be found which have no plateaus, implying that the charge and discharge processes of the TT-Nb2O5 and T-Nb2O5 samples are very fast, which are in good agreement with the characteristics of pseudocapacitance.12,41 Among the three samples, TT-Nb2O5 shows the highest reversible charge/discharge capacities of about 190 mA h g−1 which is much higher than the capacity of 110 mA h g−1 reported by Kodama et al.30 Such a high capacity might be the result of a high specific surface area and the nanosized crystallites. For the larger-sized T-Nb2O5, the reversible charge/discharge capacities in the present study are similar to the results reported in the literature.27 From here, we see that the difference is probably because of the fact that the nanoscale TT-Nb2O5 particles obtained by this sol–gel method can provide a larger surface area for Li+ de/intercalation and thereby improve the capacity.3 More capacities under different charge rates and stabilities are shown in Fig. 9.
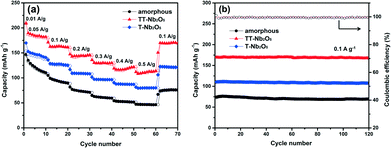 | ||
| Fig. 9 (a) Rate capability of the Nb2O5 polymorphs at different current densities and (b) cycling performance of the Nb2O5 polymorphs at a current density of 100 mA g−1. | ||
The rate performance of the a-Nb2O5, TT-Nb2O5 and T-Nb2O5 samples at various charge–discharge rates is compared in Fig. 9. The charge–discharge curves show that all these three Nb2O5 polymorphs have very regular performance. No obvious fading can be found after 120 cycles. Among these samples, the TT-Nb2O5 electrode shows the highest capacity at each current rate. The discharge capacities measured in the voltage window from 1.2 V to 3 V are 191, 164, 146, 130, 122 and 114 mA h g−1 at the current densities of 50, 100, 200, 300, 400 and 500 mA g−1, respectively. More importantly, when the current rate was reversed back to 100 mA g−1 after testing at the abovementioned different rates, the discharge capacity went back up to 172 mA h g−1 which is even higher than the initial discharge capacity at the same rate (164 mA h g−1). This implies that the decline in capacity at high rates is limited. After 120 charge–discharge cycles, the TT-Nb2O5 electrode demonstrated a specific discharge capacity of 170 mA h g−1 at a current density of 100 mA g−1, which is close to the capacity of the high-temperature phase, the pristine monoclinic phase, in a report.27 Possessing relatively stable charge–discharge performance, the T-Nb2O5 electrode showed much lower capacities than the TT-Nb2O5 electrode at each current rate mentioned above, and its specific discharge capacity was stable at 110 mA h g−1 during 120 charge–discharge cycles. Apparent fading was observed during the first 20 cycles in the rate capability of the a-Nb2O5 electrode. What's more, the a-Nb2O5 electrode showed even lower capacities at different current rates; especially, when the current rate was 500 mA g−1, the capacity fell to as low as 50 mA h g−1. The intercalation reaction is sensitive to doping, preparation temperature and particle size which was also documented in TiO2 as well as other systems.42 It should be noted that research on other transition-metal oxides such as Fe2O3 and MnOx showed that the amorphous phase possessed much enhanced performance compared to its crystalline counterpart.37,43 The reason for these results has been explained to be their unique structures which are beneficial for lithium ion storage. Upon comparing the CV curves between the amorphous and crystalline phases, only small changes could be found such as slight shifts of peak location, peak intensity or/and peak width. However, the main CV curves between the amorphous and crystalline samples are very similar, which implies that, for these two materials, there are no significant mechanism differences in the lithiation process caused by changing the crystalline material into an amorphous one. In contrast, our CV results reveal that there are either some mechanism changes in the lithiation process or some capacity deficiency because the CV curve of a-Nb2O5 significantly changed with a lack of redox reaction peaks compared with that of crystalline Nb2O5. Those different lithiation mechanisms may explain the result that crystalline Nb2O5 showed better lithium ion storage properties than a-Nb2O5 in the present work.
For the sake of comparison of the electrochemical properties of M-Nb2O5, the Nb2O5 precursor was annealed at 1100 °C in air for 3 h and the resulting monoclinic phase was obtained as confirmed by the XRD patterns shown in Fig. S1a (ESI†). The electrochemical properties (shown in Fig. S1, ESI†) turned out to be in good agreement with those reported in the literature.24 More specifically, the discharge–charge profiles showed a clear discharge plateau at a potential around 1.65 V, which is the same as that reported in the literature.24 However, the CV curves were slightly different in appearance, which was likely due to the high scan rate used in the current study as compared with that in the literature (0.2 mV s−1vs. 0.058 mV s−1). The resulting M-Nb2O5 demonstrated a high reversible capacity of 185 mA h g−1 at a low rate of 10 mA g−1. The capacity was found to be 120 mA h g−1 at a high rate of 100 mA g−1. The M-Nb2O5 in the current investigation possessed a smaller capacity as compared to TT-Nb2O5.
To investigate the electrochemical kinetics of the Li+ intercalation/deintercalation process within the Nb2O5 polymorphous electrode, EIS measurement was performed over a frequency range of 100 kHz to 0.1 Hz with the results shown in Fig. 10. The impedance data were recorded for fresh cells after 5 discharge–charge cycles at a current rate of 0.5 C. The voltage was 3.0 V under the charging status. As shown in the figure, the Nyquist plots of the a-Nb2O5, TT-Nb2O5 and T-Nb2O5 electrodes display a single semicircle in the high-frequency region and a straight line in the low-frequency range. The EIS was simulated through Z-view software using the equivalent circuit shown in the inset.35,44,45 It consists of the solution resistance (Rs), charge transfer (Rct) resistance, a constant phase element (CPE1) and diffusional component Warburg impedance (Zw). The semicircle at the high-frequency region is associated with the charge transfer resistance (Rct).46,47 The fitted Rct values are 332.9 Ω, 261.5 Ω and 400.9 Ω for the a-Nb2O5, TT-Nb2O5 and T-Nb2O5 electrodes, respectively.
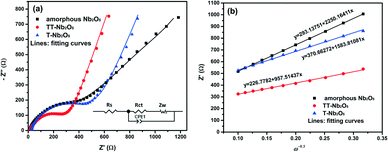 | ||
| Fig. 10 (a) EIS plots of the Nb2O5 polymorphs at 25 °C and (b) the Z′–ω−0.5 plots in the low-frequency range. | ||
With the lowest Rct value, TT-Nb2O5 demonstrates better electrical conductivity, which can be a reason for its higher capacity and cycling performance. The straight line in the low-frequency range stands for the Warburg impedance.48 The diffusion coefficient of lithium ions, DLi, as one of the main kinetic factors in the rate-determining step of the intercalation process, can be calculated using several techniques, such as CV, EIS and the galvanostatic intermittent titration technique (GITT). These testing methods are based on their respective formula derived by using Fick's law and the Nernst equation. The differences mostly come from adopting different boundary conditions, initial conditions and numerical analysis methods. The CV technique can provide quantitative information on the electrode process, and the reversibility, stability and phase transformations during the intercalation and deintercalation reactions. The GITT is established to be a reliable technique for investigating the DLi with highly resolved data for intercalation compounds of varying lithium content or potential. EIS is also a powerful technique for calculating the DLi due to the fact that the low-frequency Warburg contribution of the impedance response is directly related to the lithium-ion diffusion process in an electrode material.49 Slight differences in the diffusion coefficient may exist when different testing techniques were applied; however, DLi obtained from the same technique can still be used to analyze and compare different systems and different electrode materials. The DLi here was calculated from the plots in the low-frequency region. The equation for the calculation of DLi values by EIS can be expressed as follows:50–52
| Z′ = Rb + Rct + σω−0.5 | (5) |
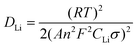 | (6) |
Based on the fitting linear equation in Fig. 10b, the Li-ion diffusion coefficients of a-Nb2O5, TT-Nb2O5 and T-Nb2O5 were calculated and the results are 1.39 × 10−21 cm2 s−1, 3.86 × 10−20 cm2 s−1 and 2.56 × 10−20 cm2 s−1, respectively. The lower surface film and charge transfer resistance, higher Li-ion diffusion coefficient and electrical conductivity of the TT-Nb2O5 electrode lead to its superior rate capabilities and other electrochemical properties. Table 1 presents a summary of BET and EIS data of the three Nb2O5 polymorphs. In general, a larger surface area provides more reaction sites and shorter diffusion paths for ionic transport and electronic conduction, leading to a higher power density.53 In this work, the crystallinity and crystal structure exhibit an obvious influence on the electrical conductivity as well as the ionic diffusivity. The surface area of a-Nb2O5 is twice as large as that of TT-Nb2O5; however, its resistance is lower than that of TT-Nb2O5. This means that during the process of crystal phase transition between the amorphous and TT phases, better crystallinity significantly reduced the value of resistance. In general, better crystallinity means more ordered arrangement of ions and fewer defects, which collectively offer more ordered passage for charge and mass transport, leading to fast diffusion and high conductivity (low resistance). During the transformation from the amorphous to TT phase, the crystals tend to be perfect. When the crystal phase transformed from the TT phase to the T phase, although the crystallinity improved to some extent, the particle size became larger, too. A larger particle size brings about long diffusion paths for ionic transport which can be a reason for high resistance.
| Samples | Specific surface area, m2 g−1 | Pore size, nm | R s, Ω | R ct, Ω | Diffusion coefficients, cm2 s−1 |
|---|---|---|---|---|---|
| a-Nb2O5 | 130 | 3.5 | 12.09 | 332.9 | 1.39 × 10−21 |
| TT-Nb2O5 | 58 | 15 | 12.38 | 261.5 | 3.86 × 10−20 |
| T-Nb2O5 | 43 | — | 12.75 | 400.9 | 2.56 × 10−20 |
Conclusions
a-Nb2O5, TT-Nb2O5 and T-Nb2O5 nanoparticles were synthesized using a facile and environmentally benign sol–gel process followed by thermal treatment. The size of the TT-Nb2O5 particles is around 20 nm, which is much smaller than that of T-Nb2O5 which is around 100 nm. The Nb2O5 nanoparticles synthesized with the addition of H2O2 are smaller and are dispersed better. Upon evaluation as anode materials for lithium-ion batteries, the a-Nb2O5, TT-Nb2O5 and T-Nb2O5 electrodes demonstrated specific discharge capacities of 149 mA h g−1, 191 mA h g−1 and 152 mA h g−1, respectively, at a current density of 50 mA g−1. The large capacity of TT-Nb2O5 is most likely attributable to its large specific surface area. All these three Nb2O5 polymorphs have very stable cyclic stability within 120 cycles at a current density of 100 mA g−1. In addition to its large capacity, TT-Nb2O5 possesses lower charge transfer resistance, a higher lithium ion diffusion coefficient and better electrical conductivity than both a-Nb2O5 and T-Nb2O5, which might be ascribed to its favourable crystal structure.Acknowledgements
Shuang Li would like to acknowledge the financial support in the form of a scholarship from the China Scholarship Council (CSC) for her Ph.D. study at the University of Washington. This research was also financially supported in part by the National Science Foundation (NSF, DMR-1505902).Notes and references
- M. Armand and J. M. Tarascon, Nature, 2008, 451, 652–657 CrossRef CAS PubMed.
- C. Liu, F. Li, L.-P. Ma and H.-M. Cheng, Adv. Mater., 2010, 22, E28–E62 CrossRef CAS PubMed.
- Q. Zhang, E. Uchaker, S. L. Candelaria and G. Cao, Chem. Soc. Rev., 2013, 42, 3127–3171 RSC.
- M. M. Rahman, R. A. Rani, A. Z. Sadek, A. S. Zoolfakar, M. R. Field, T. Ramireddy, K. Kalantar-zadeh and Y. Chen, J. Mater. Chem. A, 2013, 1, 11019–11025 CAS.
- A. Magasinski, P. Dixon, B. Hertzberg, A. Kvit, J. Ayala and G. Yushin, Nat. Mater., 2010, 9, 353–358 CrossRef CAS PubMed.
- S.-H. Ng, J. Wang, D. Wexler, K. Konstantinov, Z.-P. Guo and H.-K. Liu, Angew. Chem., Int. Ed., 2006, 45, 6896–6899 CrossRef CAS PubMed.
- L.-F. Cui, Y. Yang, C.-M. Hsu and Y. Cui, Nano Lett., 2009, 9, 3370–3374 CrossRef CAS PubMed.
- J. Fan, T. Wang, C. Yu, B. Tu, Z. Jiang and D. Zhao, Adv. Mater., 2004, 16, 1432–1436 CrossRef CAS.
- S. D. Beattie, D. Larcher, M. Morcrette, B. Simon and J.-M. Tarascon, J. Electrochem. Soc., 2008, 155, A158–A163 CrossRef CAS.
- M. R. Jo, K. M. Nam, Y. Lee, K. Song, J. T. Park and Y.-M. Kang, Chem. Comm., 2011, 47, 11474–11476 RSC.
- J. W. Kim, V. Augustyn and B. Dunn, Adv. Energy Mater., 2012, 2, 141–148 CrossRef CAS.
- A. Veronica, C. Jérémy, A. L. Michael, K. Jong Woung, T. Pierre-Louis, H. T. Sarah, D. A. Héctor, S. Patrice and D. Bruce, Nat. Mater., 2013, 12, 518–522 CrossRef PubMed.
- L. Shen, B. Ding, P. Nie, G. Cao and X. Zhang, Adv. Energy Mater., 2013, 3, 1484–1489 CrossRef CAS.
- M. Wei, Z.-M. Qi, M. Ichihara and H. Zhou, Acta Mater., 2008, 56, 2488–2494 CrossRef CAS.
- R. A. Rani, A. S. Zoolfakar, J. Z. Ou, M. R. Field, M. Austin and K. Kalantar-zadeh, Sens. Actuators, B, 2013, 176, 149–156 CrossRef CAS.
- D. Prasetyoko, Z. Ramli, S. Endud and H. Nur, Mater. Chem. Phys., 2005, 93, 443–449 CrossRef CAS.
- M. A. Aegerter, Sol. Energy Mater. Sol. Cells, 2001, 68, 401–422 CrossRef CAS.
- A. Le Viet, R. Jose, M. Reddy, B. Chowdari and S. Ramakrishna, J. Phys. Chem. C, 2010, 114, 21795–21800 CAS.
- B. Reichman and A. J. Bard, J. Electrochem. Soc., 1980, 127, 241–242 CrossRef CAS.
- B. Reichman and A. J. Bard, J. Electrochem. Soc., 1981, 128, 344–346 CrossRef CAS.
- C. Yan and D. Xue, Adv. Mater., 2008, 20, 1055–1058 CrossRef CAS.
- M. V. Reddy, R. Jose, A. Le Viet, K. I. Ozoemena, B. V. R. Chowdari and S. Ramakrishna, Electrochim. Acta, 2014, 128, 198–202 CrossRef CAS.
- M. Sasidharan, N. Gunawardhana, M. Yoshio and K. Nakashima, Mater. Res. Bull., 2012, 47, 2161–2164 CrossRef CAS.
- A. L. Viet, M. V. Reddy, R. Jose, B. V. R. Chowdari and S. Ramakrishna, J. Phys. Chem. C, 2009, 114, 664–671 Search PubMed.
- B. Guo, X. Yu, X.-G. Sun, M. Chi, Z.-A. Qiao, J. Liu, Y.-S. Hu, X.-Q. Yang, J. B. Goodenough and S. Dai, Energy Environ. Sci., 2014, 7, 2220–2226 CAS.
- G. Li, X. Wang and X. Ma, J. Energy Chem., 2013, 22, 357–362 CrossRef CAS.
- A. Le Viet, M. Reddy, R. Jose, B. Chowdari and S. Ramakrishna, Electrochim. Acta, 2011, 56, 1518–1528 CrossRef CAS.
- M. Reddy, G. Subba Rao and B. Chowdari, Chem. Rev., 2013, 113, 5364–5457 CrossRef CAS PubMed.
- I. Nowak and M. Ziolek, Chem. Rev., 1999, 99, 3603–3624 CrossRef CAS PubMed.
- R. Kodama, Y. Terada, I. Nakai, S. Komaba and N. Kumagai, J. Electrochem. Soc., 2006, 153, A583–A588 CrossRef CAS.
- N. Uekawa, T. Kudo, F. Mori, Y. J. Wu and K. Kakegawa, J. Colloid Interface Sci., 2003, 264, 378–384 CrossRef CAS PubMed.
- C. J. Brinker and G. W. Scherer, Sol–gel science: the physics and chemistry of sol–gel processing, Academic Press, 2013 Search PubMed.
- M. Schmitt and M. A. Aegerter, Electrochim. Acta, 2001, 46, 2105–2111 CrossRef CAS.
- N. Özer, M. D. Rubin and C. M. Lampert, Sol. Energy Mater. Sol. Cells, 1996, 40, 285–296 CrossRef.
- S.-Q. Guo, X. Zhang, Z. Zhou, G.-D. Gao and L. Liu, J. Mater. Chem. A, 2014, 2, 9236–9243 CAS.
- E. Uchaker, M. Gu, N. Zhou, Y. Li, C. Wang and G. Cao, Small, 2013, 9, 3880–3886 CrossRef CAS PubMed.
- J. Guo, Q. Liu, C. Wang and M. R. Zachariah, Adv. Funct. Mater., 2012, 22, 803–811 CrossRef CAS.
- B. Xu, D. Qian, Z. Wang and Y. S. Meng, Mater. Sci. Eng., R, 2012, 73, 51–65 CrossRef CAS.
- A. Kraytsberg and Y. Ein-Eli, Adv. Energy Mater., 2012, 2, 922–939 CrossRef CAS.
- E. Uchaker, Y. Z. Zheng, S. Li, S. L. Candelaria, S. Hu and G. Z. Cao, J. Mater. Chem. A, 2014, 2, 18208–18214 CAS.
- V. Augustyn, P. Simon and B. Dunn, Energy Environ. Sci., 2014, 7, 1597–1614 CAS.
- C. T. Cherian, M. Reddy, T. Magdaleno, C.-H. Sow, K. Ramanujachary, G. S. Rao and B. Chowdari, CrystEngComm, 2012, 14, 978–986 RSC.
- Y. Jiang, D. Zhang, Y. Li, T. Yuan, N. Bahlawane, C. Liang, W. Sun, Y. Lu and M. Yan, Nano Energy, 2014, 4, 23–30 CrossRef CAS.
- P. Nithyadharseni, M. Reddy, B. Nalini, M. Kalpana and B. Chowdari, Electrochim. Acta, 2015, 161, 261–268 CrossRef CAS.
- B. Das, M. Reddy, C. Krishnamoorthi, S. Tripathy, R. Mahendiran, G. S. Rao and B. Chowdari, Electrochim. Acta, 2009, 54, 3360–3373 CrossRef CAS.
- S. Hu, F. Yin, E. Uchaker, W. Chen, M. Zhang, J. Zhou, Y. Qi and G. Cao, J. Phys. Chem. C, 2014, 118, 24890–24897 CAS.
- M. Umeda, K. Dokko, Y. Fujita, M. Mohamedi, I. Uchida and J. R. Selman, Electrochim. Acta, 2001, 47, 885–890 CrossRef CAS.
- S. S. Zhang, K. Xu and T. R. Jow, Electrochim. Acta, 2004, 49, 1057–1061 CrossRef CAS.
- X. Rui, N. Ding, J. Liu, C. Li and C. Chen, Electrochim. Acta, 2010, 55, 2384–2390 CrossRef CAS.
- Y.-F. Deng, S.-X. Zhao, Y.-H. Xu and C.-W. Nan, J. Mater. Chem. A, 2014, 2, 18889–18897 CAS.
- Q. Cao, H. P. Zhang, G. J. Wang, Q. Xia, Y. P. Wu and H. Q. Wu, Electrochem. Commun., 2007, 9, 1228–1232 CrossRef CAS.
- H. Song, Y. Liu, C. Zhang, C. Liu and G. Cao, J. Mater. Chem. A, 2015, 3, 3547–3558 CAS.
- G. Cao, Synthesis, Properties and Applications, World Scientific, 2004 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ce02069g |
| This journal is © The Royal Society of Chemistry 2016 |