Open Access Article
Open Access ArticleSimple access to ionic liquids and organic salts containing the phosphaethynolate (PCO−) and Zintl (Sb113−) anions†
Maximilian
Jost
,
Lars H.
Finger
 ,
Jörg
Sundermeyer
* and
Carsten
von Hänisch
*
,
Jörg
Sundermeyer
* and
Carsten
von Hänisch
*
Fachbereich Chemie and Wissenschaftliches Zentrum für Materialwissenschaften, Philipps-Universität Marburg, Hans-Meerwein-Straße 4, 35032 Marburg, Germany. E-mail: haenisch@chemie.uni-marburg.de; jsu@staff.uni-marburg.de
First published on 22nd August 2016
Abstract
Herein we report the synthesis of the first ionic liquids with the highly reactive and synthetically valuable phosphaethynolate (PCO−) anion. They are simply synthesised by the reaction of organic methylcarbonate salts with P(SiMe3)3. The products are obtained in near to quantitative yields and they are highly viscous liquids at room temperature or have low melting points for symmetric cations. Moreover, extending this synthetic strategy towards Sb(SiMe3)3 we discovered a simple synthesis for [P(nBu)3Me]+3[Sb11]3−, the first organic cation salt comprising the Zintl-anion [Sb11]3−.
The first synthesis of the PCO− anion goes back to the early 1990s when Becker et al. isolated the lithium salt bis(1,2-di-methoxyethan-O,O′)lithoxy-methylidinphosphan (Li(OCP)·2 DME) from the reaction of [LiP(SiMe3)2] with CO(OMe)2.1 Until lately this anion has attracted little attention, also due to limitations of its synthetic access. This situation changed upon introduction of new synthetic procedures by Grützmacher and coworkers that allow large scale access to sodium and lithium PCO salts.2 Systematic investigations in recent years have brought up a fascinating diversity of PCO− anion chemistry: like the homologous pseudohalide NCO−, the PCO− anion can act as a nucleophile by binding via the oxygen or phosphorous atom. For example, the P- or O-silylated compounds R3Si–OCP and R3Si–PCO are accessible from the reaction of NaPCO with triorganosilanechlorides or -triflates. In spite of the strong Si–O bond the phosphaketene structure is thermodynamically favoured and prevails as the main product.3,4 Besides its use as a ligand in transition metal5,6 and actinide complexes,7 the PCO− anion can act as a precursor for [2+2] cycloadditions.4,8 The groups of Goicoechea and Grützmacher have developed a diversity of heterocycle forming reactions of the PCO− anion with, e.g., activated alkynes,9 carbodiimides10 and isocyanates.11 Moreover, the PCO− anion allows the synthesis of unsubstituted or N-functionalised inorganic urea analogues,12,13 and acts as a phosphide transfer reagent.14,15
The most common route to the PCO− anion starts from NaPH2, which is converted under CO pressure or with ethylenecarbonate in dimethoxyethane.12 As the number of ionic synthons for PCO− was limited to very few inorganic cations our interest was focused on developing a convenient preparation method for new organic salts containing this anion. It should be mentioned that simple ion exchange in water/methylene chloride or chloroform typically used in ionic liquid syntheses does not lead to this class of reactive organic PCO salts in the pure state. In view of the first synthesis of the PCO− anion, which started from dimethyl carbonate, the class of methylcarbonate ionic liquids (ILs) attracted our attention. The easily accessible organic methylcarbonate salts [Cat]+[OCO2Me]− can be prepared via a water and halide free procedure and have proven as versatile starting materials for ILs.16,17 They were recently employed to synthesise organic salts with hydrochalcogenide and trimethylsilylchalcogenolate anions, starting from H2E and E(SiMe3)2 (E = S, Se, Te), respectively.18,19 In the latter case the methylcarbonate anion acted not as a Brønsted base, which is its typical reactivity pattern, but as a nucleophile and a desilylating agent.20 Our original motivation was to probe similar desilylation reactions at E(SiMe3)3 (E = P, As, Sb) in order to access reactive organic salts with E(SiMe3)2− anions.
Subsequently we discovered a method to synthesise purely organic salts of the composition [Cat]+[PCO]− with varying steric demands of the cation by reacting readily available methylcarbonate precursors with P(SiMe3)3. Many new PCO− salts were obtained in almost quantitative yields from polar solvents like acetonitrile or THF without any need to use autoclaves (Scheme 1). Their formation can be explained by the nucleophilic attack of E(SiMe3)2− anions formed in situ at CO2 formed by the decay of unstable carbonate intermediate Me3SiO–COOMe followed by silyl migration and siloxane elimination.
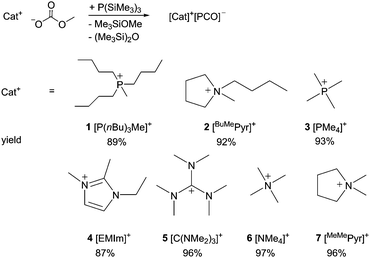 | ||
| Scheme 1 Synthesis of organic phosphaethynolate salts, yields in parentheses (BuMePyr: 1-butyl-1-methylpyrrolidinium, MeMePyr: 1,1-dimethylpyrrolidinium, EMIm: 1-ethyl-2,3-dimethylimidazolium). | ||
The compounds [P(nBu)3Me]+[PCO]− (1) and [EMIm]+[PCO]− (4) were obtained as yellow to orange, very viscous oils and can therefore be regarded as room temperature ILs; all other PCO− salts presented in this work were received as yellow to orange powders. While the salts [BuMePyr]+[PCO]− (2) and [PMe4]+[PCO]− (3) melt reversibly below 100 °C, the compound [NMe4]+[PCO]− (6) decomposes at 215 °C. Symmetric [C(NMe2)3]+[PCO]− (5) decomposes at 230 °C while asymmetric [MeMePyr]+[PCO]− (7) melts reversibly at 125 °C.
The prepared phosphaethynolate salts were investigated by 1H, 13C and 31P NMR spectroscopy. In good agreement with literature reports, the phosphorus atom of the anion displays a resonance between −382.6 ppm and −390.3 ppm21 and 13C{1H} NMR resonance signals are consistent with other published values in the range of 167.7 to 170.0 ppm, which show 1JCP coupling constants consistent with literature known compounds such as [K(18c6)]+[PCO]− with 1JCP = 62 Hz.21
The IR spectra of compounds 1–7 show two characteristic bands in the range from 1757 to 1772 cm−1 and from 1791 to 1799 cm−1 of the PCO− anion, which are in agreement with the spectra of [K(18c6)]+[PCO]− (1730 cm−1) and NaOCP·DME (1760 cm−1, 1780 cm−1).12,21
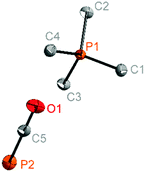
The salts [PMe4]+[PCO]− (3), [C(NMe2)3]+[PCO]− (5) and [NMe4]+[PCO]− (6) could be obtained as single crystals and were investigated by X-ray diffraction. [PMe4]+[PCO]− (3) crystallises in the orthorhombic crystal system in the space group Pnma and includes one ion pair in the asymmetric unit (Fig. 1). The phosphorus–carbon bond length in the anion is 1.579(1) Å and is thus in good agreement with the bond length in [K(18c6)]+[PCO]− (1.579(3) Å). Also the carbon–oxygen bond length in the anion ((C5–O1): 1.213(1) Å) is similar to the value described by Goicoechea (d(C–O): 1.212(4) Å).21
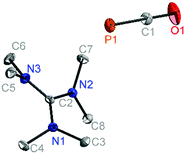
[C(NMe2)3]+[PCO]− (5, Fig. 2) crystallises in the monoclinic crystal system in the space group P21/c with two independent ion pairs and one equivalent of acetonitrile in the asymmetric unit. In comparison with 3, the P–C bond of the PCO− anion is elongated, while the C–O bond is shortened. A P–C–O angle of 179.9° suggests an almost linear geometry around the central carbon atom in the anion and is in good agreement with the P–C–O angle in [K(18c6)]+[PCO]− (178.9°).
[NMe4]+[PCO]− (6) crystallises in the monoclinic crystal system in the space group P21/m. The P–C and C–O bond lengths are 1.601(1) Å and 1.203(2) Å, respectively, and within the typical range of the PCO− anion. The P–C–O angle is 179.2° and shows the expected nearly linear geometry.
Besides the reaction of methylcarbonate ILs with P(SiMe3)3 we investigated their behaviour towards the heavier homologue Sb(SiMe3)3 in order to synthesise a compound of an unknown SbCO− anion. Its existence and stability at ambient temperature was postulated by Schaefer and co-workers on the basis of DFT calculations.22
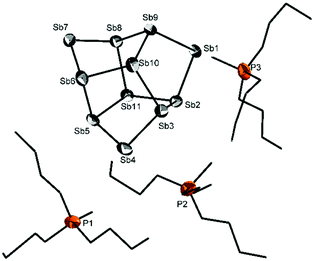
Similar to the previously mentioned procedure, [P(nBu)3Me]+[OCO2Me]− was dissolved in DME and Sb(SiMe3)3 was slowly added at 0 °C. The reaction mixture turned dark brown. Obviously a different competitive reaction path of desilylated anionic antimony species is involved. After filtration of some solid material, black plates crystallized from solution at room temperature. The XRD analysis revealed an antimony cluster of the composition [P(nBu)3Me]+3[Sb11]3− (8). We did not find any indication of the formation of analogues of a salt with the SbCO− anion. 8 crystallises in the monoclinic crystal system in the space group Cc. Fig. 3 displays the [Sb11]3− Zintl anion crystallised with three tri-n-butylmethyl-phosphonium cations. The [Sb11]3− anion and all lighter congeners have been well known in the literature for years; the [Bi11]3− anion was obtained by Dehnen and co-workers only recently.23–25 While the counterions are usually alkali metal cations, which are coordinated by crown ethers or cryptands, quaternary phosphonium counterions, the most base/nucleophile-robust among the organic cations, have been used to stabilize only the [P11]3− anion, so far.26,27
The anion consists of three Sb− (pseudo-tellurium) atoms, which are twofold bonded. The bond length between the twofold and threefold bonded antimony atoms varies from 2.752(2) to 2.766(1) Å (respectively from 2.811(1) to 2.841(1) Å for the threefold bonded antimony atoms) and matches with known [Sb11]3− clusters ([Na(2,2,2-crypt)]+3[Sb11]3−; 2.757 Å and 2.827 Å).23
The crystals are stable for several months at room temperature in the mother liquid but decompose very quickly if they are removed from the solvent. The investigation of 8 by NMR spectroscopy showed partial decomposition of the cation and as a consequence decomposition of the cluster anion by deprotonation of the methyl group, resulting in the formation of a phosphorous ylide, which shows a singlet resonance at 20.48 ppm in the 31P{1H} NMR spectra.
We have shown that the reaction between organic methylcarbonate salts and P(SiMe3)3 leads to new ionic liquids and reactive synthons of the type [Cat]+[PCO]−. Our method is comparably benign with respect to hazards. In contrast to already known procedures, no autoclaves or high reaction temperatures nor the handling of carbon monoxide and PH3 gases is needed to form these valuable PCO− synthons. The latter stand out due to excellent yields, good solubility and a simple preparative procedure. The new salts comprising an uncoordinated, highly nucleophilic PCO− anion will serve as transfer agents of this ligand to transition metal complexes or main group compounds, for the formation of crystalline metallate complexes or as stable and nicely soluble synthons in heterocyclic chemistry. Furthermore, we obtained the first organic cation salt of the Zintl ion [Sb11]3−. In future we will focus on similar reactions with As(SiMe3)3 and Bi(SiMe3)3 to establish the scope and limits of ECO− (E = As, P, As, Sb, Bi) versus Zintl anion reaction paths.
LHF and JS thank the VCI (FCI grant for LHF) and the DFG (GRK 1782) for financial support. Part of this work was funded by BMBF Project TBAfuerPV.
Notes and References
- G. Becker, W. Schwarz, N. Seidler and M. Westerhausen, Z. Anorg. Allg. Chem., 1992, 612, 72–82 CrossRef CAS.
- D. Heift, Z. Benkő and H. Grützmacher, Dalton Trans., 2014, 43, 831–840 RSC.
- D. Heift, PhD thesis, ETH Zürich, 2013.
- D. Heift, Z. Benkő and H. Grützmacher, Dalton Trans., 2014, 43, 5920–5928 RSC.
- (a) A. R. Jupp, M. B. Geeson, J. E. McGrady and J. M. Goicoechea, Eur. J. Inorg. Chem., 2016, 639–648 CrossRef CAS PubMed; (b) L. Liu, D. A. Ruiz, F. Dahcheh, G. Bertrand, R. Suter, A. M. Tondreau and H. Grützmacher, Chem. Sci., 2016, 7, 2335–2341 RSC.
- L. Weber, G. Bassmann and H. Stammler, Organometallics, 1996, 15, 128–132 CrossRef CAS.
- C. Camp, N. Settineri, J. Lefèvre, A. R. Jupp, J. M. Goicoechea, L. Maron and J. Arnold, Chem. Sci., 2015, 2, 6379–6384 RSC.
- L. Liu, J. Zhu and Y. Zhao, Chem. Commun., 2014, 50, 11347–11349 RSC.
- X. Chen, S. Alidori, F. F. Puschmann, G. Santiso-Quinones, Z. Benkő, Z. Li, G. Becker, H.-F. Grützmacher and H. Grützmacher, Angew. Chem., Int. Ed., 2014, 53, 1641–1645 CrossRef CAS PubMed.
- D. Heift, Z. Benkő and H. Grützmacher, Angew. Chem., Int. Ed., 2014, 53, 6757–6761 CrossRef CAS PubMed.
- D. Heift, Z. Benkő, H. Grützmacher, A. R. Jupp and J. M. Goicoechea, Chem. Sci., 2015, 6, 4017–4024 RSC.
- F. F. Puschmann, D. Stein, D. Heift, C. Hendriksen, Z. a. Gal, H. F. Grützmacher and H. Grützmacher, Angew. Chem., Int. Ed., 2011, 50, 8420–8423 CrossRef CAS PubMed.
- A. R. Jupp, G. Trott, É. Payen de la Garanderie, J. D. G. Holl, D. Carmichael and J. M. Goicoechea, Chem. – Eur. J., 2015, 21, 8015–8018 CrossRef CAS PubMed.
- (a) A. M. Tondreau, Z. Benkö, J. R. Harmer and H. Grützmacher, Chem. Sci., 2014, 5, 1545–1554 RSC; (b) L. Liu, D. A. Ruiz, D. Munz and G. Bertrand, Chem, 2016, 1, 147–153 CrossRef.
- T. P. Robinson, M. J. Cowley, D. Scheschkewitz and J. M. Goicoechea, Angew. Chem., Int. Ed., 2015, 54, 683–686 CrossRef CAS PubMed.
- L. Rehmann, E. Ivanova, J. L. Ferguson, H. Q. N. Gunaratne, K. R. Seddon and G. M. Stephens, Green Chem., 2012, 14, 725–733 RSC.
- J. D. Holbrey, R. D. Rogers, S. S. Shukla and C. D. Wilfred, Green Chem., 2010, 12, 407–413 RSC.
- L. H. Finger, F. Wohde, E. I. Grigoryev, A.-K. Hansmann, R. Berger, B. Roling and J. Sundermeyer, Chem. Commun., 2015, 51, 16169–16172 RSC.
- L. H. Finger and J. Sundermeyer, Chem. – Eur. J., 2016, 22, 4218–4230 CrossRef CAS PubMed.
- L. H. Finger, B. Scheibe and J. Sundermeyer, Inorg. Chem., 2015, 54, 9568–9575 CrossRef CAS PubMed.
- A. R. Jupp and J. M. Goicoechea, Angew. Chem., Int. Ed., 2013, 52, 10064–10067 CrossRef CAS PubMed.
- Y. Lu, H. Wang, Y. Xie, H. Liu and H. F. Schaefer, Inorg. Chem., 2014, 53, 6252–6256 CrossRef CAS PubMed.
- T. Hanauer and N. Korber, Z. Anorg. Allg. Chem., 2006, 632, 1135–1140 CrossRef CAS.
- F. Kraus and N. Korber, Chem. – Eur. J., 2005, 11, 5945–5959 CrossRef CAS PubMed.
- B. Weinert, A. R. Eulenstein, R. Ababei and S. Dehnen, Angew. Chem., Int. Ed., 2014, 53, 4704–4708 CrossRef CAS PubMed.
- N. Korber and H. G. von Schnering, Chem. Ber., 1996, 129, 155–159 CrossRef CAS.
- N. Korber, J. Daniels and H. G. Von Schnering, Angew. Chem., Int. Ed. Engl., 1996, 35, 1107–1110 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental and crystallographic details. CCDC 1450250–1450252 and 1454441. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c6cc06620h |
| This journal is © The Royal Society of Chemistry 2016 |