A new optical sensor for selective quantitation of uranium by the immobilization of arsenazo III on an agarose membrane
Abstract
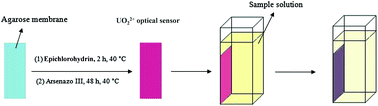
A highly selective optical sensor was introduced for the determination of UO22+ through the chemical immobilization of arsenazo III on a transparent agarose membrane. Spectrophotometric studies of complex formation between arsenazo III and several metal ions at pH 2 show a significantly larger stability constant for the uranyl ion complex compared to the others. Consequently, arsenazo III was used as an appropriate ionophore for the preparation of a selective UO22+ optical sensor, through its chemical immobilization on a transparent agarose film. The immobilization conditions were optimized and the prepared optical sensor showed a distinct color change from purple to dark violet upon increasing the analyte concentration in a buffered solution at pH 2. The effects of pH, the ionophore concentration, the ionic strength and the reaction time on the response of the sensor were carefully studied. The experimental points were best fitted to two distinct lines within the concentration ranges of 3.8 × 10−7 to 7.4 × 10−5 mol L−1 and 7.4 × 10−5 to 2.1 × 10−3 mol L−1 with R2 values of 0.982 and 0.994, respectively. No significant interference from 100 times the concentration of a number of potentially interfering ions was detected during uranyl ion determination. The optical sensor was successfully applied to the determination of UO22+ in spiked water samples.

 Please wait while we load your content...
Please wait while we load your content...