Development of a protease-resistant reporter to quantify BCR–ABL activity in intact cells†
Abstract
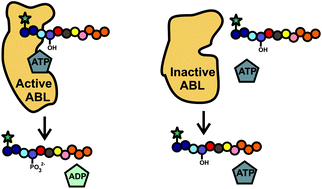
A peptidase-resistant ABL kinase substrate was developed by identifying protease-susceptible bonds on an ABL substrate peptide and replacing flanking amino acids with non-native amino acids. After an iterative design process, the lead, or designed, peptide X-A possesses a six-fold longer life in a cytosolic lysate than that of the starting peptide. The catalytic efficiency (kcat/KM) of purified ABL kinase for the lead peptide (125 s−1 μM−1) is similar to that of the starting peptide (103 s−1 μM−1) demonstrating preservation of the peptide's ability to serve as a kinase substrate. When incubated in cytosolic lysates, the lead peptide is slowly degraded into 4 fragments over time. In contrast, when loaded into intact cells, the peptide is metabolized into 5 fragments, with only 2 of the fragments corresponding to those in the lysate. Thus the two environments possess differing peptidase activities, which must be accounted for when designing peptidase-resistant peptides. In both settings, the substrate is phosphorylated by BCR–ABL providing a readout of BCR–ABL activity. A small panel of tyrosine kinase inhibitors verified the substrate's specificity for BCR–ABL/ABL kinase activity in both lysates and cells in spite of the multitude of other kinases present. The designed peptide X-A acts as a long-lived BCR–ABL kinase reporter in the leukemic cells possessing the BCR–ABL mutation.
- This article is part of the themed collection: In memory of Craig Lunte

 Please wait while we load your content...
Please wait while we load your content...