Open Access Article
Open Access ArticleDevelopment and use of in vitro alternatives to animal testing by the pharmaceutical industry 1980–2013
Jen-Yin
Goh
a,
Richard J.
Weaver
b,
Libby
Dixon
a,
Nicola J.
Platt
a and
Ruth A.
Roberts†
*c
aThe Association of the British Pharmaceutical Industry (ABPI), 7th Floor, Southside, 105 Victoria Street, London, SW1E 6QT, UK
bInstitut de Recherches Internationales Servier (IRIS), rue Carnot, 92150 SURESNES, France
cAstraZeneca, Alderley Park, SK10 4TG, UK. E-mail: ruth.roberts@apconix.com; Tel: +44 (0)7733 014396
First published on 30th June 2015
Abstract
We examined the use of in vitro (including in silico) techniques in preclinical safety testing by the pharmaceutical industry between 1980 and 2013 to determine patterns, drivers and challenges in uptake. Data were collected via a survey sent to the Association of the British Pharmaceutical Industry (ABPI) member companies from the Nonclinical and Biological Discovery Expert Network (NaBDEN) requesting the number of compounds screened using in vitro and in silico tests at 5-year intervals between 1980 and 2005 then yearly from 2008 onwards. A utility score from 1 (poor) to 5 (excellent) for each assay was also requested. Four pharmaceutical companies and 3 contract research organisations (CROs) responded to the survey, providing >895![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 data points across all years and all assays. Overall, there was a steady increase in the use of in vitro tests by the pharmaceutical industry between 1980 and 2013; indeed >20% of all in vitro tests reported were conducted in the last year of the survey window (2013) and >70% of all in vitro tests reported were conducted since 2010. Use of in vitro tests peaked at >190
000 data points across all years and all assays. Overall, there was a steady increase in the use of in vitro tests by the pharmaceutical industry between 1980 and 2013; indeed >20% of all in vitro tests reported were conducted in the last year of the survey window (2013) and >70% of all in vitro tests reported were conducted since 2010. Use of in vitro tests peaked at >190![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tests per annum in 2012; >99% of this usage was in the three main areas reported of ADME, safety pharmacology and genotoxicity. Trends and step changes in uptake were most notable in the three main areas of ADME, safety pharmacology and genotoxicity and may be explained by the timing of adoption of the relevant International Committee on Harmonisation (ICH) guidelines. Trends in uptake may also be explained by perceptions of utility where scores varied from poor (Eye Irritation – flourescein leakage) to excellent (Genotoxicity – Ames and Skin irritation – EpiSkin/Epiderm). In summary, the data show a large increase and a continuing upwards trend in development and adoption of in vitro alternatives to animal testing in pharmaceutical drug development providing new opportunities to improve success rates coupled with a strong commitment to the 3Rs.
000 tests per annum in 2012; >99% of this usage was in the three main areas reported of ADME, safety pharmacology and genotoxicity. Trends and step changes in uptake were most notable in the three main areas of ADME, safety pharmacology and genotoxicity and may be explained by the timing of adoption of the relevant International Committee on Harmonisation (ICH) guidelines. Trends in uptake may also be explained by perceptions of utility where scores varied from poor (Eye Irritation – flourescein leakage) to excellent (Genotoxicity – Ames and Skin irritation – EpiSkin/Epiderm). In summary, the data show a large increase and a continuing upwards trend in development and adoption of in vitro alternatives to animal testing in pharmaceutical drug development providing new opportunities to improve success rates coupled with a strong commitment to the 3Rs.
Introduction
Preclinical safety testing of new drug candidates is a crucial step in pharmaceutical drug development and depends on a sequential series of in silico, in vitro and in vivo tests before administration to humans. Currently, in vivo testing is a vital part of safety assessment, and is a regulatory requirement before a drug can progress into clinical trials.1,2 However, in recent years, many in vitro assays have been developed and validated for early stage screening aimed at filtering out molecules with a higher potential for toxicity and in some cases replacing or reducing the use of certain in vivo tests.The pharmaceutical industry's interest in developing new in vitro assays has arisen from the need to support the early identification of promising drug candidates but also through legislation requiring adherence to the 3Rs, a set of principles that outlines the replacement, reduction and refinement of the use of animals in research.3
These 3Rs have long been embedded in the UK Animals (Scientific Procedures) Act (ASPA) 1986,4 recently revised to transpose European Directive 2010/63/EU5 into new legislation. The European Medicine Agency's (EMA) paper on replacement of animal studies by in vitro models6 provides information on the conditions and strategy for regulatory acceptance of 3R alternative methods. Additionally, since its establishment in 2004, the UK National Centre for the 3Rs (NC3Rs) has played an important role in promoting awareness of the 3Rs and in leading and driving the discovery, use and commercialisation of new non-animal technologies and alternative techniques.7 The UK government has also demonstrated its support for the 3Rs, committing in 2010 to work towards reducing the use of animals in research, and recently publishing a Delivery Plan8 that details current and future initiatives to reduce the use of animals in research. Additionally, organisations that support regulatory validation of alternatives methods have been established, such as the European Union Reference Laboratory for Alternatives to Animal Testing (EURL ECVAM). This has resulted in validation of a number of in vitro assays such as the bovine corneal opacity and permeability (BCOP) test and Cytosensor microphysiometer test that are used in eye irritation testing9 and are now part of international regulatory guidelines. Finally, new technological and scientific advances, such as powerful computational models, and ‘omics’ technologies, have facilitated the development of many new in vitro assays.
The pharmaceutical industry has shown a strong commitment to the 3Rs principles,10–13 as demonstrated by their strong links with the NC3Rs5 and their contributions to the Concordat on Openness on Animal Research in the UK.11 The industry is increasingly working to replace, reduce and refine the use of animals in drug development and especially in toxicology testing;12–14 legislative and regulatory changes, coupled with technological and scientific developments have provided opportunities to support the adoption of in vitro and in silico alternatives to in vivo testing. Here, we have examined how the use of in vitro techniques within the pharmaceutical industry has evolved from 1980 to 2013 and report on where the uptake of in vitro and in silico techniques have been most notable both within pharmaceutical companies and CROs.
Methods
Collection of data
Excel spreadsheet was designed and developed by the Association of The British Pharmaceutical Industry (ABPI) Nonclinical and Biological Discovery Expert Network Group (NaBDEN)15 to capture information on the historical use of in vitro assays (including in silico assays) in pharmaceutical R&D. The spreadsheet listed in vitro and in silico assays within the field of preclinical safety testing (Table 1) and provided fields to capture number of compounds put through each of the assays at 5-year intervals between 1980 and 2005 and each year from 2008 onwards. For each assay, the following information was requested: (i) the number of compounds screened per year using the assay and (ii) a utility score of 0–5 based on the percieved value of the assay to the contributing company (Table 2). In addition to providing data requested, participants were invited to provide additional examples of in vitro assays used in their organisations.| Discipline/field | Test/study type |
|---|---|
| DEREK: Deductive estimation of risk from existing knowledge; MNU: Micronucleus assay; REACH: Registration, evaluation, authorisation and restriction of chemicals; BCOP: Bovine corneal opacity and permeability; HET CAM: Hen's Egg test-chorioallantoic membrane; ECVAM: European centre for the validation of alternative methods; FP6: Framework programme 6; FP7: Framework programme 7; EURL: European Union Reference Laboratory; SAR: Structure activity relationship; ADRs: Adverse drug reactions; PK: Pharmacokinetics; NRU: Neutral red uptake. | |
| Genotoxicity | COM cell transfection assays |
| DEREK + other in silico projects | |
| Ames II, MNU, Greenscreen and Blue screen assays | |
| Impurity testing in silico for REACH | |
| Mouse lymphoma assay | |
| In vitro micronucleus test | |
| Ames | |
| In vitro chromosome aberration test | |
| In silico Ames prediction GWS | |
| SOS UMU | |
| Safety pharmacology | Electrophysiology testing |
| Cardiovascular | |
| Human recombinant activity | |
| In silico prediction for off target panel screen | |
| Radioligand binding & enzyme – off target panel screen | |
| Safety screen – cellular/functional | |
| Skin irritation | Irritation (OECD 439) |
| EpiSkin (VRM) | |
| SkinEthic RHE | |
| EpiDerm SIT | |
| Skin corrosion (OECD 430/431/435 | OCED 431 In Vitro Skin Corrosion (EpiDerm) |
| Dermal absorption | Skin absorption: in vitro/radio-analysis/LC-MS/MS |
| Eye irritation | BCOP |
| Isolated chick eye | |
| Cytosensor microphysiometer | |
| Fluorescein leakage | |
| HET-CAM (ECVAM Validation) | |
| Human corneal epithelium | |
| Development/reprotox/endocrine | Differential gene expression, mechanisms of action |
| FP6 and FP7 repro and neurotoxicity | |
| ReproTECT | |
| Endocrine disruptors – battery testing in vitro | |
| Carcinogenicity Studies | FDA/Regulators encouraging use of bioassays |
| CarcinoGenomics FP6 | |
| EURL ECVAM recommendation | |
| Immunotoxicity | Human and mouse artificial lymph nodes |
| ADME | SAR before in vitro testing and in vivo testing for metabolic stability |
| CaCO2 efflux assays and transporter assays for prediction of ADRs | |
| PK properties (assay below) | |
| - CYP inhibition/induction | |
| - Protein binding in vitro | |
| In vitro metabolic stability | |
| Mechanistic studies | |
| In vitro reactive metabolites | |
| LINK programme – CYP expression | |
| Microfluidics/culex | |
| PD samples quantification (plasma, brain, tumours) | |
| Phototoxicity | 3T3 NRU |
| Score | Reason |
|---|---|
| 0 | Do not believe in assay |
| 0 | Did not exist at this time |
| 0 | Not engaged in R&D requiring this |
| 0 | Not valued/does not work |
| 1 | Not predictive |
| 2 | Not very predictive |
| 3 | Reasonable |
| 4 | Good but not exhaustive |
| 5 | Excellent |
The survey was distributed to ABPI NaBDEN member companies with guidance together with a guarantee of anonimity for participating companies.
Data handling
Completed surveys were anonimised and collated by the number of reported tests by year and utility scores values.Data returned on some tests were excluded from analysis, either because they are not in vitro tests or because data from both in vivo/in vitro tests were combined and could not be separated in a meaningful way. These are listed in Table 3.
| Discipline/field | Tests/study/project type |
|---|---|
| Safety pharmacology | Zebrafish |
| Developmental/reproductive/endocrinology toxicology | Fingerprint biomarkers (inhibin B) |
| Carcinogenicity | Transgenic onco mouse |
| ADME | In vivo bioavailability |
| In vivo absorption | |
| In vivo reactive metabolites | |
| Metabolic identification (in vivo/in vitro) |
Where respondents provided data in absolute values, e.g. 7, 30, 699, 78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 098 these were used in the analysis. Where respondents indicated a range (as given at the column head in the collection spreadsheet), midpoints of the ranges were used to provide a numerical figure for the analysis. For assays where more than 10
098 these were used in the analysis. Where respondents indicated a range (as given at the column head in the collection spreadsheet), midpoints of the ranges were used to provide a numerical figure for the analysis. For assays where more than 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 compounds were tested, a higher end value of a range could not be determined and thus, the lower end value of ‘10
000 compounds were tested, a higher end value of a range could not be determined and thus, the lower end value of ‘10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000’ was used (Table 4).
000’ was used (Table 4).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 times, the ‘midpoint’ of 10
000 times, the ‘midpoint’ of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 was used
000 was used
| Range | Midpoint/lower end value |
|---|---|
| 1–10 | 6 |
| 11–50 | 31 |
| 51–250 | 151 |
| 251–500 | 376 |
| 500–1000 | 750 |
| 1000–5000 | 3000 |
5000–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
7500 |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000+ 000+ |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
Where companies provided data that did not cover all of the 1980–2013 window, exclusion was considered to prevent any spurious impact on potential trends (see results for details). In these cases, the utility scores and any comments given were included in the qualitative analyses.
Utility scores of ‘0’ with accompanying reasons of ‘did not exist at this time’, ‘not engaged in R&D requiring this’ and ‘do not believe in assay’ were omitted during data analysis as these options do not reflect the utility of the tests (but provided useful context to the data during analysis).
Analysis of data
The relative use of in vitro assays in different fields was evaluated by comparing the total number of tests being carried out in each field with the overall total tests being carried out. In addition, comparisons were made between overall use and fields of use of in vitro tests in pharmaceutical companies and CROs from 1980 to 2013.
Historial trends in overall use of in vitro tests between CRO were revealed by normalising data for CROs and for Pharma. This allowed trends to be viewed irrespective of the large variation in the total number of tests performed by these two groups. To do this, the total number of all in vitro tests carried out across all years (1980–2013), was calculated for CROs and for Pharma; the total number of in vitro tests carried out in each individual year was then divided by the total number of tests carried out by CRO or by Pharma across all years and multiplied by 100 to give the percentage of tests carried out in each year.
Utility scores
Utility scores were combined for each contributor to give an average for each test. Where no utility scores were recorded they were excluded from the final data analysis.Results
Responses
Responses from seven companies (four pharmaceutical companies and three contract research organisations (CROs)) were received in response to the ABPI survey. Some companies submitted data for multiple sites, including global locations, whereas others submitted data from UK sites only. Four of the seven respondents provided data in absolute values wheares three of the companies supplied data in ranges.Five of the seven companies provided data across the full period of 1980 to 2013 although there were relatively low numbers of in vitro tests reported before 2005. One company provided data from 2005 onwards only – these data were included in the historical trends analysis since missing data from before 2005 for this one company had a minimal effect on the analysis of historical trends against a background of low numbers of tests reported up to 2005 for the other five companies. In contrast, one company provided data only from 2009 to 2013; responses from this company were not included in the analysis of historical use of in vitro tests since data from other companies showed that many of the tests were in use before 2009 and inclusion of the 2009–2013 data from this one company would result in a larger apparent increase from 2008 to 2009, thereby skewing the results. Although omitted from the historical trends analysis, data from this same company were used in analysis of breakdown of in vitro tests according to fields/disciplines where trends were not analysed historically. The scores and comments from this one company were also used in qualitative analysis of data and to support interpretation and discussion of the overall trends.
Trend of use of in vitro tests
Fig. 1 displays the overall trend of use of in vitro tests between 1980 and 2013. Raw data (1A) showed a general year on year increase including a large step up from 2000 to 2005 but with a small downturn in 2011 and 2013. However, normalised data (1B) showing trends across all companies irrespective of the total number of tests carried out by each company showed a year by year increase and confirmed a strong upward trend in the use of in vitro tests between years 2000 and 2005.Breakdown of in vitro tests used by the pharmaceutical industry by fields/disciplines
Fig. 2 illustrates that in vitro tests in the fields of absorption, distribution, metabolism and excretion (ADME), safety pharmacology and genotoxicity account for 99.9% of the test carried out across the time period 1980–2013. Only a small proportion (0.1%) of the in vitro tests that were carried out was from other areas. A focused breakdown of this 0.1% revealed tests for dermal absorption, skin irritation, eye irritation and skin corrosion accounting for the majority with a few inhalation, endocrine disruption, development/reprotoxicology and phototoxicity tests also being reported. There was no reported use of in vitro techniques in the fields of carcinogenicity and immunotoxicology.Comparisons between use of in vitro tests by pharmaceutical companies and CROs
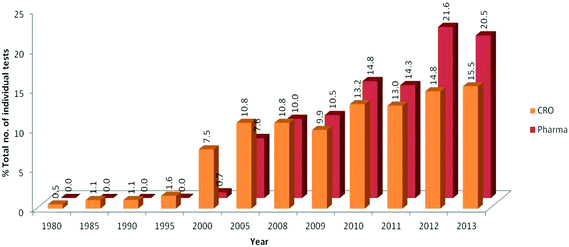
Fig. 3A shows the overall trends of use of in vitro tests by pharmaceutical companies (top) and CROs (bottom) from 1980 to 2013. Overall, a large and steady increase in the use of in vitro tests was observed for both pharmaceutical companies and CROs since 2000. Of note was the large difference in total numbers of compounds between pharmaceutical companies and CROs with pharmaceutical companies peaking at >180![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 and CROs peaking at >500 tests per annum. In addition, the use of in vitro tests in pharmaceutical companies showed a slight drop in the use of in vitro tests between 2012 and 2013 in contrast to a marked increase in use for CROs in the same time period.
000 and CROs peaking at >500 tests per annum. In addition, the use of in vitro tests in pharmaceutical companies showed a slight drop in the use of in vitro tests between 2012 and 2013 in contrast to a marked increase in use for CROs in the same time period.
Fig. 3B illustrates the use of in vitro tests in pharmaceutical companies and CROs by discipline. The pharmaceutical companies primarily used in vitro assays in three fields: ADME, safety pharmacology and genotoxicity. In contrast, CROs carried out in vitro assays in diverse fields such as eye irritation, dermal absorption and skin irritation. However, in common with data from the pharmaceutical companies, ADME also accounted for a large proportion (46%) of tests carried out by CROs.
Fig. 4 shows a comparison using normalised data between the use of in vitro tests in the three largest disciplines (genotoxicity, safety pharmacology and ADME) by pharmaceutical companies and CROs between 1980 and 2013. CROs reported the earliest use of in vitro tests in these disciplines with 0.5% of the overall CRO usage occurring in 1980 followed by the earliest upturn in use in the period 2000–2008. The use of these tests by pharmaceutical companies was only evident from 2000 but since then has shown a steady increase.
Trend of use: in vitro tests from the three main disciplines
Fig. 5 shows the pattern in historical uptake of tests within the three main disciplines of genotoxicity, safety pharmacology and ADME in the period 1980 to 2013. For genotoxicity, use remained relatively low until 2010 but since then has continued to increase, with a particularly prominent increase in use of tests in this field from 2011–2012 (Fig. 5A). For safety pharmacology, the use of in vitro assays has shown little increase, remaining at similar levels since the initial significant implementation around 2005 (Fig. 5B). For ADME, there was some usage as early as 1980 which remained low but constant until a step change in 2000 followed by a steady increase (Fig. 5C).Trend of use: some examples of individual tests
Fig. 6 shows the historical trends of use for two in vitro genotoxicity tests and one skin absorption test. DEREK and in silico tests in genotoxicity testing was noted in 1995 followed by a large increase to 2005 (Fig. 6A) and a slight increase in the use of these tests since this time. In contrast, since initial use in the mid-1990s, use of Ames II, Bluescreen and related assays (Fig. 6B) has increased but in an apparently inconsistent manner.Fig. 6C shows the historical trends of use for in vitro tests of skin absorption. The usage of these tests has increased year on year since their initial introduction in 2000 except between 2008 and 2009 where usage remained constant.
Utility
Fig. 7 shows the assessment of the perceived utility for each of the individual in vitro tests in the survey. Overall, there was a high level of confidence (4: good but not exhaustive to 5: excellent) in the field of genotoxicity with a few notable exceptions; SOS UMU scored low on utility (2: not very predictive) and in silico REACH scored average (3: reasonable). All tests listed for skin irritation/absorption scored ≥4.9 whereas tests for ADME consistently scored between 3.8 and 4.4. The biggest variability within a discipline was seen in the utility scores for the tests for eye irritation where responses ranged from 1 (not predictive) for flourescein leakage to 4 (good but not exhaustive) for the BCOP.Discussion
In this study, we sought to examine how the use of in vitro techniques in the pharmaceutical industry has changed from 1980 to 2013. We also aimed to identify whether there has been a greater focus on developing in vitro techniques in some fields than others and compared the trends in use of in vitro assays between pharmaceutical companies and the CROs.The data presented show a large and continuing increase in the use of in vitro tests by the pharmaceutical industry. The slight plateau between 2012 and 2013 resolves when data are normalised such that each company's figures act as their own control suggesting the apparent plateau is caused by a downturn in use by one or two big contributors to the survey. Overall, >20% of all in vitro tests reported were conducted in the last year of the survey window (2013) and >70% of all in vitro tests reported were conducted since 2010. This increase is encouraging in the context of the 3Rs, a set of principles that outlines the replacement, reduction and refinement of the use of animals in research.3 Indeed, expenditure on research and development by the pharmaceutical industry has grown by £1 billion since 200216 yet the Home Office figures for animal use in the UK17 by commercial organisations have stayed largely unchanged with around 1–1.5 million procedures a year reported since 1995.17 This increased investment in UK R&D in the absence of an substantial increase in animal use could be attributed at least in part to the increased use of in vitro tests described here. It is however worth noting that certain approaches with high reported use herein such as DEREK offer new technological possibilities rather than replacing existing in vivo assays per se. Nonetheless, such in silico approaches could be viewed as helping to select and prioritise compounds for development with a better profile and hence probability of success.
The comparisons of in vitro work carried out by CROs with that carried out by pharmaceutical companies reveal interesting trends. For example, CROs conducted only 0.25% of the tests reported in the period 1980 to 2013 reflecting their relatively smaller size but nonetheless conducted a much wider range of in vitro tests when compared with pharmaceutical companies. For example, 99.9% of the reported in vitro assays conducted by pharmaceutical companies were in the three fields of ADME, safety pharmacology and genotoxicity possibly reflecting a trend in the industry to focus resources in certain higher throughout areas and outsource to CROs tests carried out less frequently. In contrast, although CROs did conduct work in the 3 areas of ADME, safety pharmacology and genotoxicity, 44% of the in vitro tests at CROs were in diverse fields such as eye irritation, dermal absorption and skin irritation. In contrast, <0.01% of all in vitro tests done by the pharmaceutical companies was from disciplines other than the top three. A further analysis of trends over time in these three main areas (genotoxicity, safety pharmacology and ADME) suggests that CROs were engaged in conducting in vitro tests in one or more of these fields earlier than pharmaceutical companies, with some use reported since the 1980s.
Comparisons of trends over time in the three main areas reported (genotoxicity, safety pharmacology and ADME) suggests differences in patterns of uptake. There was a slow but steady increase in the use of genotoxicity tests since 1980 whereas there was a step change in reported use of both genotoxicity and safety pharmacology assays since 2005. Interestingly, there was a small but steady use of ADME from the start of the survey in 1980 with a steady increase thereafter. Although the reasons behind these uptake trends are likely to be multifactorial, introduction of new and modifications of existing International Committee on Harmonisation (ICH) guidelines may explain some of the data. For example, ICH S2A (Regulatory Genotoxicity Tests for Pharmaceuticals) was finalised in 1995 followed in 1997 by ICH S2B18 which outlines the standard battery for genotoxicity testing and provides recommendations on the evaluation of test results. Together, these two guidelines are likely to explain some uptake in 1995–2000, the small step seen in 2005 and the subsequent steady rise in the use of in silico and in vitro tests for genotoxicity testing for pharmaceuticals. In this context, it's worth noting that ongoing ICH revisions and their adoption often tend to be driven by research into and validation of in vitro alternatives conducted by and published in collaboration between pharmaceutical companies, CROs and academia.
Regarding safety pharmacology, ICH S7A which addresses definition, objectives and scope of safety pharmacology studies for pharmaceuticals was finalised in 2000.18 Only 44 tests were reported for safety pharmacology up to and including 2000 but then >28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tests were reported in 2005 alone with a steady level and slight rise thereafter.
000 tests were reported in 2005 alone with a steady level and slight rise thereafter.
The earlier uptake of tests for ADME could be explained by the earlier adoption of a guideline in this area; S3A (Guidance on Toxicokinetics) reached step 4 (finalization) in 1994;18 the data presented here suggest some lag in uptake with only 12 tests reported in 1990 and in 1995 but then a steady increase to >6300 tests per annum from 2000 onwards. Interestingly both ICH S7A and ICH S3A largely describe in vivo tests rather than in vitro alternatives, but it could be argued that clarity on acceptable biological endpoints can provide a base for developing in vitro alternatives irrespective of the origin of the endpoints.
A focused analysis of two of the clusters of in vitro genotoxicity tests (DEREK/other in silico tests versus Ames II/MNU/Greenscreen/Bluescreen) revealed very different trends in uptake. ‘DEREK/other in silico tests’ showed a rapid uptake between 2000 and 2005 with little further increase; this suggests that these test may already have been used to maximum effect since their introduction, or have not been developed further. Another explanation is the relatively low utility score returned on DEREK with responses between ‘reasonable’ and ‘good’ compared with some of the other tests in the genotoxicity battery that were rated as good or excellent.
Since initial uptake in the mid-1990s, the use of Ames II/MNU/Bluescreen/Greenscreen related assays has increased but inconsistently. The use of MS in testing skin absorption has also increased steadily since its initial introduction around 2000. This is likely to be a reflection of steadily increasing demand coupled with increasing accessibility and an overall high score in utility; there have been incremental improvements in the accessibility and reliability of the technologies required for this assay year by year.
The approach taken in this paper has helped to quantify the uptake of in vitro tests over a period of 33 years. The approach was never intended to be exhaustive since it depends upon companies retrieving historical data; indeed it is likely that many other tests were carried out that could not be accounted for, especially at the earlier time points. Areas such as reprotoxicology also appear to be underrepresented in the data set for similar reasons. Thus the data set most likely under-report the actual number of tests conducted but nevertheless offer encouraging insight on the upward trends in in vitro alternatives to in vivo testing to complement the regulatory requirements for the safety evaluation of candidate drugs before clinical trials.
Overall, the survey and approach used to quantify use of in vitro tests confirm our recent conclusions drawn from published literature13 that the pharmaceutical industry has a strong commitment to the development and uptake of in vitro test methods and has seen significant success in key areas such as genetic toxicology, skin absorption and reproductive toxicology.19 Indeed, many of the on-going in vitro initiatives to seek and formally validate alternative and in vitro tests are focused on pharmaceuticals as illustrated by Chapman et al.19 Developments in validation and regulatory guidance around in vitro techniques have also facilitated uptake and adoption – this is notable in the timing of uptake connected to the introduction of new guidance such as that from ICH. However, continued uptake of in vitro alternatives depends on reliable and relevant models and there is still much to achieve in this area. This study has highlighted the need for further investment in the development of in vitro tests in particular fields, such as immunotoxicity, as well as a need to continue refining assays that are currently used. The data suggest that implementation can also be a challenge; some tests with high scores on utility took many years to implement or are only used in a few companies. Many of the in vitro assays that have been developed in areas such as genetic toxicology and electrophysiology score low on utility – this could be due to a high level of false positive results which makes extrapolation to the human situation difficult. In addition, the reliability of in silico testing to predict safety signals remains in its infancy and has even been called into question in a recent paper from Cook et al. (2014).20
Many of the tests reported in this study reduce and refine animal use by allowing early high throughput screening of compounds, reducing the number of ineffective or unsafe compounds progressing to in vivo studies. On the other hand, others are direct alternatives to in vivo methods. One example is the EpiSkin test that uses reconstructed human epidermis as a replacement for the rabbit skin irritation tests, and has been validated and endorsed by organizations including ECVAM.21 The first use of the EpiSkin alternative reported in this study was in 2008 with continued increase in use from 2008–2013, reflecting its validation by ECVAM in 2007. Another encouraging example is the uptake by industry since 2008 of the Bovine Corneal Opacity and Permeability (BCOP) test, an in vitro test developed as an alternative to the rabbit Draize eye irritation test. The BCOP assay uses excised animal tissue to replace the in vivo studies, and when used in combination with other in vitro tests, could fully replace the use of an animal model in the future.22
In summary, the data show a large increase and a continuing upwards trend in development and adoption of in vitro alternatives to animal testing in pharmaceutical drug development providing new opportunities to improve success rates coupled with a strong commitment to the 3Rs. However despite the encouraging trend there is still much to be done; there is a pressing need to improve success rates in the pharmaceutical industry and also to make failure less costly perhaps via the development and validation of further in silico and in vitro laboratory tests that could address the main reasons for failure: unexpected toxicity and/or lack of efficacy.20 Collaboration across industry, CROs, academia and government23 will be key to future success via identifying and exploiting the best knowledge and expertise.
References
- ICH Guideline M3(R2), Guidance on Nonclinical Safety Studies for the Conduct of Human Clinical Trials and Marketing Authorization for Pharmaceuticals, 2009, http://www.ich.org/products/guidelines/multidisciplinary/article/multidisciplinary-guidelines.html.
- ICH Guideline S9, Nonclinical Evaluation for Anticancer Pharmaceuticals, 2009, http://www.ich.org/products/guidelines/safety/article/safety-guidelines.html.
- W. M. S. Russell and R. L. Burch, The Principles of Humane Experimental Technique, Methuen, London, 1959 Search PubMed.
- Animals (Scientific Procedures) Act 1986.
- EU Directive 2010 on the protection of animals used for scientific purposes. Directive 2010/63/EU.
- EMA Revised Concept paper on the need for revision of the position on the replacement of animal studies by in vitro models (CPMP/SWP/728/95) CPMP/SWP/728/95), 2012, http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2012/07/WC500130365.pdf.
- NC3Rs, 2014, http://www.nc3rs.org.uk/about-us [accessed 18th October 2014].
- Home Office, Department for Business Innovation & Skills and Department of Health, Working to reduce the use of animals in scientific research, 2014, https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/277942/bis-14-589-working-to-reduce-the-use-of_animals-in-research.pdf [accessed 18th October 2014].
- S. Ward, Applicability domain of BCOP and ICE extended to identifying ocular non-irritants, 2013, http://alttox.org/applicability-domain-of-bcop-and-ice-extended-to-identifying-ocular-non-irritants-2/ [accessed 18th October 2014].
- NC3Rs, Working with the pharmaceutical industry: pioneering better science 2004–2014, NC3Rs, London, 2014, pp. 1–31 Search PubMed.
- Understanding Animal Research, Concordat on openness on animal research in the UK, Understanding Animal Research, London, 2014, pp. 1–10 Search PubMed.
- EFPIA, Putting animal welfare principles and 3Rs into action: European pharmaceutical industry 2011 report, EFPIA, Brussels, 2012, pp. 1–21 Search PubMed.
- EFPIA, Putting animal welfare principles and 3Rs into action: European pharmaceutical industry 2012 update, EFPIA, Brussels, 2013, pp. 1–15 Search PubMed.
- S. Cunningham, N. Partridge and R. Roberts, Quantifying the pharmaceutical industry's contribution to published 3Rs research 2002–2012. Manuscript draft, Elsevier Editorial System (tm) for Toxicology Reports, 2014.
- ABPI Expert Networks, http://www.abpi.org.uk/about-us/how-we-work/Pages/working-members.aspx.
- ABPI, UK Pharmaceutical R&D Expenditure 2000–2012, 2012. http://www.abpi.org.uk/industry-info/knowledge-hub/randd/Pages/expenditure.aspx#6.
- Home Office, Statistics of scientific procedures on living animals, Great Britain 2013, 2013, https://www.gov.uk/government/statistics/statistics-of-scientific-procedures-on-living-animals-great-britain-2013.
- ICH, 2014http://www.ich.org/products/guidelines/safety/article/safety-guidelines.html.
- K. Chapman, H. Holzgrefe, L. Black, M. Brown, G. Chellman, C. Copeman, J. Couch, S. Creton, S. Gehen, A. Hoberman, L. Kinter, S. Madden, C. Mattis, H. Stemple and S. Wilson, Reg. Tox. Pharm., 2013, 66, 88–103 CrossRef CAS PubMed.
- D. Cook, D. Brown, R. Alexander, R. March, P. Morgan, G. Satherwaite and M. Panagalos, Nat. Rev. Drug Discovery, 2014, 13, 419–431 CrossRef CAS PubMed.
- H. Spielmann, et al. The ECVAM international validation study on in vitro tests for acute skin irritation: report on the validity of the EPISKIN and EpiDerm assays and on the Skin Integrity Function Test, Altern. Lab. Anim., 2007, 35, 559–601 CAS.
- OECD, 2009. OECD Guidelines for Testing of Chemicals; Test Guideline 437; Bovine Corneal Opacity and Permeability Test Method for Identifying Ocular Corrosives and Severe Irritants.
- J. Hunter, Drug Discovery World, Spring, 2014, 9–15 Search PubMed.
Footnote |
| † Now at ApconiX, BioHub at Alderley Park, SK10 4TG. |
| This journal is © The Royal Society of Chemistry 2015 |