A copper acetate/2-aminobenzenthiol complex supported on magnetite/silica nanoparticles as a highly active and recyclable catalyst for 1,2,3-triazole synthesis†
Abstract
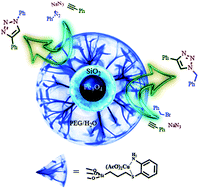
A novel and recoverable Cu(II)/2-aminobenzenthiol complex immobilized on magnetite/silica nanoparticles (Fe3O4/SiO2 NPs) catalyzed the synthesis of 1,2,3-triazole derivatives via three-component condensation of terminal alkynes, benzyl or alkyl halides, and sodium azide in wet polyethylene glycol (PEG 400/H2O) as a green medium at ambient temperature. The catalytic potentials of some transition metal salt complexes adhered on Fe3O4/SiO2 NPs (5–10 nm) were employed for the one pot 1,2,3-triazole synthesis. The order of catalytic activity of the various transition metal salt complexes for a model reaction was found to be Cu(II) > AgI ∼ CuI > CuCl > Ni(OAc)2 ∼ Co(OAc)2 > ZnCl2. Moreover, the order of catalytic activity of Cu(II) salts supported on Fe3O4/SiO2 NPs was found to be Cu(OAc)2 > CuSO4 ≫ CuCl2 > Cu(NO3)2. The catalytic potential of the complex has been improved significantly when core–shell magnetite/silica has been used instead of magnetite for successful wide-spread syntheses of 1-aryl-1,2,3-triazoles under mild reaction conditions. 1-Aryl-1,2,3-triazoles syntheses require harsh reaction conditions.

 Please wait while we load your content...
Please wait while we load your content...