Rational design of the first furoquinolinol based molecular systems for easy detection of Cu2+ with potential applications in the area of membrane sensing†
Manoj Kumar‡
a,
Lokesh Kumar Kumawat‡a,
Vinod Kumar Guptaab and
Anuj Sharma*a
aDepartment of Chemistry, Indian Institute of Technology Roorkee, Roorkee-247667, India. E-mail: anujsharma.mcl@gmail.com; anujsfcy@iitr.ac.in; Fax: +9113 3227 3560; Tel: +9113 3228 4751
bDepartment of Applied Chemistry, University of Johannesburg, Johannesburg, South-Africa
First published on 8th December 2015
Abstract
Two molecular probes based on the furoquinolinol (FQ) framework have been rationally designed for the rapid detection and trace level quantification of Cu2+ in organic and semi-aqueous mediums. Synthesized FQs were extensively examined for their Cu2+ sensing abilities by UV-vis/fluorescence experiments, NMR titrations and DFT based calculations. Higher binding constant [2.11 × 104 M−1 (FQ1) and 1.87 × 104 M−1 (FQ2)], low detection limit [1.52 × 10−7 M (FQ1) and 2.13 × 10−7 M (FQ2)], high selectivity, fast response, wide operational pH range, and repeated usability are some of the salient features of the reported sensors. Furthermore, these compounds (FQs) retained their metal detection abilities in thin PVC membranes.
Introduction
Cu2+ is an ion of ubiquitous presence in living systems with a prominent role in oxidative metabolism and homeostatic regulation.1a The normal functioning of certain enzyme classes such as superoxide dismutase (SOD1), cytochrome-c-oxidase (Cyt-O), tyrosinase and ceruloplasmin rely on the Cu+/Cu2+ redox couple.1a–e While under a normal state, this redox chemistry is essential for life, under “overexpressed” or “misregulated” state, it becomes equally life threatening. It is believed that an excess amount of free Cu2+ triggers the production of ROS (reactive oxygen species) by “Fenton” type reactions, which in turn, is responsible for several pathological states including neurodegenerative diseases and disorders such as Wilson diseases, familial amyotrophic lateral sclerosis, Alzheimer's disease (AD), Parkinson disease (PD) etc.1a–e Excess free Cu2+ concentration is also linked with the reduction of micro-algal biomass and also causes a drastic shift in diatoms/cyanobacteria population in aquatic ecosystems.1fThis population change, generally implicated in various forms such as loss of self cleaning, self healing and reprocessing capabilities of water bodies such as ponds and rivers.1f–g Consequently, detection and trace level quantification of Cu2+ in biological and ecological samples is a research goal of significant interest.
Although instrumental methods such as atomic absorption spectroscopy (AAS), inductively coupled plasma mass spectroscopy (ICP-MS) and voltammetry have been traditionally employed for copper detection, but higher operational cost, requirement of expert handling, destructive nature and low portability have seriously compromised their applicability.2 Design of small molecular systems, sensitive to metal ions and their potential utility as “probing agent” can be an effective solution. In fact, this is an area of growing interest with hundreds of research reports every year. Fluorescent based sensors are particularly important in this regard owing to their advantageous features such as higher sensitivity, portability, quick response and non destructive nature.3
Till date, several molecular systems have been tested for their Cu2+ sensing abilities with varying degree of success.4 Most of these chemical probes are the structural modifications of very few typical scaffold classes such as coumarin,4a–d bispine-dyeconjugate,4e rhodanine–aldazine,4f–g diamines,4h thiadiazole,4i Schiff bases4j–k etc. with common limitations such as slow response, off target selectivity, irreversible nature, operation range at non physiological pH etc. Considering these limitations, design and discovery of molecular probes based on novel structural framework, can be a useful exercise.4
Our group has a longstanding interest in the discovery and design of novel molecular systems with metal sensing abilities.5 In the present report, we have introduced two new annulated furanones based chemosensors 3-(4-bromophenyl)-2-((2-morpholinoethyl)amino)furo[3,2-c]quinolin-4-ol (FQ1), 3-(4-chlorophenyl)-2-(pentylamino)furo[3,2-c]quinolin-4-ol (FQ2) for Cu2+ detection along with their potential applicability in the area of “membrane sensor” and “environment sample analysis”. To best of our knowledge this is the first report of its kind, where metal sensing abilities of amino substituted furoquinolinols (FQs) have been studied in depth.
Results and discussion
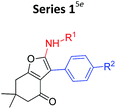
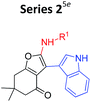
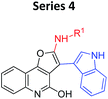
Our group has recently investigated the Al3+ sensing abilities of amino substituted 4-keto-4,5,6,7-tetrahydrobenzofurans.5e During this work, we found that out of 14 compounds studied; only two compounds responded to Al3+. Structurally both the compounds were similar and contained “indole moiety” as a substituent. At that point, we got interested to know the contribution of the core scaffold in their ability to metal binding/sensing. It was based upon our belief that had substituent pattern been only responsible for binding, then a change at framework level would not affect the fluorescent response to a large extent.In order to test our hypothesis, we replaced “dihydrobenzofuranone” by “furoquinolones” not only changed the fluorescent response, but also changed the detection abilities of the compounds. Nearly all the synthesized compounds now sensed Cu2+ in place of expected Al3+ while indoyl substituted furoquinolones (indoyl-FQs) displayed a very slow turn on fluorescence response towards Al3+ alongside turn off fluorescence toward Cu2+ (Table 1).
| Cu2+ (no response) | Cu2+ (no response) | Cu2+ (turn off, fluorescence) | Cu+2 (turn off, fluorescence) |
| Al3+ (no response) | Al3+ (turn on, fluorescence) | Al3+ (no response) | Al3+ (turn on, fluorescence) |
| No response | Fast response (2–3 min) | Fast response (2–3 min) | Slow response (24–48 h) |
| No sensing ability | Sensor for Al3+ | Sensor for Cu2+ | Sense both Cu2+ and Al3+ but not practical as a sensor |
Some important inferences that can be drawn from these observations are, (1) both sensing phenomenon (Al3+ and Cu2+ sensing) might be operated through either by different kind of mechanisms or different portion of compounds might be involved in these interaction. (2) For Al3+ sensing “indoyl” substituent was critical for the turn on fluorescense response (series 2 and 4, Table 1), while for Cu2+, probably furoquinolone framework itself seemed to be important (series 3 and 4). (3) As expected series 4 (indoyl substituent with furoquinolone ring) sensed both the metals, but because of very long response time of 24–48 hours, it would not be practical to use this series as chemical probe. On the other hand, since compounds of series 3 displayed fast response within 2–3 minutes, hence two representative members of this series FQ1 and FQ2 (FQs) were studied for their potential as chemical probe for Cu2+ (Fig. 1).
Synthesis of ionophore FQs6a–c
Both FQs were synthesized by a three step procedure (Scheme 1). At the beginning, fusion of aniline and Meldrum acid (Pechmann reaction) at 90 °C for 9 hours afforded half acid N-phenyl-malonic acid (1) after acid work-up. Acid (1) was then cyclized to quinoline-2,4-diol in the presence of Eaton's reagent (P2O5 in PPA). Desired furoquinolinols were then synthesized by a microwave assisted multicomponent reaction of C–H acid (2) with respective aldehydes and isocyanide. Mechanistically, this three component condensation relied on [4 + 1] cycloaddition between an in situ generated enone and isonitrile.Cu2+ sensing abilities of sensors FQs
Extensive UV-vis and fluorescence studies were carried out to get insight into Cu2+ sensing phenomenon. While representative absorption spectra of synthesized FQs displayed three intense bands centred near 275, 320 and 370 nm, emission spectra had only one broad band at 465 nm in DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (v/v, 1
MeOH (v/v, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9). Addition of 10 equivalents of Li+, K+, Na+, Ca2+, Mg2+, Mn2+, Co2+, Fe2+, Ni2+, Zn2+, Pb2+, In3+, Al3+, Cr3+, Gd3+ and Fe3+ had no apparent effect on UV absorption/fluorescent spectra. On the other hand, addition of just one equivalent of Cu2+ to the solution of FQs (20 μM) totally quenched the emission with slight colour change from yellow to colourless (response time 2–3 min). These effects were also apparent in corresponding UV-vis/fluorescence spectra of FQs in the presence of Cu2+ (Fig. 2–4 and SS7–SS11†). FQs retained this distinct behaviour in other organic solvents such as MeOH, ACN, CHCl3, DMF and mixed aqueous-organic mixtures such as ACN
9). Addition of 10 equivalents of Li+, K+, Na+, Ca2+, Mg2+, Mn2+, Co2+, Fe2+, Ni2+, Zn2+, Pb2+, In3+, Al3+, Cr3+, Gd3+ and Fe3+ had no apparent effect on UV absorption/fluorescent spectra. On the other hand, addition of just one equivalent of Cu2+ to the solution of FQs (20 μM) totally quenched the emission with slight colour change from yellow to colourless (response time 2–3 min). These effects were also apparent in corresponding UV-vis/fluorescence spectra of FQs in the presence of Cu2+ (Fig. 2–4 and SS7–SS11†). FQs retained this distinct behaviour in other organic solvents such as MeOH, ACN, CHCl3, DMF and mixed aqueous-organic mixtures such as ACN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water (v/v, 9
water (v/v, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9), MeOH
9), MeOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water (v/v, 9
water (v/v, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9).
9).
 | ||
Fig. 2 Fluorescence responses of FQ1 in the presence of different metal ion in DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (v/v, 1 MeOH (v/v, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) solution. 9) solution. | ||
 | ||
Fig. 3 UV-vis spectra of FQ1 [20 μM in DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (v/v, 1 MeOH (v/v, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9)] in the presence of 10 equiv. of different ions. The distinct behaviour of Cu2+ (equimolar) is apparent from figure. 9)] in the presence of 10 equiv. of different ions. The distinct behaviour of Cu2+ (equimolar) is apparent from figure. | ||
 | ||
Fig. 4 Fluorescence response of FQ1 [20 μM in DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (v/v, 1 MeOH (v/v, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9)] toward different metal ions (10 equivalents). In the presence of equimolar amount of Cu2+ peak at 465 nm completely disappeared. 9)] toward different metal ions (10 equivalents). In the presence of equimolar amount of Cu2+ peak at 465 nm completely disappeared. | ||
Since selectivity is an important performance indicator of a chemical sensor, hence interference effects of other competitive ions on Cu2+–FQs interaction profile were studied. Gratifyingly, presence of other cations didn't produce any noticeable change either in emission spectra or sensing properties of both FQs, indicating the distinct behaviour of FQs toward Cu2+ (Fig. 5, SS14 and SS15†).
 | ||
Fig. 5 Examination of selectivity of FQ1 [20 μM in DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (v/v, 1 MeOH (v/v, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9)] towards Cu2+ in the presence of interfering ions (λems = 465 nm). 9)] towards Cu2+ in the presence of interfering ions (λems = 465 nm). | ||
When excited at 370 nm both the receptors exhibited high fluorescence quantum yield, Φ = 0.3915 (FQ1), 0.3856 (FQ2) (standard reference: coumarin-1 in ethyl acetate; Φ = 0.99, λex = 355 nm). On the other hand, receptor–Cu2+ complex exhibited very low fluorescence quantum yield, Φ = 0.0253 (FQ1 + Cu2+), 0.0597 (FQ2 + Cu2+). This data ensured significant quenching of fluorescence during the Cu2+ sensing process.
To understand complexometric and stoichiometric aspects of binding, fluorescent titration were carried out (Fig. 6, SS12 and SS13†). Band at 465 nm displayed a gradual decrease in intensity with the incremental addition of Cu2+. Detection limits for Cu2+ was calculated 1.52 × 10−7 M (FQ1) and 2.13 × 10−7 M (FQ2) by universal method (LOD = 3σ/slope) respectively. These results were comparable or better than some of the already known sensors.4,8 In Job's plot intensity minima were noticed, when molar fraction of Cu2+ approached a value of 0.5, indicating 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometric relationship (Fig. SS17 and SS18†) and this fact was further confirmed by ESI-HRMS (Fig. SS19 and SS20†). Binding constants for FQ1 and FQ2 were calculated 2.11 × 104 M−1 and 1.87 × 104 M−1 respectively by plotting measured 1/I−I0 (at 465 nm) against 1/Cu2+ concentrations.7 High correlation was observed between data with Pearson's coefficients (r2) of 0.992 and 0.993 respectively (Fig. 6, SS12 and SS13†).
1 stoichiometric relationship (Fig. SS17 and SS18†) and this fact was further confirmed by ESI-HRMS (Fig. SS19 and SS20†). Binding constants for FQ1 and FQ2 were calculated 2.11 × 104 M−1 and 1.87 × 104 M−1 respectively by plotting measured 1/I−I0 (at 465 nm) against 1/Cu2+ concentrations.7 High correlation was observed between data with Pearson's coefficients (r2) of 0.992 and 0.993 respectively (Fig. 6, SS12 and SS13†).
 | ||
Fig. 6 Fluorescent titration of FQ1 [20 μM in DMSO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MeOH (v/v, 1 MeOH (v/v, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9)] with Cu2+ (from 0.0 equivalent to 2.5 equivalents) at excitation wavelength 370 nm. 9)] with Cu2+ (from 0.0 equivalent to 2.5 equivalents) at excitation wavelength 370 nm. | ||
Furthermore, fluorescence response of FQs toward Cu2+ was found to be pH dependent. Optimum responses were obtained in the pH range of 5.5–10, pointing out that FQs could be utilised in a wide pH range (Fig. 7).
 | ||
| Fig. 7 Dependence of fluorescence response of FQs–Cu2+ over pH of the medium. (λems = 465 and 460 nm respectively for FQ1 and FQ2). | ||
Reversibility study
Reversibility studies were performed to examine the recyclability/reusability of the reported sensors. Addition of 1.0 equiv. of Cu2+ to the equimolar solution of FQ1 caused the expected loss of fluorescence under UV light. It was then treated with aliquots from 1.0 mM solution of EDTA with the gradual appearance in fluorescence, indicating regeneration of free FQ1. To ensure the reusability of FQ1, this mixture was washed with brine solution two times followed by extraction with excess of ethyl acetate and dried over MgSO4. The fluorescent of recovered (90% recovery) FQ1 was again quenched by Cu2+ and again reappeared by EDTA. The binding constants for both fresh and recovered FQ1 were found comparable, indicating the reversibility of interactions. Slight excess of EDTA displaced Cu2+ from its site of interactions with the regeneration of fluorescence. Similar results were obtained for other fluorophores FQ2. These results ensured the repeated applicability of the sensors for Cu2+ detection (Fig. 8).Moreover this cyclic response of FQs towards Cu2+/EDTA is similar to a molecular switch and can be best explained with the help of suitable truth table and logic gate. Two input signal were input-1 (Cu2+) and input-2 (EDTA), presence and absence of an input was denoted by “1” and “0” respectively. The resultant fluorescence response could be used as an output (fluorescence on = 1, fluorescence off = 0) with the condition that presence of Cu2+ alone will quench the fluorescence. This behaviour can be epitomised by “OR” gate with a “NOT” at input-1 (Fig. 9).
 | ||
| Fig. 9 Output response (A) truth table (B) and respective logic gate diagram (C) for reversibility test. | ||
1HNMR titrations
Two probable metal binding sites “site A” (with furan oxygen), “site B” (with quinolinone carbonyl) are shown in Fig. 10. In order to get a deeper insight into binding a 1HNMR titrations were performed.Cu2+ is paramagnetic in nature and is known to have a significant effect over NMR signals of vicinal/coordinating groups. NMR spectra were recorded in the presence (0.25 equivalent to 1.0 equivalent) and absence of copper in DMSO-d6 (Fig. 11, SS22 and SS23†). Signal of NH proton (morpholinoethylamino group) at 6.32 ppm gradually diminished with some initial broadening. Finally this signal completely disappeared at equimolar level of Cu2+, indicating the direct involvance of this peripheral nitrogen in coordination with metal ion. The proton signal of four proton of morpholinoethylamino group of FQ1 at 2.47 also displayed a slight downfield shift toward 2.50 ppm and ultimately merged with DMSO protons. As compare to these protons, aromatic protons (δH = 7–8) and quinolinone NH proton at 11.62 ppm largely remained unchanged during NMR titration. This titration unambiguously proved that site A was directly involved in coordinating with Cu2+ and in satisfying its secondary valency.
 | ||
| Fig. 11 Disappearance of NH peak at 6.32 ppm and shift of aliphatic proton at 2.47 ppm during IHNMR titration (DMSO-d6 was used as NMR solvent and TMS as internal standard). | ||
Theoretical investigation
We believe that quenching response of FQs in the presence of Cu2+ can be best explained by some electron transfer process (ET) that is also in agreement with some of the previous reports. To confirm this possibility, DFT calculations based on B3LYP/6-31G(d,p) methods were performed (Fig. 12 and SS25†).The relevant molecular orbitals (HOMOs and LUMOs) of free FQs and FQ–Cu2+ were explored for this purpose. From Fig. 12, it is obvious that while frontier orbitals of FQs were localised over rigid and conjugated furoquinolinol moiety which acts as actual fluorophore. After coordination with Cu2+, charges were redistributed and most of the electron density was transferred to the receptor site “A”, leaving the rigid fluorophore electron deficient. This flow of charge/electrons from fluorophore to receptor–quencher chelating site is sometimes termed as CHEQ (Chelation Enhanced Fluorophore Quenching) and seemed to be operative in present case.
Comparative study
With good binding constant of 104 and high LOD of 10−7 order of magnitude FQs are well comparable or better than most of the recently reported Cu2+ sensors. High selectivity, instantaneous response, wide operation pH range and repeated usability make FQs a perfect sensor for several important purposes. All these facts are evident from Table SS1† (see also Table SS2† for comparison of sensing properties of two sensors).Applications of the proposed sensors
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9, pH = 7.4). Fluorescent intensity at the band near 465 (FQ1) were recorded and calibration curve (1/fluorescence intensity vs. concentration of added Cu2+) thus obtained was used for the calculation of the concentration of unknown samples. In all the cases, triplicate reading were recorded and average values were taken for used (Table 2). Results were in good agreement with that obtained by AAS (atomic absorption spectroscopy, result A) with relative error less than ±3.0%, indicating the utility of proposed sensors in environmental sample monitoring.
9, pH = 7.4). Fluorescent intensity at the band near 465 (FQ1) were recorded and calibration curve (1/fluorescence intensity vs. concentration of added Cu2+) thus obtained was used for the calculation of the concentration of unknown samples. In all the cases, triplicate reading were recorded and average values were taken for used (Table 2). Results were in good agreement with that obtained by AAS (atomic absorption spectroscopy, result A) with relative error less than ±3.0%, indicating the utility of proposed sensors in environmental sample monitoring.
| Sample | Concentration (A) | Concentration (B) | Relative error in % (B − A/A) × 100 |
|---|---|---|---|
| Tap water | 5.2 × 10−6 M | 5.3 × 10−6 M | +1.92 |
| Canal water | 10.46 × 10−6 M | 10.24 × 10−6 M | −2.10 |
| Industrial waste water | 32.08 × 10−6 M | 32.48 × 10−6 M | +1.24 |
Experimental section
All solvent were distilled prior to use and all chemicals were purchased from sigma-Aldrich® and used without further purification. All NMR spectra were recorded on a Jeol Resonance ECX-400II spectrometer. Chemical shifts are reported in parts per million and are referenced to TMS. Spectra were processed using MestReNova-6 software. Mass spectrometry (HRMS) was performed using a Bruker daltronics microTOF-QII® spectrometer using ESI ionization, with less than 5 ppm error for all HRMS analyses. IR spectra were recorded on a PerkinElmer FT-IR spectrometer in the range 4000–400 cm−1. The UV-vis absorption spectra were recorded on a Shimadzu UV-2450 spectrophotometer and the fluorescent spectra on a ShimadzuRF-5301PC spectrofluorophotometer analytical thin layer chromatography (TLC) was performed on a silica gel plate (Merck® 60F254). For statistical analysis and graphical representation of data Origin 6.0 software was used.Microwave irradiation experiment
All microwave experiments were carried out in a dedicated Anton Paar Monowave 300 reactor®, operating at a frequency of 2.455 GHz with continuous irradiation power of 0 to 300 W. The reactions were performed in a G-4 borosilicate glass vial sealed with Teflon septum and placed in a microwave cavity. Initially, microwave of required power was used and temperature was being ramped from room temperature to a desired temperature. Once this temperature was attained, the process vial was held at this temperature for required time. The reactions were continuously stirred. Temperature was measured by an IR sensor. After the experiments a cooling jet cooled the reaction vessel to ambient temperature.Synthetic procedures
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water (1
water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) or isopropanol
4) or isopropanol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) water (1
water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) was added into it and the precipitated solids (FQs) were filtered.
4) was added into it and the precipitated solids (FQs) were filtered.Analytical data
UV-vis and fluorescence study
For UV and fluorescence studies, stock solutions of the compounds and metal ions Li+, K+, Na+, Ca2+, Mg2+, Mn2+, Co2+, Fe2+, Ni2+, Zn2+, Pb2+, In3+, Al3+, Cr3+, Gd3+ and Fe3+ were prepared (1000 μM) in DMSO: MeOH (v/v, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9). For spectral recordings, the stock solutions were further diluted to 20 μM. All fluorescence spectra were recorded from 390 to 700 nm in a quartz cell (1 cm path length) at room temperature (0.4 nm excitation and emission slit). Solutions were added through a Hamilton burette equipped with 1 ml syringe.
9). For spectral recordings, the stock solutions were further diluted to 20 μM. All fluorescence spectra were recorded from 390 to 700 nm in a quartz cell (1 cm path length) at room temperature (0.4 nm excitation and emission slit). Solutions were added through a Hamilton burette equipped with 1 ml syringe.
Quantum yield calculations
Quantum yields of samples were determined by using standard reference solution of coumarin-1 (Φr = 0.99 in ethyl acetate at an excitation wavelength of 355 nm) and quantum yield is calculated by using following equation:Φs and Φr are the quantum yields of sample and the reference respectively, Ar and As are the respective absorbance of the reference and the sample, Is and Ir are the areas of emission for sample and reference respectively, ηr and ηs are the refractive indices of the sample and reference solutions.
Synthesis of polymeric membrane
5 mg of ionophore (FQ1), plasticizer (o-NPOE), additive NaTPB and high molecular weight PVC were dissolved in THF with continuous stirring. This viscous glue like mixture was then poured into a polyacrylate ring placed on a smooth surface. After evaporation of solvent a fluorescent membrane of about 0.5 mm thickness were obtained.9,10Theoretical calculations
DFT calculations were carried out using Gaussian 09 software package.11 Initially, free FQs and FQ–Cu2+ were optimized using B3LYP exchange function followed by TD-DFT. For FQ–Cu2+ LanL2DZ effective core potential (ECP) basic set was employed, while all other atoms were subjected to ordinary 6-31G(d,p) set. For input–output visualization and for plotting purpose GaussView 5.0 was used.Conclusion
In summary, two new annulated furanones based chemosensors (FQs) were synthesized by an isocyanide based three component condensation. The synthesized probes displayed higher affinity (10+4 M−1), greater selectivity and micromolar detection (10−7 M) for Cu2+ in organic and semi-aqueous solvents. NMR titrations and DFT based calculations predicted the possibility of CHEQ (Chelation Enhanced Fluorophore Quenching) mechanism. The utility of reported systems were demonstrated in the area of PVC based membrane sensing.Acknowledgements
MK and LK would like to thank CSIR and UGC, New Delhi for the award of research fellowship. The authors also thank the departmental Instrumentation lab (IITR) and Institute Instrumental Centre (IIC) for providing the instrumental facilities.Notes and references
- (a) P. Verwilst, K. Sunwoo and J. S. Kim, Chem. Commun., 2015, 51, 5556–5571 RSC; (b) M. C. Linder and M. H. Azam, Am. J. Clin. Nutr., 1996, 63, 797S–811S CAS; (c) L. M. Gaetke, H. S. C. Johnson and C. K. Chow, Arch. Toxicol., 2014, 88, 1929–1938 CrossRef PubMed; (d) Z. L. Harris and J. D. Gitlin, Am. J. Clin. Nutr., 1996, 63, 836S–841S CAS; (e) C. Shen and E. J. New, Metallomics, 2015, 7, 56–65 RSC; (f) C. A. Flemming and J. T. Trevors, Water, Air, Soil Pollut., 1989, 44, 143–158 CrossRef CAS; (g) Y. Zhao, X.-B. Zhang, Z.-X. Han, L. Qiao, C.-Y. Li, L.-X. Jian, G.-L. Shen and R.-Q. Yu, Anal. Chem., 2009, 81, 7022–7030 CrossRef CAS PubMed.
- (a) J. A. Cotruvo, A. T. Aron, K. M. R. Torres and C. J. Chang, Chem. Soc. Rev., 2015, 44, 4400 RSC; (b) N. Pourreza and R. Hoveizavi, Anal. Chim. Acta, 2005, 549, 124–128 CrossRef CAS; (c) A. P. S. Gonzáles, M. A. Firmino, C. S. Nomura, F. R. P. Rocha, P. V. Oliveira and I. Gaubeur, Anal. Chim. Acta, 2009, 636, 198–204 CrossRef PubMed; (d) J. S. Becker, M. V. Zoriy, C. Pickhardt, N. P. Gallagher and K. Zilles, Anal. Chem., 2005, 77, 3208–3216 CrossRef CAS PubMed; (e) V. Beni, V. I. Ogurtsov, N. V. Bakunin, D. W. M. Arrigan and M. Hill, Anal. Chim. Acta, 2005, 552, 190–200 CrossRef CAS; (f) A. A. Ensafi, T. Khayamian, A. Benvidi and E. Mirmomtaz, Anal. Chim. Acta, 2006, 561, 225–232 CrossRef CAS.
- (a) D. Karak, S. Lohar, A. Sahana, S. Guha, A. Banerjee and D. Das, Anal. Methods, 2012, 4, 1906–1908 RSC; (b) Y. J. Zheng, J. Orbulescu, X. J. Ji, F. M. Anrreopoulos, S. M. Pham and R. M. J. Leblanc, J. Am. Chem. Soc., 2003, 125, 2680–2686 CrossRef CAS PubMed; (c) Y. J. Zheng, X. H. Cao, J. Orbulescu, V. Konka, F. M. Andreopoulos, S. M. Pham and R. M. Lebac, Anal. Chem., 2003, 75, 1706–1712 CrossRef CAS; (d) Y. T. Li and C. M. Yang, Chem. Commun., 2003, 2884–2885 RSC; (e) D. Karak, A. Banerjee, S. Lohar, A. Sahana, S. K. Mukhopadhyay, S. S. Adhikari and D. Das, Anal. Methods, 2013, 5, 169–172 RSC; (f) I. Majumder, P. Chakraborty, S. Das, H. Kara, S. K. Chattopadhyay, E. Zangrando and D. Das, RSC Adv., 2015, 5, 51290–51301 RSC; (g) G. Sivaraman, T. Anand and D. Chellappa, Analyst, 2012, 137, 5881–5884 RSC; (h) G. Sivaraman, T. Anand and D. Chellappa, RSC Adv., 2012, 28, 10605–10609 RSC; (i) G. Sivaraman, B. Vidya and D. Chellappa, RSC Adv., 2014, 58, 30828–30831 RSC; (j) S. Adhikari, S. Mandal, A. Ghosh, S. Guria and D. Das, Dalton Trans., 2015, 44, 14388–14393 RSC; (k) S. Adhikari, A. Ghosh, S. Mandal, A. Sahana and D. Das, RSC Adv., 2015, 5, 33878–33884 RSC; (l) M. Iniya, D. Jeyanthi, K. Krishnaveni and D. Chellappa, RSC Adv., 2014, 48, 25393–25397 RSC; (m) A. Misra, P. Srivastava and M. Shahid, Analyst, 2012, 137, 3470–3478 RSC; (n) M. Shahid, P. Srivastava and A. Misra, New J. Chem., 2011, 35, 1690–1700 RSC; (o) A. Biswas, R. Pandey, D. Kushwaha, M. Shahid, V. K. Tiwari, A. Misra and D. S. Pandey, Tetrahedron Lett., 2013, 32, 4193–4197 CrossRef.
- (a) H. S. Jung, P. S. Kwon, J. W. Lee, J. I. Kim, C. S. Hong, J. W. Kim, S. Yan, J. Y. Lee, J. H. Lee, T. Joo and J. S. Kim, J. Am. Chem. Soc., 2009, 131, 2008–2012 CrossRef CAS PubMed; (b) Y. Shan, Y. Sun, N. Sun, R. Guan, D. Cao, K. Wang, Q. Wu and Y. Xu, Inorg. Chem. Commun., 2015, 59, 68–70 CrossRef CAS; (c) S. Areti, J. K. Khedkar, S. Bandaru, R. Teotia, J. Bellare and C. P. Rao, Anal. Chim. Acta, 2015, 873, 80–87 CrossRef CAS PubMed; (d) S. S. Razi, P. Srivastava, R. Ali, R. C. Gupta, S. K. Dwivedi and A. Misra, Sens. Actuators, B, 2015, 209, 162–171 CrossRef CAS; (e) D. Brox, P. Comba, D.-P. Hertena, E. Kimmle, M. Morgen, C. L. Rühl, A. Rybina, H. Stephan, G. Storch and H. Wadepohl, J. Inorg. Biochem., 2015, 148, 78–83 CrossRef; (f) Y. Fang, Y. Zhou, Q. Rui and C. Yao, Organometallics, 2015, 34, 2962–2970 CrossRef CAS; (g) Q. Meng, R. Zhang, H. Jia, X. Gao, C. Wang, Y. Shi, A. V. E. Dass and Z. Zhang, Talanta, 2015, 143, 294–301 CrossRef CAS PubMed; (h) L. Qu, C. Yina, F. Huob, Y. Zhang and Y. Li, Sens. Actuators, B, 2013, 183, 636–640 CrossRef CAS; (i) O. Çimen, H. Dinçalp and C. Varlikli, Sens. Actuators, B, 2015, 209, 853–863 CrossRef; (j) U. N. Yadav, P. Pant, S. K. Sahoob and G. S. Shankarling, RSC Adv., 2014, 4, 42647–42653 RSC; (k) Z. Chen, L. Wang, G. Zou, J. Tang, X. Cai, M. Teng and L. Chen, Spectrochim. Acta, Part A, 2013, 105, 57–61 CrossRef CAS.
- (a) V. K. Gupta, N. Mergu and L. K. Kumawat, Sens. Actuators, B, 2016, 223, 101–113 CrossRef CAS; (b) V. K. Gupta, N. Mergu, L. K. Kumawat and A. K. Singh, Talanta, 2015, 144, 80–89 CrossRef CAS PubMed; (c) V. K. Gupta, A. K. Singh and L. K. Kumawat, Sens. Actuators, B, 2014, 204, 507–514 CrossRef CAS; (d) L. K. Kumawat, N. Mergu, A. K. Singh and V. K. Gupta, Sens. Actuators, B, 2015, 212, 389–394 CrossRef CAS; (e) M. Kumar, L. K. Kumawat, V. K. Gupta and A. Sharma, ChemistryOpen, 2015, 4, 626–632 CrossRef CAS PubMed; (f) V. K. Gupta, A. K. Singh, L. K. Kumawat and N. Mergu, Sens. Actuators, B, 2016, 222, 468–482 CrossRef CAS.
- (a) M. Kumar, T. Kaur, V. K. Gupta and A. Sharma, RSC Adv., 2015, 5, 17087–17095 RSC; (b) M. Kumar, S. Bagchi and A. Sharma, RSC Adv., 2015, 5, 53592–53603 RSC; (c) S.-J. Park, J.-C. Lee and K.-I. Lee, Bull. Korean Chem. Soc., 2007, 28, 1203–1205 CrossRef CAS.
- H. A. Benesi and J. H. Hildebrand, J. Am. Chem. Soc., 1949, 71, 2703–2707 CrossRef CAS.
- (a) J. Wang and Q. Zong, Sens. Actuators, B, 2015, 216, 572–577 CrossRef CAS; (b) H. Ye, F. Ge, Y.-M. Zhou, J.-T. Liu and B.-X. Zhao, Spectrochim. Acta, Part A, 2013, 112, 132–138 CrossRef CAS PubMed.
- V. K. Gupta, A. K. Singh and L. K. Kumawat, Electrochim. Acta, 2013, 95, 132–138 CrossRef CAS.
- L. Liu, A. Wang, G. Wang, J. Li and Y. Zhou, Sens. Actuators, B, 2015, 215, 388–395 CrossRef CAS.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision A.02, Gaussian Inc., Wallingford, CT, 2009 Search PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra21862d |
| ‡ Contributed equally as co-first authors. |
| This journal is © The Royal Society of Chemistry 2015 |