H2O2 in WERSA: an efficient green protocol for ipso-hydroxylation of aryl/heteroarylboronic acid†
Eramoni Saikia,
Sankar Jyoti Bora and
Bolin Chetia*
Department of Chemistry, Dibrugarh University, Dibrugarh-786004, Assam, India. E-mail: bolinchetia@dibru.ac.in
First published on 25th November 2015
Abstract
A mild, green, economic and efficient protocol has been developed for ipso-hydroxylation of aryl/heteroarylboronic acids to phenols using 30% aqueous H2O2 as an oxidant and WERSA (water extract of rice straw ashes) as a neat reaction medium. All reactions were carried out without using metal, ligand, activator or hazardous organic solvent with excellent yield within a very short reaction time at room temperature. Therefore, this appears to be the cleanest and greenest alternative protocol for ipso-hydroxylation of aryl/heteroarylboronic acids.
Introduction
Phenols are ubiquitous in nature as they are the key structural constituents of various pharmaceuticals,1 agrochemicals,2 polymers3 and naturally occurring compounds mainly as polyphenols4 (natural antioxidants). Traditional methods for the preparation of phenols involve nucleophilic substitution of activated aryl halides, Cu-catalysed conversion of diazoarenes5 and Pd-catalysed conversion of aryl halides6 to phenols. But these existing routes require harsh reaction conditions due to inactive nature of starting materials. However, as a result of continuous effort of many research groups, arylboronic acids have replaced the traditional inactive synthetic precursor for phenols. Ipso-hydroxylation, the generalized method for the conversion of arylboronic acids to phenols is gaining enormous importance due to higher availability, stability and greater functional group diversity of arylboronic acids. As a result various protocols for ipso-hydroxylation of arylboronic acids were put forwarded using different catalysts such as biosilica–H2O2,7 I2–H2O2,8 Al2O3–H2O2,9 PEG–H2O2,10 NH2OH,11 KOH–TBHP,12 CuSO4–phenanthroline,13 H3BO4–H2O2,14 Amberlite IR-120 resin,15 supported silver nano particle,16 organic hypervalent iodine(III)17 etc.Even with all the existing protocols, majority of which found to be effective for conversion of arylboronic acids to phenols via ipso-hydroxylation, there is a serious need for “green” protocols with greater environmental and economical viability due to some unavoidable drawbacks of the reported protocols such as use of transition metal as catalyst, ligands, bases and harmful organic solvents. Recently Saikia et al., following the green chemistry tools have explored natural feedstock as green alternatives for carrying out two popular organic reactions Suzuki–Miyaura cross coupling18 and Dakin reaction.19 Inspired by their work and following the motion of “greening-up” here we wish to report a superb protocol for ipso-hydroxylation of arylboronic acids to phenols using WERSA (water extract of rice straw ashes) a highly abundant, cheap natural feedstock as neat reaction media and 30% aqueous H2O2 as oxidant. To the best of our knowledge this is the first report on ipso-hydroxylation of arylboronic acids to phenols using WERSA and we therefore believe that this is an outstanding alternative protocol for synthesis of phenols from arylboronic acids, as reaction condition is mild, straightforward and without use of metal, ligand, bases and solvents. Additional beauty of the protocol is the use of H2O2 as oxygen source rather than other oxygen sources like NaClO2,20 N-oxide,21 HOF22 as H2O2 is richer source of oxygen and forms water as byproduct.
Experimental
At the beginning we tried our first reaction with phenyl boronic acid (1 mmol) using 30% aqueous H2O2 (0.10 mL) as oxidant in WERSA (2 mL) (Scheme 1) which was prepared using the established procedure23 to check the feasibility of the reaction. We monitored the reaction by TLC in every min and to our delight phenyl boronic acid fully converted to the desired product phenol within 10 minutes at room temperature. After that we moved on for optimization of oxidant H2O2 needed for full conversion taking phenyl boronic acid as model substrate. As a result of optimization as depicted in Table 1 we found that 0.20 mL of 30% aqueous H2O2 oxidant were sufficient to complete the reaction within 5 minutes. Along with mild reaction condition, excellent yield, lesser toxicity, operational plainness, cost effectiveness makes this hydroxylation protocol one of the cleanest and greenest alternative protocol ever.With the optimized condition, we then carried out numbers of reactions with different electron-rich and electron-poor arylboronic acid to study the substrate scope using this newly designed protocol. According to our expectation, reactions proceeded very smoothly converting various electron-donating and electron-withdrawing groups bearing arylboronic acids to phenols with very good to excellent yield within very short reaction time (Table 2). Results showed that substrate containing electron withdrawing groups were more efficient than electrons donating ones. Moreover, we readily carried out the reaction with some heteroarylboronic acids (Table 2). Till now, there is no earlier report on ipso-hydroxylation using such a mild, green and economic reaction condition. Therefore, this seems to be an evergreen protocol with a wide range of substrate scope for ipso-hydroxylation of arylboronic acids to phenols.
| Entry | Starting materials | Products | Time (min) | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: aryl/heteroarylboronic acid (1 mmol), 30% H2O2 (0.2 mL for each substrate) in WERSA (2 mL) at room temperature.b Isolated yields. | ||||
| 1 |  |
 |
5 | 98 |
| 2 |  |
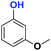 |
10 | 92 |
| 3 |  |
 |
7 | 95 |
| 4 | 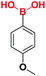 |
 |
10 | 94 |
| 5 |  |
 |
15 | 90 |
| 6 |  |
 |
10 | 92 |
| 7 |  |
 |
6 | 95 |
| 8 |  |
 |
8 | 94 |
| 9 |  |
 |
8 | 97 |
| 10 |  |
 |
10 | 92 |
| 11 |  |
 |
15 | 90 |
| 12 |  |
 |
15 | 91 |
Results and discussion
Nucleophilic activation of H2O2 is a general fact to use H2O2 as oxidant; as a result numbers of bases have already been used to activate H2O2 by abstracting a proton so far. Here in this protocol we used only WERSA with H2O2 as a neat reaction media for ipso-hydroxylation of arylboronic acids to phenols. Perhaps the exact role of WERSA for the reaction is not clearly understood yet. However, literature24 reveals that rice straw ashes contains oxides of SiO2 (74.31%), Al2O3 (1.40%), Fe2O3 (0.73%), TiO2 (0.02%), CaO (1.61%), MgO (1.89%), K2O (11.30%), Na2O (1.85%), P2O5 (2.65%) as primary ingredients. Thus water extract of rice straw ashes may contain KOH, NaOH which may be responsible for making WERSA basic enough for nucleophilic activation of H2O2 by abstracting its proton. Therefore we proposed a plausible mechanism for ipso-hydroxylation of arylboronic acid to phenol using H2O2 in WERSA (Fig. 1).Recyclability of WERSA is another attractive feature of this protocol. We reused WERSA upto 5th cycle without significant lose of efficiency (Table 3). Taking phenyl boronic acid as model substrate we carried out the WERSA recyclability test. After completion of the reactions, products were extracted with diethylether and WERSA separated from product was washed with more diethylether and reused. Thus we can consider WERSA as a reusable homogeneous catalyst.
Conclusions
In conclusion, we have developed a green, mild, efficient and aerobic protocol for ipso-hydroxylation of arylboronic acids to phenols using H2O2 as oxidant in WERSA at room temperature. With these mild reaction conditions, our method is very compatible with various electron-donating and electron-withdrawing groups at ortho, meta and para positions on the aromatic ring. In addition, all reactions occur in shortest reaction time without using any activating agent, metal, ligand and organic solvent. From the environmental and economical point of view, we believe that this is the most greener and efficient protocol for ipso-hydroxylation of arylboronic acids to phenols. All these advantages make H2O2–WERSA a very adaptable and competitive catalyst system, consequently can be used as clean and safer alternative for various other reactions in laboratory as well as in industry in near future.Acknowledgements
BC gratefully acknowledges UGC, New Delhi (grant no. 42357/2013) and DST-SERB (No. SB/FT/CS-161/2012) for financial assistance. ES is thankful to the Department of Science and Technology (Government of India) for INSPIRE-Fellowship and SAIF, NEHU-Shillong for spectral data.Notes and references
- Z. Rappoport, The Chemistry of Phenols, Wiley-VCH, Weinheim, Germany, 2003 Search PubMed.
- J. H. P. Tyman, Synthetic and Natural Phenols, Elsevier, New York, 1996 Search PubMed.
- (a) S. Quideau, D. Deffieux, C. Douat-Casassus and L. Pouysegu, Angew. Chem., Int. Ed., 2011, 50, 586–621 CrossRef CAS PubMed; (b) Y. Ji, P. Li, X. Zhang and L. Wang, Org. Biomol. Chem., 2013, 11, 4095–4101 RSC.
- R. W. Owen, A. Giacosa, R. Haubner, B. Spiegel-hader and H. Bartsch, Eur. J. Cancer, 2000, 36, 1235–1247 CrossRef CAS PubMed.
- P. Hanson, J. R. Jones, A. B. Taylor, P. H. Walton and A. W. J. Timms, J. Chem. Soc., Perkin Trans. 2, 2002, 1135–1150 RSC.
- G. A. Sergeev, T. Schultz, C. Torbog, A. Spenenberge and H. Neumann, Angew. Chem., Int. Ed., 2009, 48, 7595–7599 CrossRef PubMed.
- A. Mahanta, P. Adhikari, U. Bora and A. J. Thakur, Tetrahedron Lett., 2015, 56, 1780–1783 CrossRef CAS.
- A. Gogoi and U. Bora, Synlett, 2012, 1079–1081 CAS.
- A. Gogoi and U. Bora, Tetrahedron Lett., 2013, 54, 1821–1823 CrossRef CAS.
- M. Gohain, M. D. Plessis, J. H. Tonder and B. C. B. Bezuidenhoudt, Tetrahedron Lett., 2014, 55, 2082–2084 CrossRef CAS.
- E. Kinmehr, M. Yahyaee and K. Tabatabai, Tetrahedron Lett., 2007, 48, 2713–2715 CrossRef.
- S. Guo, L. Lu and H. Cai, Synlett, 2013, 24, 1712–1714 CrossRef CAS.
- J. Xu, X. Wang, C. Shao, D. Su, G. Cheng and Y. Hu, Org. Lett., 2010, 12, 1964–1967 CrossRef CAS PubMed.
- K. Gogoi, A. Dewan, A. Gogoi, G. Borah and U. Bora, Heteroat. Chem., 2014, 25, 127–130 CrossRef CAS.
- N. Mulakayala, Ismail, K. M. Kumar, R. K. Rapolu, B. Kandagatla, P. Rao, S. Oruganti and M. Pal, Tetrahedron Lett., 2012, 53, 6004–6007 CrossRef CAS.
- T. Begum, A. Gogoi, P. K. Gogoi and U. Bora, Tetrahedron Lett., 2015, 56, 95–97 CrossRef CAS.
- N. Chatterjee and A. Goswami, Tetrahedron Lett., 2015, 56, 1524–1527 CrossRef CAS.
- P. R. Boruah, A. A. Ali, B. Saikia and D. Sarma, Green Chem., 2015, 17, 1442–1445 RSC.
- B. Saikia, P. Borah and N. C. Barua, Green Chem., 2015, 17, 4533–4536 RSC.
- P. Gogoi, P. Bezboruah, J. Gogoi and R. C. Boruah, Eur. J. Org. Chem., 2013, 7291–7294 CrossRef CAS.
- C. Zhu, R. Wang and J. R. Falck, Org. Lett., 2012, 14, 3494–3497 CrossRef CAS PubMed.
- J. Gatenyo, I. Vints and S. Rozen, Chem. Commun., 2013, 7379–7381 RSC.
- P. R. Boruah, A. A. Ali, M. Chetia, B. Saikia and D. Sarma, Chem. Commun., 2015, 51, 11489–11492 RSC.
- B. M. Jenkins, R. R. Bakker and J. B. Wei, Biomass Bioenergy, 1996, 4, 177–200 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra21354a |
| This journal is © The Royal Society of Chemistry 2015 |


