Integrating three-dimensional graphene/Fe3O4@C composite and mesoporous Co(OH)2 nanosheets arrays/graphene foam into a superior asymmetric electrochemical capacitor†
Abstract
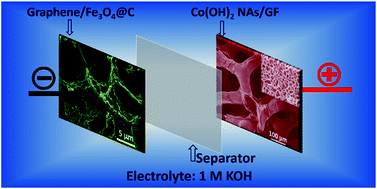
Aqueous electrolyte-based asymmetric electrochemical capacitors (AECs) are promising in the field of energy storage because of their wider potential windows compared to the symmetric capacitors and higher ionic conductivity compared to the organic electrolytes. Most of the research works on AECs are directed towards cathode materials, while anode materials have rarely been investigated. Herein, a novel AEC is constructed, in which two highly conductive and lightweight graphitic substrates, graphene framework and graphene foam, are hybridized with Fe3O4@C core–shell nanoparticles (anodes) and mesoporous Co(OH)2 nanosheets arrays (NAs) (cathodes), respectively. The as-assembled AEC device shows extended cell voltage (0.0–1.6 V) and excellent cycle stability (72% retention after 8000 cycles). More importantly, a high specific energy of 75 W h kg−1 is achieved at a specific power of 400 W kg−1. Even at a 10.3 s charge/discharge rate, specific energy as high as 33 W h kg−1 can be retained.

 Please wait while we load your content...
Please wait while we load your content...