Spherical 3-nitro-1,2,4-triazol-5-one (NTO) based melt-cast compositions: heralding a new era of shock insensitive energetic materials†
Abstract
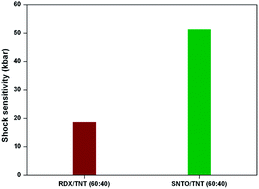
3-Nitro-1,2,4-triazol-5-one (NTO) is an unique candidate among military high explosives and is explored as a potential bomb filler with TNT in melt cast formulations. In the present study, we attempted to replace sensitive RDX with NTO with spherical morphology to develop a less hazardous, thermally stable shock insensitive composition. A bimodal mixture (150 and 25 μm in 70/30 ratio) of spherical NTO powders with superior flowability was chosen for the formulations and achieved 60% solid loading. The temperature sensitivity of the formulations was assessed by calculating the activation energy for the flow. The velocity of detonation and shock sensitivity of the composition were also determined. The study demonstrated that the spherical-NTO/TNT (60 : 40) was found to be 2.5 times more shock and 2 times more friction insensitive than composition B which consisted of RDX/TNT (60 : 40). The activation energy for thermal decomposition was determined to assess the thermal hazards and a vacuum stability test was carried out to ensure the storage life of the composition.

 Please wait while we load your content...
Please wait while we load your content...