A novel epoxy-functionalized hyperbranched polysiloxane (HPSi) endowing methyl phenyl silicone resin (Si603)/epoxy systems with enhanced compatibility and fire retardancy performance
Pei Jia,
Hanchao Liu,
Youchuan Wang and
Xufu Cai*
Department of Polymer Science and Engineering, Sichuan University, Chengdu 610065, China. E-mail: caixf2008@scu.edu.cn
First published on 2nd November 2015
Abstract
A novel hyperbranched polysiloxane (HPSi) with a great amount of epoxy groups was synthesized as a compatibilizer of epoxy resin (EP)/methyl phenyl silicone resin (Si603) blends. The structure of HPSi has been analyzed by Fourier transform infrared spectroscopy (FTIR) and nuclear magnetic resonance (1H-NMR) spectroscopy. The compatibility of EP/HPSi/Si603/DDM was characterized by differential scanning calorimetry (DSC) and scanning electron microscopy (SEM), and the results showed that HPSi significantly improved the compatibility of EP/Si603. The flame retardancy and thermal degradation behavior of the EP/HPSi/Si603/DDM systems were investigated using the limiting oxygen index (LOI), UL-94 vertical burning, thermogravimetric analysis (TG), FTIR and SEM. The LOI value of the modified EP resin with 10% HPSi and 30% Si603 was 31 (about 1.4 times the corresponding value of the original EP resin), and passed V-1 of the UL-94 rating. Specifically, its combustion residue at 800 °C was about 2.24 times that of the original EP resin. Moreover, structural analysis of the combustion residue by FTIR showed the formation of polyaromatic carbons. Additionally, the morphology of the residue char showed the compact, smooth, and tight structure of the EP/HPSi/Si603/DDM systems. Mechanical properties such as tensile strength, flexural strength and flexural modulus of EP/DDM and EP/HPSi/Si603/DDM systems were evaluated and the result showed that these polymers also possess good mechanical properties. These outstanding integrated properties would make EP/HPSi/Si603/DDM systems attractive for practical applications.
1. Introduction
Epoxy resin (EP) is one of the most important commercial thermosetting materials owing to its high tensile strength and modulus, outstanding adhesive properties, good chemical resistance and superior dimensional stability.1–4 It is widely used as adhesives, coatings, semiconductor encapsulation and matrices for advanced composites in electronic/electrical industries where a remarkable grade of flame-retardancy is required. However, the flammability is a major drawback of epoxy resin and limits its applications, thus, the improvement of the flame retardancy of EP is an urgent problem and has attracted great attention.5,6 To protect the environment and human health, environmentally friendly halogen-free fire retardancy for epoxy resins has always been the focus,7 and silicon-containing compounds are considered a promising choice due to their high flame-retardant efficiency.8,9As one of the silicon-containing compounds, methyl phenyl silicone resin (Si603) with a high thermal stability and carbon residue was selected to modify epoxies. However, it is important to note that the introduction of polysiloxane flame retardant into the resin not only demonstrated poor compatibility with the polymer matrix, but also lowered the glass transition temperature (Tg).10 Therefore, how to overcome these drawbacks without a decrease of the integrated properties of polysiloxane is worth investigating.
Recently, studies on hyperbranched polymers used as compatibilizers have received a growing prominence. Because of the hybrid inorganic–organic nature,11,12 the hyperbranched polymer possessing epoxy group, which can serve to increase the compatibility with polymers, can improve the thermal stability and the flame retardancy of the EP composites. In addition, the existence of highly reactive groups provides the possibility for chemical modifications.13 A typical example is the fully end-capped hyperbranched polysiloxane with a large branching degree and amine-groups (AmeHBPSi), which was used to modify BMI resin to improve the flame retardancy, toughness, strength and thermal stability.14 Juhua Ye15 synthesized a novel phosphorus-containing hyperbranched polysiloxane which was then used to improve the flame retardancy of cyanate ester resins.
The subject of this work is to improve the compatibility of methyl phenyl silicone resin (Si603)/epoxy resin with a hyperbranched polymer. Integrated performances (including compatibilization, thermal degradation behavior, and the flame retardancy) of HPSi/methyl phenyl silicone resin/epoxy resin systems were intensively investigated.
2. Experimental
2.1. Materials
Diglycidyl ether of bisphenol A (DGEBA, EP, epoxy value = 0.44 mol/100 g) was purchased from Wuxi Bluestar Chemical Co. Ltd. China. Methyl phenyl silicone resin (Si603) was supplied by WACKER, Germany. γ-(2,3-Epoxypropoxy)propyltrimethoxysilane (KH560) and dimethoxydimethylsilane (DEMS) were bought from Jinan Yijia Chemical Co. Ltd. China. Distilled water, 4,4-diaminodiphenylmethane (DDM), ammonia, and anhydrous ethanol were obtained from Chengdu Kelong Chemical Reagent Factory. All materials were used without further purification.2.2. Synthesis of polysiloxane (HPSi)
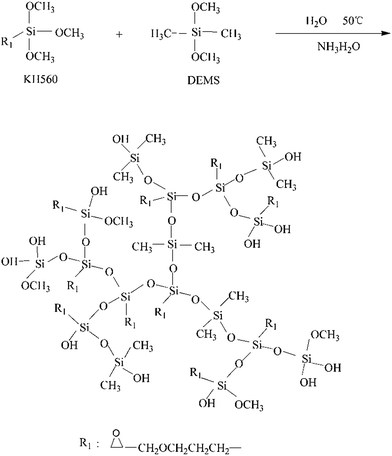
23.6 g KH560, 18 g DEMS and 150 mL anhydrous ethanol were added to a 250 mL three-necked round bottom flask equipped with a stirrer, a thermometer, and a condenser. Then 0.4 g ammonia and 7.56 g distilled water were both added into the flask. The temperature was raised to 50 °C and kept at that temperature for 4.5 h. After that, 0.25 g 1-chlorotrimethylsilane was added as a capping agent. Finally, the product was dried to eliminate methanol, ethanol, and water in a vacuum oven. Finally, a transparent and viscous liquid, hyperbranched polysiloxane (coded as HPSi), was obtained. The reaction mechanism of this system is shown in Scheme 1.2.3. Preparation of cured HPSi/EP/Si603/DDM blends
HPSi and epoxy with a ratio of 0.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (mass) was blended with vigorous stirring at 130 °C for 20 min to obtain a prepolymer. Then an appropriate amount of Si603 (10 wt%, 20 wt%, 30 wt%) was put into the above prepolymer. Then, stoichiometric DDM was cast into the blends above to dissolve at 100 °C. Subsequently, the blends were cast into a preheated aluminium mold for curing and postcuring following the protocol of 80 °C/2 h + 150 °C/3.5 h + 165 °C/1.5 h. Then the epoxy resin specimens were obtained and subsequently machined to the desirable size for further testing.
1 (mass) was blended with vigorous stirring at 130 °C for 20 min to obtain a prepolymer. Then an appropriate amount of Si603 (10 wt%, 20 wt%, 30 wt%) was put into the above prepolymer. Then, stoichiometric DDM was cast into the blends above to dissolve at 100 °C. Subsequently, the blends were cast into a preheated aluminium mold for curing and postcuring following the protocol of 80 °C/2 h + 150 °C/3.5 h + 165 °C/1.5 h. Then the epoxy resin specimens were obtained and subsequently machined to the desirable size for further testing.
2.4. Measurements
3. Results and discussion
3.1. Synthesis and characterization of HPSi
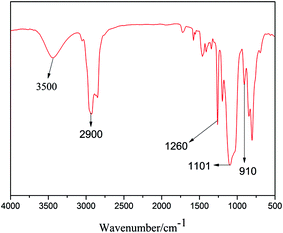
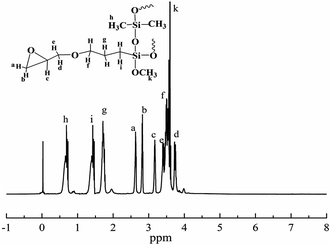
In order to confirm the molecular structure of HPSi, FTIR, and 1H-NMR analyses were carried out. Fig. 1 is the FTIR spectrum of HPSi. The FTIR spectrum of HPSi shows that the strong and wide peak at 1101 cm−1 was attributed to the bending vibration of the Si–O–Si groups, indicating that the alkoxy groups were successfully changed into Si–O–Si through the hydrolysis and condensation; however the broad peak centered at 3500 cm−1 representing the Si–OH groups can also be observed, demonstrating that the Si–OH groups in the raw material do not completely disappear during the condensation.16 The emergence of the chemical shift at 2.79 ppm assigned to –OH groups in the 1H-NMR of HPSi (Fig. 2) also supports this statement. Moreover, a stretching vibration of the oxirane ring group at 910 cm−1 demonstrates that polymerization between KH560 and DEMS occurred. We can also see a vibration peak (2840 cm−1) assigned to methoxy, and the peaks in the range of 750 to 870 cm−1 indicate the existence of Si–CH3. To further prove the structure of HPSi, Fig. 2 shows the 1H-NMR spectrum of HPSi (ppm): 2.6 (a), 2.79 (b), 3.18 (c), 3.67 (d), 3.32 (e), 3.48 (f), 1.62 (g), 3.52 (k), 0.67 (i), 7.12–7.66 (j).17 It suggests that the HPSi is successfully synthesized.3.2. Compatibility of cured HPSi/EP/Si603/DDM blends
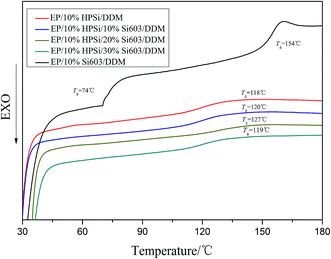
The compatibility of EP/Si603/DDM improved by HPSi can be characterized by measuring the glass transition temperature (Tg). Generally speaking, the homogeneous morphology of the mixed epoxy systems exhibits only one Tg, while incompatible mixtures exhibit two or more Tg values. Differential scanning calorimetry (DSC) can accurately detect the Tg of the blends.18 Fig. 3 gives the DSC curves of various cured EP/Si603/DDM, EP/HPSi/DDM, EP/HPSi/Si603/DDM mixtures. EP/Si603/DDM exhibits two separated Tg values at 74 and 154 °C, which are close to the Tg of Si603 and pure epoxy resin, respectively. Except EP/Si603, the rest of the curing systems have a single Tg between 119 and 127 °C, which indicates that HPSi greatly improved the compatibility of this system. This phenomenon can be mainly interpreted by the fact that HPSi contains a large amount of epoxy groups and Si–O–Si bonds, which have a similar structure to the EP and Si603 resin, respectively. It can also be observed that with the increase of Si603, the Tg of the whole system increases until it reaches the maximum value of 127 °C, following which there is an obvious decrease. Owing to a relatively large space volume, Si603 resin usually has higher average free volume which plays a negative role in increasing the concentration of the chain segment of modified resins. At the same time, in the case of the EP/Si603/HPSi/DDM system, there are strong interactions between Si603 and HPSi, which can conspicuously increase the packing density of the polymer. The positive factor is dominant when the content of Si603 is between 10 and 20 wt%, while when the content of Si603 increases from 20 to 30 wt% the negative factor will offset this positive effect. From the data analysis of differential scanning calorimetry (DSC), the addition of HPSi successfully improves the compatibility of EP/Si603/DDM, which proves that Si603 increases the flame retardancy of EP.To further explore the influence of HPSi in the cured system, SEM morphologies of the fractured surfaces of the samples are shown in Fig. 4. The sample of EP/Si603/DDM resin is laminated with a translucent yellow bottom and a white opaque upper layer. Fig. 4a is the SEM photograph of the EP/30% Si603/DDM cured sample. Holes and balls are almost invisible because Si603 has migrated to the surface of the matrix in the process of blending and curing. In addition, forming balls in the process of blending was also responsible for the serious phase separation. Such phenomena demonstrate the poor compatibility of EP/Si603, as Si603 can’t be well dispersed in the EP matrix in the absence of a compatibilizer. The morphology of the EP/10% HPSi/DDM cured sample shows a compact, smooth, and tight surface in Fig. 4b,which demonstrates the outstanding compatibility of EP/HPSi.
Cured EP/10% HPSi/30% Si603/DDM is transparent and uniformly yellow. It can be observed in Fig. 4c that the fractured surface is flat. What’s more, small balls and circular holes appear on the fractured surface. These balls are evenly distributed in the matrix, which could be attributed to the fact that the Si603 is ideally dispersed in the EP matrix in the presence of HPSi. After being placed in anhydrous ethanol for 20 hours, a large number of circular holes appear on the fractured surface of EP/10% HPSi/30% Si603/DDM (Fig. 4d), which is caused by the dissolution of part of the balls occupied by Si603 resin.
3.3. Flammability
LOI, the lowest volume concentration of oxygen sustaining the burning of materials in a mixture of nitrogen and oxygen gases,19 is an important parameter for evaluating the ease of extinguishment of polymeric materials in the same atmosphere. The flame retardant properties of the cured EP/DDM and EP/HPSi/Si603/DDM systems were evaluated by LOI values and UL-94. Data are listed in Table 1. It should be noted that the LOI value gradually increases from 26 to 31 (about 1.4 times of the LOI value of the EP resin) as the Si603 content increases from 10% to 30%. At the same time, less smoke was produced after adding Si603 resin. What’s more, the residue with Si603 resin could keep the original shape and form a hollow shell after burning. The test results show that the vertical combustion performance of the EP/10% HPSi/30% Si603/DDM system can achieve a V-1 level and shows a good anti-drip property. However, the LOI value of pure EP is 21.5 and the UL-94 rating is not V-2, with an unsatisfying anti-drip property. This indicates that the compatibilizing effect of HPSi have a good influence on the flammability of the EP/HPSi/Si603/DDM systems.| Sample | LOI (%) | UL-94 |
|---|---|---|
| EP/DDM | 21.5 | NR |
| EP/10% HPSi/DDM | 23 | NR |
| EP/10% HPSi/10% Si603/DDM | 26 | V-2 |
| EP/10% HPSi/20% Si603/DDM | 28.5 | V-2 |
| EP/10% HPSi/30% Si603/DDM | 31 | V-1 |
3.4. Thermal analysis
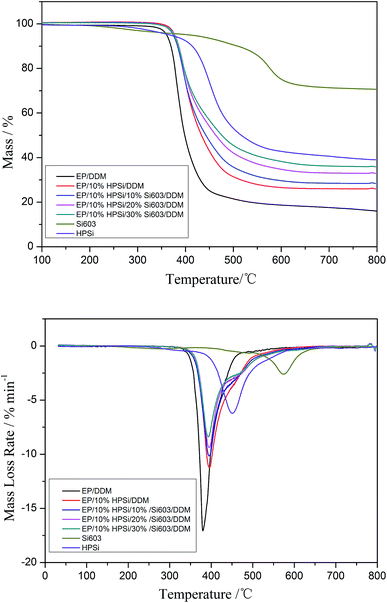
TG and DTG traces of the cured EP/HPSi/Si603/DDM systems provided additional information in regard to their thermal stability and thermal degradation behavior.20 The thermal degradation behavior of the various samples in a nitrogen atmosphere are shown in Fig. 5. The corresponding T5%, the Tmax, and char yield (Yc) at 800 °C are summarized in Table 2. T5% represents the temperature at which the mass loss is 5% for the system (the initial decomposition temperature), and T1max and T2max represent the temperature of the first maximum mass loss rate for the system and the second maximum mass loss rate for the system, respectively. Yc represents the char yield at 800 °C.| Sample | T5%/°C | R1peak/(% °C−1) | T1max/°C | Yc at 800 °C | |
|---|---|---|---|---|---|
| Cal. | Exp. | ||||
| EP/DDM | 350.8 | −17.07 | 379 | — | 16.01 |
| Si603 | 410 | −5.22 | 574 | — | 70 |
| HPSi | 368 | −6.22 | 460 | — | 39 |
| EP/10% HPSi/DDM | 377.5 | −11.24 | 396 | 18.1 | 25.99 |
| EP/10% HPSi/10% Si603/DDM | 375 | −10.16 | 395 | 21.34 | 28.5 |
| EP/10% HPSi/20% Si603/DDM | 376 | −9.39 | 395 | 26.08 | 32.94 |
| EP/10% HPSi/30% Si603/DDM | 376.2 | −8.39 | 393 | 29.22 | 35.88 |
It can be observed from Fig. 5 that all cured resins except HPSi have one-stage decomposition processes, demonstrating that the original and modified EP resins have similar degradation processes.
Fig. 5 shows that the char residue at 800 °C is 39% and the T5% and Tmax of cured HPSi are 368 °C and 457 °C, respectively. The degradation of HPSi in a nitrogen atmosphere has gone through two processes: unzipped degradation occurs before 400 °C, and silicone chain scission rearrangement follows.21 When terminal hydroxyl groups exist, a poorer heat resistance of the silicone resin is presented. As can be seen from the FTIR spectrum of HPSi (Fig. 1), HPSi contains a small amount of silicone hydroxy, which can initiate unzipped degradation through the backbiting reaction, which mainly produces low molecular weight annulations, and thus thermal weight loss happens. A small amount of unzipped degradation occurred during the degradation processes probably because HPSi contains a large number of branches, which greatly reduce the occurrence of unzipped degradation. At 457 °C, cured HPSi shows a Tmax, which is mainly caused by the cleavage and rearrangement reaction of Si–O–Si bonds.
TG shows that the addition of HPSi (10 wt%) enhances the thermal stability of the EP/DDM system: T5% and T1max of EP/HPSi/DDM are higher than that of EP/DDM. This phenomenon originates from the decomposition of HPSi at an inferior temperature (368 °C) that leads to the formation of the silicone-containing group, which will participate in the cross-linked carbonization. Moreover, the Si–O group of HPSi is able to absorb more thermal energy and its vibration could dissipate the thermal decomposition energy.22 However, with the increase of Si603 resin, T5% and T1max of the EP/HPSi/Si603/DDM systems are almost the same, while the char residue at 800 °C remarkably increases from 16.01% to 35.88% with Si603 content increasing from 0% to 30%. As discussed above, silicone resins have the ability to convert the usual organic decomposition to partial inorganic decomposition by forming a carbon–silicon residue which acts as thermal insulation11,12,23,24 to prevent gas evolution, and achieve ultimate improvement of the flame retardation of this silicone-containing epoxy system.
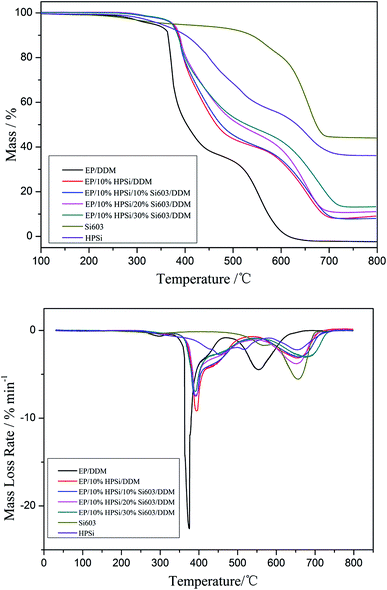
Fig. 6 depicts the TG and DTG curves of cured EP/DDM, HPSi and EP/HPSi/Si603/DDM resins in an air atmosphere and the corresponding T5%, the Tmax, and char yield (Yc) at 800 °C are summarized in Table 3.
| Sample | T5%/°C | R1peak/% °C−1 | T1max/°C | R2peak/% °C−1 | T2max/°C | Yc at 800 °C | |
|---|---|---|---|---|---|---|---|
| Cal. | Exp. | ||||||
| EP/DDM | 334 | −22.59 | 375 | −4.47 | 555 | — | 0 |
| Si603 | 380 | −5.43 | 566 | −4.06 | 656 | — | 44 |
| HPSi | 350 | −2.76 | 452 | −2.20 | 653 | — | 36.16 |
| EP/10% HPSi/DDM | 373.5 | −9.18 | 394 | −3.13 | 654 | 3.29 | 9.08 |
| EP/10% HPSi/10% Si603/DDM | 374 | −7.43 | 390 | −3.06 | 661 | 6.68 | 9.85 |
| EP/10% HPSi/20% Si603/DDM | 375 | −7.52 | 391 | −3.79 | 654 | 9.55 | 11.1 |
| EP/10% HPSi/30% Si603/DDM | 373 | −6.95 | 392 | −3.06 | 674 | 12 | 13.21 |
As can be seen, HPSi experiences three stages of degradation processes which occur at the temperature ranges of 300–510 °C, 510–600 °C and 600–750 °C in an air atmosphere, and the largest thermal degradation peaks appear at 452 °C, 540 °C and 653 °C. When the temperature reaches 350 °C, initial decomposition occurs and the char yield is 36.16% when the temperature reaches 800 °C. Thus, due to the presence of oxygen, the decomposition mechanism of cured HPSi has changed. For HPSi in an air atmosphere, thermo-oxidative degradation occurs, which includes the thermal degradation of the main chain and the oxidation reaction of the organic groups. As can be seen from the figure, before the main chain degradation (600 °C), two peaks appear, which are due to the thermo-oxidative degradation of the side groups generating small molecules such as carbon monoxide, carbon dioxide, water and so on.
For EP/DDM resin, bits of decompositions appear before 300 °C, which can be attributed to the decomposition of unstable alkyl groups in the hybrids. The chain scission of the isopropylidene linkage takes place, leading to the release of EP and its derivates at the initial decomposition stage.25 The thermal degradation of EP resin can be divided into two independent steps: the first thermal decomposition occurs between 362 and 470 °C, and the second stage is from 470 to 700 °C with 0% char residue at 800 °C, which demonstrates that pure EP shows poor charring ability.
The initial thermal decomposition of EP/HPSi/Si603/DDM systems, occurring between 350 and 600 °C, are wider than that of the EP/DDM resin, and the second stage is from 600 to 700 °C. What’s more, the Tmax of the EP/HPSi/Si603/DDM systems become higher, especially the T2max, with the content of Si603 resin increasing. The improvement can be attributed to silicon in the condensed phase which leads to the formation of a surface glassy char acting as a barrier to heat and mass transfer at high temperature. As is shown in Table 3, as the content of Si603 increases, the char residue of the overall system significantly enhances; when the amount of Si603 increases from 0% to 30%, the char residue of the EP/HPSi/Si603/DDM system combusted in an air atmosphere increases from 0% to 13.21%, indicating that HPSi enhances the thermal stability and char formation ability of EP. This is reasonable as we can see from the TG curve that the Si603 resin possesses high thermal stability. When it is over 400 °C, decomposition occurs in the Si603 resin and the solid char yield in nitrogen and air at 800 °C is 70% and 44%, respectively. It demonstrates that the charring ability and thermal stability of the Si603 resin is significantly higher than that of other resins.
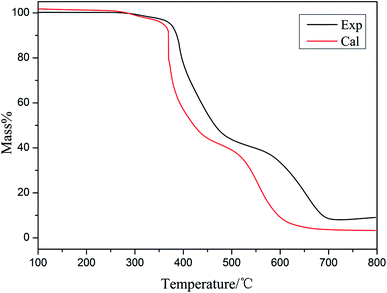
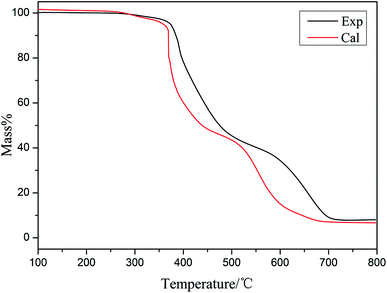
Fig. 7 and 8 show the experimental and theoretical TG curves of the EP/10% HPSi/30% Si603/DDM system in N2 and air, respectively. The theoretical curve was calculated based upon the mass percentage of the ingredient in the EP system. The formula is as follows:
| Mcal = wtHPSi% × MHPSiexp + wtEP% × MEPexp + wtSi603% × MSi603exp |
As can be seen in N2, when the temperature is below 350 °C, the experimental and theoretical curves are similar. However, the experimental mass of the residual char exceeds the theoretical one after 400 °C. The experimental and theoretical TG curves of the EP/HPSi/Si603/DDM system in air are different after 350 °C: the experimental mass of residual char surpasses the theoretical one in the whole temperature range and both decomposition stages occur at higher temperatures. It could be deduced that the addition of organic silicon obviously improves the mass of carbon residue and organic silicon generates a more stable substance at high temperatures, which effectively improves the char-formation ability and thermal stability of the matrix.
The higher char yield of the Si603 resin indicates that the carbonization mechanism indeed plays an important role in flame retardation. It is well known that the general flame retarding mechanism of organic silicone resin-based materials is thought to be the formation of a protective barrier during combustion. The improved flame retardancy can be explained by a flame retarding mechanism: providing a barrier for heat and mass transfer in the condensed phase, and at the same time preventing melted EP from burning.26
3.5. Structural analysis of the combustion residue by FTIR
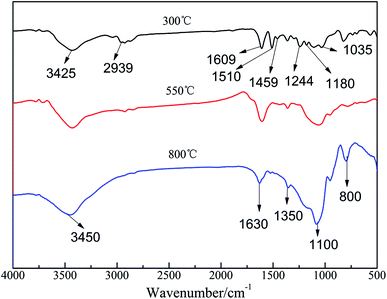
In order to understand the chemical alteration in the condensed phase of the EP/10% HPSi/30% Si603/DDM system, the combustion residue after thermogravimetry under an air atmosphere was investigated by FTIR. The FTIR spectra at different degradation temperatures (300, 550 and 800 °C) are shown in Fig. 9.At 300 °C, the absorptions of –OH (3450 cm−1), the stretching vibrations of –CH3 and –CH2– (2939 cm−1), C–C stretching vibrations of the aromatic ring (1609, 1510, 1459 cm−1), C–H vibrations of –C6H4–O–CH2– (1244, 1035 cm−1) and C–O stretching vibration (1180 cm−1) are the characteristic absorptions of pure epoxy resin.27 At 550 °C, it is found that the absorption peaks at 2939, 1609, 1459, 1244 and 1035 cm−1 disappear and then decrease in relative intensity. Of the aromatic components, the related bonds (1510 cm−1) and ether bonds (1180 and 1035 cm−1) were detected, indicating that the main decomposition happened in this stage, which is consistent with the TG results. According to the information shown, the absorption of aliphatic components and the C–C stretching vibration of the aromatic ring at 1510 cm−1 totally disappear at 800 °C. However, the bending vibration peak belonging to Si–O–Si groups centered at 1100 cm−1 becomes stronger and wider, and characteristic peaks belonging to the polyaromatic, centered at 800 cm−1 and 1630 cm−1 become broader, indicating the formation of polyaromatic carbons.
3.6. Morphology of the residue char
In order to explore how the structure of char determines the flame retardancy of EP, we investigated the residues of char after LOI testing by SEM. Fig. 10 presents SEM micrographs of char residue of EP/10% HPSi/30% Si603/DDM and EP/DDM. According to Fig. 10a, for cured EP, there are many big holes due to insufficient char formation or less condensed char during the burning process. This poor char quality could not effectively protect the underlying EP from degradation during combustion; therefore, EP can’t pass UL 94-V0. However, the char surface of EP with both HPSi and Si603, illustrated in Fig. 10b, is compact, smooth, and tight. This structure of the char for EP composites could prevent heat transfer between the flame Si603 and the substrate and thus protect the underlying materials from further burning and pyrolysis, which endows the material with much higher LOI values. In addition, this char structure could offer a good shield to prevent melted EP from burning, which was proved in vertical flammability tests (Table 1).3.7. Mechanical properties
Tensile strength is a measurement of the force required to pull a sample to the point where it breaks, and reflects the fracture resistance of materials. Flexural strength is usually used for evaluating the mechanical properties of a material because the flexural loading is very complicated and may contain multi-type loadings such as tensile, shearing and/or compressing loadings.28 Therefore, to evaluate the integrated mechanical properties of a material, flexural strength is usually selected as a typical parameter to evaluate the integrated mechanical properties. The flexural modulus reflects the inherent energy of a material, and the ability to resist strain. Moreover, it is also a property reflecting the stiffness of a material.29 Therefore, the flexural modulus is usually selected as a typical parameter to evaluate the stiffness of the materials.29,30The tensile strength, flexural strength and flexural modulus of EP/HPSi/Si603/DDM composites are compared and the results are presented in Table 4. Overall, a continuous reduction in flexural strength, tensile strength and flexural modulus were evident as the addition of Si603 increased. However, there is only an almost negligible decline in the tensile strength, flexural strength and flexural modulus of EP/10% HPi/10% Si603/DDM compared with that of the EP/DDM, which is attributed to the better compatibility of EP/Si603/DDM improved by HPSi and the improved homogeneity of the system to some extent offsetting the negative effect the Si603 brings to the mechanical property. As the addition of Si603 increases, the reduction in tensile strength, flexural strength and flexural modulus are observed. Owing to a large amount of unoccupied structure, a hyperbranched polymer usually has a higher average volume of free cavities that play a negative role in decreasing the concentration of the chain segment of modified resins, and the large number of rigid pendant groups (benzene rings) from Si603 decreased the flexibility of the compound provided by the siloxane linkages, hence the EP/HPSi/Si603/DDM with a large content of Si603 (>20 wt%) possesses decreased toughness. These results are in line with those in the literature, in detail, a suitable content of phosphorus-containing flame retardant will contribute a satisfactory flame retardancy to a resin, but meanwhile this usually brings a negative effect to the flexural strength of the resin system.31,32
| Specimen | Mechanical properties | ||
|---|---|---|---|
| Tensile strength/MPa | Flexural strength/MPa | Flexural modulus/MPa | |
| EP/DDM | 45.87 | 97.77 | 3579.6 |
| EP/10% HPi/10% Si603/DDM | 45.03 | 97.55 | 3553.5 |
| EP/10% HPSi/20% Si603/DDM | 40.76 | 89.57 | 3326.7 |
| EP/10% HPSi/30% Si603/DDM | 29.56 | 68.92 | 3182.6 |
4. Conclusions
A novel hyperbranched polysiloxane (HPSi) with epoxy groups was successfully synthesized as a compatibilizer by controlling hydrolysis and condensation between γ-(2,3-epoxypropoxy)propyltrimethoxysilane (KH560) and dimethoxydimethylsilane (DEMS). Experiments suggested that HPSi significantly improved the compatibility of the EP/Si603/DDM systems.A series of EP/10% HPSi/Si603/DDM hybrids containing 0 wt%, 10 wt%, 20 wt% and 30 wt% content of Si603 were prepared. The flame retardancy of the products was examined by an LOI measurement and UL-94 vertical test. The LOI value of EP/10% HPSi/30% Si603/DDM was 31, which was about 1.4 times that of the corresponding value of neat EP, and a V-1 grade for the UL-94 can be achieved. TG showed that the addition of HPSi (10 wt%) enhanced the thermal stability of the EP/DDM system.
The high silicon content and rich aromatic group structures of Si603 contributed an excellent flame retardancy to epoxy resins. Specifically, the char residues of the EP/10% HPSi/30% Si603/DDM system could reach 35.88% in N2 and 13.21% in air at 800 °C. Structural analysis of the combustion residue by FTIR indicated the formation of polyaromatic carbons which slowed heat and mass transfer between the gas and condensed phases. Based on this, the speculated degradation process of silicon resin was obtained. These properties would make this epoxy attractive for practical applications such as flame-retardant laminates.
Acknowledgements
We would like to thank the generous support of Sichuan Applied Basic Research Program and the Experiment Center of Polymer Science and Engineering Academy, Sichuan University.References
- R. R. Jones, Chapman & Hall, London, Chemistry and technology of epoxy resin, 1993, vol. 8, pp. 256–302 Search PubMed.
- M. Morell, X. Ramis, F. Ferrando, Y. F. Yu and A. Serra, Polymer, 2009, 50, 5374–5383 CrossRef CAS.
- B. Wetzel, F. Haupert and M. Qiu Zhang, Compos. Sci. Technol., 2003, 63, 2055–2067 CrossRef CAS.
- D. Rosu, C. N. Cascaval, F. Mustata and C. Ciobanu, Thermochim. Acta, 2002, 383, 119–127 CrossRef CAS.
- W. J. Wang, L. H. Perng, G. H. HPSiue and F. C. Chang, Polymer, 2000, 41, 6113 CrossRef CAS.
- G. H. HPSiue, W. J. Wang and F. C. Chang, J. Appl. Polym. Sci., 1999, 73, 1231 CrossRef.
- F. H. Gojny, M. H. G. Wichmann, B. Fiedler, I. A. Kinloch, W. Bauhofer, A. H. Windle and K. Schulte, Polymer, 2006, 47, 2036–2045 CrossRef CAS.
- S. D. Smith, T. E. Long and J. E. McGrath, J. Polym. Sci., Part A: Polym. Chem., 1994, 32, 1747–1753 CrossRef CAS.
- R. P. Kambour, H. J. Klipfer and S. A. Smith, J. Appl. Polym. Sci., 1981, 26, 847 CrossRef CAS.
- S. T. Lin and S. K. Huang, J. Polym. Res., 1994, 1, 151 CrossRef CAS.
- X. Wang, Y. Hu, L. Song, W. Y. Xing and H. D. Lu, J. Polym. Sci., Part B: Polym. Phys., 2010, 48, 693–705 CrossRef CAS.
- C. L. Chiang and C. C. M. Ma, Polym. Degrad. Stab., 2004, 83, 207 CrossRef CAS.
- S. Mahapatra and K. N. Singha, J. Mater. Sci., 2009, 46, 296–303 CAS.
- D. Zhuo, A. Gu, G. Liang, J. Hu, L. Yuan and X. Chen, J. Mater. Chem., 2011, 21, 6584–6594 RSC.
- J. Ye, G. Liang, G. Aijuan and Z. Zhang, Polym. Degrad. Stab., 2013, 98, 597–608 CrossRef CAS.
- G. B. Zhang, X. D. Fan, Y. Y. Liu, J. Kong and S. J. Wang, J. Polym. Environ., 2007, 7, 644–646 Search PubMed.
- S. A. Torry, A. Campbell, A. V. Cunliffe and D. A. Tod, Int. J. Adhes. Adhes., 2006, 26, 40–49 CrossRef CAS.
- W. J. Wang, L. H. Perng, G. H. HPSiue and F. C. Chang, Polymer, 2000, 41, 6113–6122 CrossRef CAS.
- Y. Ling Liu, Y. Chan Chiu and C. S. Wu, J. Appl. Polym. Sci., 2003, 87, 404–411 CrossRef.
- Z. Tang, Y. Li and Y. J. Zhang, Polym. Degrad. Stab., 2012, 97(4), 638–644 CrossRef CAS.
- T. T. Howard and T. C. Kenick, J. Polym. Sci., Part B: Polym. Phys., 1969, 7, 537–549 CrossRef.
- J. E. Curry and J. D. Byrd, J. Appl. Polym. Sci., 1965, 9, 295 CrossRef CAS.
- H. Kanai, V. Sullivan and A. Auerback, J. Appl. Polym. Sci., 1994, 53, 527 CrossRef CAS.
- R. P. Kambour, W. V. Ligon and R. P. Russell, J. Polym. Sci., Polym. Chem. Ed., 1978, 16, 327 CrossRef CAS.
- C. S. Wu, Y. L. Liu and Y. S. Chiu, Polymer, 2002, 43, 4277–4284 CrossRef CAS.
- R. P. Kambour, J. Appl. Polym. Sci., 1981, 26, 861 CrossRef CAS.
- X. Wang, Y. Hu, L. Song, W. Y. Xing, H. D. Lu and P. Lv, Polymer, 2010, 51, 2435–2445 CrossRef CAS.
- S. Xavier and A. Misra, Polym. Compos., 1985, 6, 93–99 CrossRef CAS.
- C. Karthikeyan and S. Sankaran, Polym. Adv. Technol., 2007, 18, 254–256 CrossRef CAS.
- Q. Shao and P. Lee-Sullivan, Polym. Test., 2000, 19, 239–250 CrossRef CAS.
- Z. K. Chen, G. Yang, J. P. Yang, S. Y. Fu, L. Ye and Y. G. Huang, Polymer, 2009, 50, 1316–1323 CrossRef CAS.
- B. Li and J. He, Polym. Degrad. Stab., 2004, 83, 241–246 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2015 |